Research Article - (2025) Volume 4, Issue 1
Comparison of Ammonia and Water Vapor Steam Distillation for Thin Layer Chromatography of Essential Oils from Rosmarinus Officinalis, Salvia Officinalis, and Lavandula Species
Received Date: Jan 24, 2025 / Accepted Date: Feb 27, 2025 / Published Date: Mar 03, 2025
Copyright: ©Â©2025 Alessandro Careglio Frazione. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Frazione, A. C. Comparison of Ammonia and Water Vapor Steam Distillation for Thin Layer Chromatography of Essential Oils from Rosmarinus Officinalis, Salvia Officinalis, and Lavandula Species. Int J Clin Med Edu Res, 4(1), 01-06.
Abstract
This study project investigates the extraction of essential oils (E.O.) from lavender, sage, and rosemary using ammonia vapor generated by the reaction of ammonium sulfate, ammonium phosphate, and sodium hydroxide with water vapor. This type of extraction could represent a new way to extract volatile plant compounds, presenting some advantages such as a higher yield and a greater selectivity towards apolar compounds compared to polar ones, such as camphor, which can decrease the sensory properties or eventually thujone due to its toxicity. The essential oils are compared using thin layer chromatography.
For rosemary and lavender, there is a higher yield of essential oil and an improvement in sensory characteristics (subjective opinion).
From the chromatographic analysis, the extraction with ammonia does not show the evident formation of compounds different from the extraction with water, so there should be no risk of potential toxicity. Furthermore, ammonia is not soluble in essential oils and remains in the mother liquors.
Keywords
Essential Oils, Thin Layer Chromatography, Ammonia Vapor Distillation
Introduction
This study uses a solution of sodium hydroxide (NaOH) with ammonium sulfate ((NH4)2SO4) and ammonium phosphate ((NH4)3PO4).
The reaction between sodium hydroxide (NaOH) and ammonium sulfate (and phosphate) ((NH4)2SO4) generates ammonia (NH3), water (H2O), and sodium sulfate (Na2SO4). The complete chemical reaction is as follows:
2 NaOH + (NH4)2SO4 → 2 NH3 + Na2SO4 + 2 H2O
In this reaction, sodium hydroxide acts as a strong base and ammonium sulfate as an acidic salt. Ammonia is a basic gas that is produced by the deprotonation of the ammonium ion (NH4+) by sodium hydroxide. Sodium sulfate is a neutral salt that remains in solution and creates an ebullioscopic rise, resulting in an increase in vapor temperature and improved extraction yield.
Ammonia in its vapor state takes on the characteristics of a solvent with a polarity represented by the molecular dipole moment of 1.42D, which is lower than that of water (1.85D) [5,4].
A molecular dipole is the electric dipole present in a molecule, due to the non-uniform distribution of electric charge. The dipole moment represents the vector sum of all the bond moments present [6].
As a consequence, apolar compounds are extracted to a greater extent, as evidenced by the size of the spots near the solvent front in the figures below.
Materials and Methods
Distillation
The fresh flowering tops of rosemary (Rosmarinus oficinalis) and sage (Salvia oficinalis) are manually cut from the plants, while the dried inflorescences of lavender (Lavandula hybrid genus) are used.
Source of plant material: Careglio farm
The plants are distilled in a 30-liter stainless steel still with a grid at the bottom to prevent the formation of foam that would impede distillation.
Steam distillation is carried out by adding tap water in the quantities indicated in the table at the bottom of the still, loading the still with the herbs (quantity indicated in the table) and heating, in my case with a LPG flame, cooling the vapors with tap water.
Ammonia vapors are generated by adding water and salts in the quantities indicated in the table to the still, separating them from the herbs with a metal mesh to prevent the formation of foam. A slightly higher than stoichiometric amount of caustic soda is used.
|
Plant name |
Rosm OE W |
Sage OE W |
Lav OE W |
Rosm OE Amm |
Sal OE Amm |
Lav OE Amm |
|
plant weight kg |
1,5 |
1 |
1 |
1,25 |
1,25 |
0,6 |
|
water weight kg |
3 |
2,5 |
5 |
2,5 |
2,5 |
2,5 |
|
sulfate weight g |
0 |
0 |
0 |
50 |
50 |
50 |
|
phosphate weight g |
0 |
0 |
0 |
50 |
50 |
0 |
|
soda weight g |
0 |
0 |
0 |
100 |
100 |
50 |
|
ml EO obtained |
4 |
1 |
14 |
6 |
1 |
10 |
Table 1: Quantity of Water, Plant and Salts used and Essential Oil obtained in Distillations.
TLC plates 10X5 silica gel on glass (Fluka) are used. Fluorescent at 254 nanometers.
An eluent solution of toluene-ethyl acetate (97:3) is prepared.
1 ml of essential oil is diluted in 9 ml of toluene and 40 microliters are spotted on each run with a microsyringe.
Images are taken with the tablet camera under 254 nanometer Wood's lamp illumination.
Detection with vanillin-sulfuric acid (VS) reagent 1% ethanolic vanillin (solution I). 10% ethanolic sulfuric acid (solution II).
The plate is sprayed with 10 ml of solution I, followed immediately by 10 ml of solution II. After heating at 110°C for 5-10 min under observation, the plate is evaluated at visible light.
Detection e.g. components of essential oils (terpenoids, phenylpropanoids) [3].
Results
Essential Oil Yield
Ammoniacal steam distillation yielded a higher quantity of essential oil compared to traditional distillation, except for sage which showed no variation, as shown in figure 1.
Figure 1: Comparison of Yield in Distillations.
Thin Layer Chromatography Results:
TLC Lavender E.O.
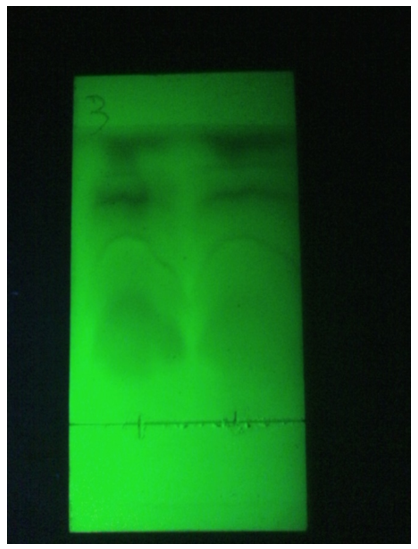
Figure 2: Chromatographic comparison between Lavender Essential Oil with Water Vapors (run 1) and Ammonia Vapors (run 2), a larger spot is noted on the solvent front which indicates a greater quantity of Apolar compounds extracted.

Figure 3: Chromatographic comparison between Lavender Essential Oil with Water Vapors (run 1) and Ammonia Vapors (run 2), detection with Vanillin-Sulfuric Acid, the plate is evaluated in visible light.
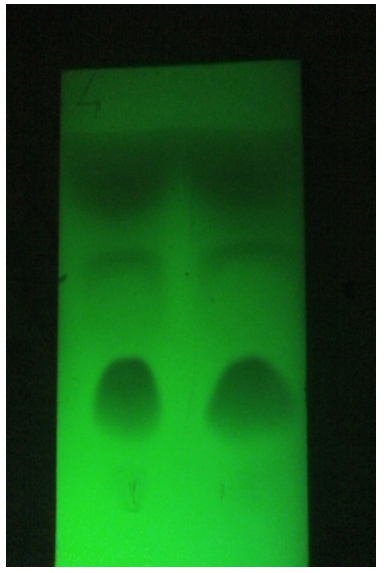
Figure 4: Chromatographic comparison between Rosemary Essential Oil with Water Vapors (run 1) and Ammonia Vapors (run 2), a larger spot is noted on the solvent front which indicates a greater quantity of Apolar compounds extracted.
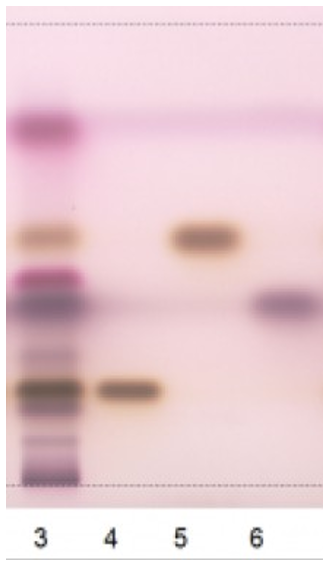
Figure 5: Chromatography with Rosemary Essential Oil (run 3), referred to Borneol (run 4) and Bornyl Acetate (run 5) Cineol (run 6) [1].
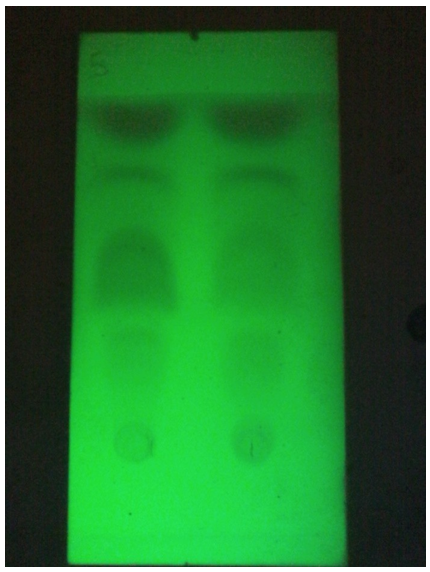
Figure 7: Chromatographic comparison between Sage Essential Oil with Water Vapors (run 1) and Ammonia Vapors (run 2), there are no visibly evident differences between the two types of extraction.
Conclusion
Essential oils extracted with ammonia vapors and steam are compared using thin layer chromatography, resulting in an improved organoleptic profile of the EOs (subjective opinion).
For example, rosemary EO extracted with ammonia vapors acquires incense-like notes because the more volatile compounds (pinene, camphene, limonene) are extracted to a greater extent, as evidenced by the size of the spots in the TLC (Fig. 4) compared to the data on composition and retention times of Szumny et al. [1].
An increase in yield was obtained for lavender and rosemary oils (ammonia vapors), while for sage there were no evident changes in the chromatographic profile but the scent was better (subjective opinion).
This type of analysis, although not extremely precise, has the function of preliminary analysis and does not clearly show the formation of new compounds, as far as the possible use of this application in the commercial field is concerned. Further investigations would be desirable.
An advantage of this type of extraction could be in the use of essential oils as a transdermal drug delivery vehicle, as the more apolar compounds better overcome the epidermal membranes [2].
Laboratory Notes
Draft of a Study Project by Alessandro Careglio. Safety Notes If you repeat the distillation carried out in this study, pay attention to the following:
• The reaction between sodium hydroxide and ammonium sulfate can be dangerous if not performed correctly.
• Sodium hydroxide is a strong base that can cause chemical burns.
• Ammonia is an irritating gas that can cause respiratory problems.
It is important to wear safety glasses, gloves, and a lab coat when performing this reaction. It is also important to work in a well-ventilated area to avoid inhaling ammonia.
It is important to be aware of the potential hazards of this experiment and to take appropriate safety precautions.
Conflict of Interest
There are no conflicts of interest.
Funding
The study project is self-financed.
References
- Szumny, A., Figiel, A., Gutiérrez-Ortíz, A., & Carbonell- Barrachina, Á. A. (2010). Composition of rosemary essential oil (Rosmarinus officinalis) as affected by drying method. Journal of Food engineering, 97(2), 253-260.
- Caliskan, U. K., & Karakus, M. M. (2020). Essential oils as skin permeation boosters and their predicted effect mechanisms. Journal of Dermatology and Skin Science, 2(3).
- Wagner, H., & Bladt, S. (2009). Plant drug analysis (eBooked.). Springer Berlin Heidelberg.
- https://en.wikipedia.org/wiki/Ammonia
- https://it.wikipedia.org/wiki/Dipolo_molecolare
- https://www.scienzeascuola.it/lezioni/legami-chimici/il-momento-dipolare



