Case Report - (2023) Volume 2, Issue 1
Clear Cell Carcinoma of The Sublingual Salivary Gland: A Case Report and Literature Review.
2Alexandria University Medical college, Egypt
Received Date: Sep 14, 2023 / Accepted Date: Oct 16, 2023 / Published Date: Oct 18, 2023
Copyright: ©©2023 Alaa Abdulrahim, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Abdulrahim, A., AlEnezi, A., Abushara, k., Zahran, M. (2023). Clear Cell Carcinoma of The Sublingual Salivary Gland: A Case Report and Literature Review. World J Otolaryngol Res, 2(1), 01-04.
Abstract
Clear Cell Carcinoma (CCC) is a rare type of neoplasm that affecting minor salivary glands. In this article, we present a rare case of clear cell carcinoma that affects the sublingual region.
Furthermore, the anatomy of the salivary glands, etiologies of CCC, and how to approach CCC will be highlighted.
Introduction
In head and neck region, tumors associated with salivary gland are relatively uncommon, affecting predominantly major sal- ivary glands. Minor salivary gland neoplasm is rare accounting for 9-23% of all salivary gland neoplasms, out of which clear cell carcinoma (CCC) accounts for just 0.2%. This malignancy was first described by Milchgrub and col- leagues3 in 1994 as a minor salivary gland carcinoma made up of clear cells forming cords and nests in a hyalinized stroma [1]. Here we will present a case of clear cell carcinoma in the right sub-lingual area in Kuwait.
Case Report
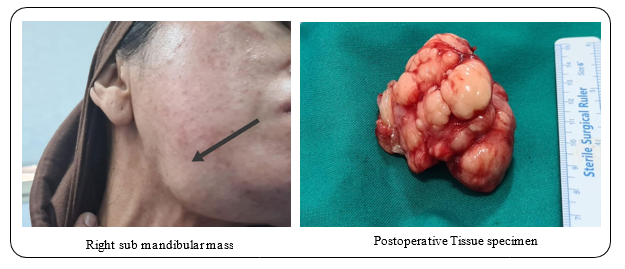
A 43-year-old female patient, previously healthy, presented to the out-patient clinic with 4 months history of right sub-lingual swell- ing, that gradually increase in size. Associated with restriction in opening the mouth. On physical examination, she was conscious, alert, and fully oriented. Her vital signs were within normal range. She had right sub-lingual swelling, which is about 5x5x2 cm in size. The facial sensation was intact. No palpable lymph nodes in cervical and auricular region. Laboratory studies showed white count level of 6.7x109 /L , a hemoglobin level of 120 g/L, creat- inine level of 47 mmol/ L, sodium of 134 mmol/L and potassium of 3.7 mmol/L. CT Neck was done and showed A sizable mass lesion is seen involving the right sublingual and submandibular spaces (right side of floor of the mouth), showing the following CT criteria: It is seen located just anterior to the right submandibular gland, displacing it posteriorly, however being inseparable from it at a certain point within its superior part with suggested areas of glandular invasion. There is prime involvement of the right sublin- gual space with infiltrative medial border, medially displacing and being inseparable from the right genioglossus muscle. Inferiorly, it extends medially involving and towards the midline displacing the right anterior belly of digastric muscle with no separable cleavage plane. There is encroachment upon the right anterolateral aspect of the oropharynx, which appears relatively narrowed. It is measur- ing about 5 x 5 x 2.5 cm in maximum AP, CC & TR dimensions. It shows heterogeneous pattern of contrast enhancement pattern with multiple internal cystic-like areas, denoting internal braking down/ degenerative necrosis. The right body of the mandible presents fo- cal area of inner cortical rarefaction with suspected altered-tex- tured lytic intra-medullary involvement.
Thus, the patient underwent mass excision under general anesthe- sia. The operation started by an incision 2 fingers below the free border of the right mandible, the mass was dissected from the sur- rounding structures and delivered from the oral cavity, through the sublingual site. The mass was send for histopathology. Post-oper- atively the patient was doing well, kept on IV antibiotic (klacid), and IV dexamethasone for 10 days, then discharged on oral klacid for one more week. PET scan done two months after the operation and showed: No evidence to suggest metabolic disease involve- ment (local/ distant). The histopathology result showed Pathologic stage: pT2 with Additional findings of: The tumor shows interven- ing areas of hyalinization, edema and focal necrosis, along with superficial ulceration of squamous mucosa.The tumor is positive for EWSR1 (22q12) gene. Later, the patient was following with Kuwait Cancer Center for adjuvant therapy.
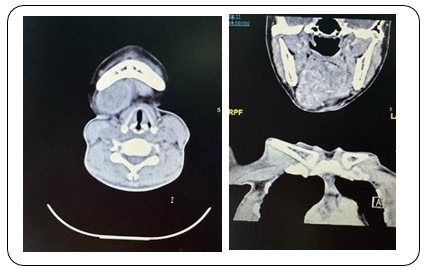
Right floor of the mouth mass lesion seen within sublingual and submandibular spaces showing extension, features and mass effect,mea- suring about 5 x 5 x 2.5 cm in maximum ap, cc & tr dimensions.
Discussion
Anatomy
The salivary glands are exocrine glands. They are divided into two main types: the major salivary glands, which include the parot- id, submandibular and sublingual glands, and the minor salivary glands, which line the mucosa of the upper aerodigestive tract and the overwhelming entirety of the oral cavity [2]. The sublingual gland (SLG) is the smallest of the three major salivary glands [3]. lies beneath the mucosa of the floor of the oral cavity and superior to the mylohyoid muscle. it lies between the muscles of the floor of the oral cavity which include the geniohyoid muscle, hyoglossus muscle medially, and the mylohyoid muscle inferiorly. Medially, between the base of the tongue and the sublingual gland, the sub- mandibular duct, and the sublingual nerve can be found. Rather than having one main duct, it contains a series of short ducts that project directly into the floor of the oral cavity, the ducts of Rivi- nus, and a common duct, known as Bartholin's duct that connects with the submandibular gland's duct at the sublingual caruncula [2]. The sublingual glands receive their parasympathetic input via the chorda tympani nerve, which is a branch of the facial nerve via the submandibular ganglion. The nerve functions in a secretomo- tor capacity. The chorda tympani branches from the motor branch of the facial nerve in the middle ear cavity, which then exits the middle ear through the petrotympanic fissure. The chorda tympani nerve then travels with the lingual nerve to synapse at the subman- dibular ganglion [3].
Physiology
The sublingual tissue is predominantly a mucous gland, however, is considered a mixed serous and mucous gland. It is made up of mainly mucous acini with serous demilunes. It is the only unen- capsulated major salivary gland. Sublingual tissue primarily pro- duces a thick mucinous fluid and lubricates the oral cavity which allows for swallowing, initiating digestion, buffering pH, and den- tal hygiene [3]. The composition of saliva depends on the salivary flow rate and can vary given each gland's flow rate and overall contribution. Saliva is comprised of both organic and inorganic components. Inorganic components include electrolytes, urea, and ammonia. The organic elements of saliva contain immunoglobu- lins, enzymes, and proteins [3].
Pathogenesis
Histologically, typical CCCs comprised bland polygonal or round cells with clear cytoplasm. In contrast with typical CCCs, HGT tumor cells exhibited nuclear pleomorphism, high nuclear-to- cy- toplasmic ratios, high mitotic activity, and necrosis. Rare morpho- logic features such as pseudo papillae, gland-like spaces, and en- trapped ducts were also observed. Occasionally, tumors involving the oral cavity might arise from the overlying epithelium of the mucosal surface. Immunohistochemically, all the cases expressed p63, p40, and CK5/6, while myoepithelial-related markers were uniformly negative in all cases. HGT (high grade transformation) exhibited a wild type p53 expression pattern. FISH demonstrated EWSR1 rearrangement (10/10) and EWSR1–ATF1 fusion (4/5); however, MAML2 remained intact (0/3) [4].
Etiology
Clear cell tumors of the oral mucosa, jaws, and salivary glands constitute a heterogeneous group of lesions which may be either odontogenic, salivary gland, or metastatic in origin [5]. Clear cell myoepithelial carcinoma shows sheets of clear cells sometimes admixed with spindle shaped and other myoepithelial cells. These cells are positive for cytokeratins, S-100 and actin (alpha smooth muscle actin). The tumor cells are also positive for glycogen. Clear-cell variants of acinic cell carcinoma are usually never pure and cells with periodic acid Schiff positive and diastase resistant cytoplasmic zymogen granules are also present. Because of the lack of awareness, CCC is often misdiagnosed as poorly differen- tiated carcinoma, squamous cell carcinoma, acinic cell carcinoma, mucoepidermoid carcinoma and EMEC [6].
Clinical Features and Presentation
These tumors occur in patients in the 22-63-year age range with no sex predilection. They have indolent clinical courses and the history may go back many months or even years. They are at low risk of lymph node and distant metastasis [7]. Patients may present with a long history of facial pain or numbness along the affected side, on examination it may show a trigeminal neuralgia.
Differential Diagnosis
By definition CCC contains a significant proportion of clear cells, but does not fit into any other recognized neoplastic entities. When clear cells predominate, along with salivary gland tissue, definitive diagnosis may be problematic as the differential diagnosis includes various clear cell variant of primary tumor of salivary gland. The microscopic differential diagnosis of primary CCC of salivary gland includes mucoepidermoid carcinoma, acinic cell carcino- ma, myoepithelial carcinoma, epithelial-myoepithelial carcinoma, and oncocytoma. In present reported case due to the presence of clear cells along with trace of minor salivary gland t issues, various other histopathological differential diagnoses such as mucoepider- moid carcinoma and clear cell odontogenic carcinoma were also included [1].
Treatment
Because of the rarity of these tumors, there are no adequate clinical trials that define the optimal therapeutic approach. Surgery is the mainstay of treatment of minor salivary gland tumors. Data on the efficacy of chemoradiotherapy are limited. There are no specific recommendations regarding the site of origin. Surgery +/- postop- erative irradiation was preferred [7].
Conclusion
Clear cell carcinoma in the head and neck region associated with the salivary glands are considered rare , which may be odonto- genic, metastatic or of salivary gland origin. Tumors occur in pa- tients in the 22-63-year age range with no sex predilection. They have indolent clinical courses and the history may go back many months or even years. Surgery is the mainstay of treatment of sal- ivary gland tumors. Surgery +/- postoperative irradiation with a regular follow up with cancer center is required.
Consent
Informed consent was taken from the patient before and after sur- gery.
Conflict of interest
The authors declare that there are no conflicts of interest regarding the publication of this article.
References
- Agrawal, J., Kumar, Y. P., Damera, D. A., & Kumar, S. (2014). Clear cell carcinoma of minor salivary gland: A case of clini- cal dilemma. Contemporary Clinical Dentistry, 5(3), 389.
- Ghannam, M. G., & Singh, P. (2019). Anatomy, head and neck, salivary glands.
- Grewal, J. S., Bordoni, B., Shah, J., & Ryan, J. (2018). Anato- my, head and neck, sublingual gland.
- Xuan, L., Wang, S., Wei, J., Yuan, J., & Liu, H. (2022). Clini- copathological and molecular study of 10 salivary gland clear cell carcinomas, with emphasis on rare cases with high grade transformation and occurring in uncommon sites. Diagnostic Pathology, 17(1), 18.
- Maiorano, E., Altini, M., & Favia, G. (1997, August). Clear cell tumors of the salivary glands, jaws, and oral mucosa. In Seminars in diagnostic pathology (Vol. 14, No. 3, pp. 203- 212).
- Rodríguez, M. S., Reija, M. F. G., & Rodilla, I. G. (2013).Primary clear cell carcinoma of parotid gland: Case report and review of literature. Journal of oral and maxillofacial patholo- gy: JOMFP, 17(1), 101.
- Arifi, S., Hammas, N., & Erraisse, M. A. (2022). Clear Cell Carcinoma of Minor Salivary Glands: The Nasopharynx, an Uncommon Site of Origin. Cureus, 14(5).




