Research Article - (2026) Volume 7, Issue 1
Characterization of A Novel Polyfunctional “Metalloglyco-Protein/Polypeptide Organochlorine” Bioflocculant Containing Saturated Nitro Compounds, Sulfones, Polysulfides, Phosphorus Chlorine Compounds, Magnesium Oxide, and Metal Chlorides, Produced from Pseudomonas aeruginosa Strain F29, Isolated from Porcine Feces in Nigeria
Received Date: Dec 17, 2025 / Accepted Date: Jan 05, 2026 / Published Date: Jan 21, 2026
Copyright: ©2026 I. K. M. Okorie, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Okorie, I. K. M., Ogunjobi, A. A. (2026). Characterization of A Novel Polyfunctional â??Metalloglyco-Protein/ Polypeptide Organochlorineâ? Bioflocculant Containing Saturated Nitro Compounds, Sulfones, Polysulfides, Phosphorus Chlorine Compounds, Magnesium Oxide, and Metal Chlorides, Produced from Pseudomonas aeruginosa Strain F29, Isolated from Porcine Feces in Nigeria. J Pla Che Pla Pro Res, 7(1), 01-13
Abstract
Antibiotic resistance has reached universal proportions, and the discovery of effective alternatives to the common antibiotics currently used, could aid in solving this problem. The aim of this study was to characterize a bioflocculant produced from Pseudomonas aeruginosa strain F29, accession number OQ734844, that exhibited effective antibacterial activity against two antibiotic resistant bacteria, viz, Staphylococcus aureus SO183, and an identified strain of Pseudomonas aeruginosa, in another study. FTIR detected saturated nitro compounds, sulfones, polysulfides, phosphorus- chlorine bonds, magnesium-oxide bonds and metal-chloride bonds. FTIR also detected the following functional groups: carboxyl, amide/peptide, aromatic alcohol, alkene, and halo. SEM showed a clumped and flaky bioflocculant surface, while EDX detected chlorine (56.00%), carbon (20.50%), sodium (12.50%), oxygen (4.00%), phosphorus (3.00%), sulfur (2.43%) magnesium (1.06%), potassium (0.32%) and nitrogen (0.30%). HPLC and MS detected varied peaks of glucose, galactose, inositol and mannose, D-ribose, arabinose, rhamnose and xylose. The phenol sulfuric acid method calculated the concentration of these sugars as 0.0059 g/L. The bioflocculant is a polymeric compound composed mainly of carbohydrates, proteins/polypeptides and organochlorines, possibly a novel “metalloglyco- protein/ polypeptide organochlorine” bioflocculant. The presence of the metals: sodium, potassium and magnesium; the nonmetals: phosphorus, sulfur and nitrogen; and multiple moieties, likely contributed to the antibacterial activity of the bioflocculant produced from Pseudomonas aeruginosa strain F29. From available documentation, this is the first report of a polyfunctional “metalloglyco-protein/polypeptide organochlorine” bioflocculant, that naturally contains saturated nitro compounds, sulfones, polysulfides, phosphorus chlorine compounds, magnesium oxide and metal chlorides; and of a bioflocculant produced from porcine feces in Africa.
Keywords
Characterization, Novel “Metalloglyco-Protein/Polypeptide Organochlorine” Bioflocculant, Polyfunctional, Pseudomonas aeruginosa Strain F29, Antibiotic Resistance
Introduction
Antibiotic resistance has reached universal proportions, and the discovery of effective alternatives to the common antibiotics currently used, could aid in solving this problem. Antibiotic resistance occurs when bacteria are no longer susceptible to the usual drugs designed to kill them (bactericidal antibiotics) or to retard their growth (bacteriostatic antibiotics). Antibiotic resistance is the main subdivision of the larger group of antimicrobial resistance (AMR), and is defined as the development, by bacteria, of resistance to specific drugs invented to kill them, or to repress their growth [1]. Alternative antibacterial agents are therefore being sought. It has been reported that bioflocculants possess bioactivities against bacteria and certain other groups of microorganisms [2].
Bioflocculants are biological flocculants and are therefore, more generally defined as flocculants of organic origins and not solely microbial [3]. Bioflocculants, can be obtained from microorganisms, animals and vegetation [4]. With respect to microorganisms, bioflocculants are extracellular polymeric substances that are produced as secondary metabolites and cause flocculation in a given medium [5]. These microbial bioflocculants are a result of cytolysis and the release of cellular products by different groups of microbes [6]. Bioflocculants aggregate particles and then remove these particles, from the liquid medium in which they were suspended. On the whole, flocculants have the capacity to clump small-sized substances suspended in a medium; these clumped substances are termed flocs, and sediment over time [7].
The specific mechanism of action, by which bioflocculants operate, is still unknown. A bioflocculant may trigger the construction of a bridge between particles of the medium and its own molecules; or may kick-start a neutralization of charges present in the medium [8]. Other hypotheses exist, nevertheless, the precise mechanism of bioflocculant antibacterial bioactivity, may be deduced from the inherent capacity of bioflocculants to form flocs, that later sediment. A bioflocculant may therefore, initiate the clumping together of the cell wall components of the bacterium being acted upon, and this could more likely, produce tears in the bacterial cell membrane, with varying degrees of fragmentation. The compromise in bacterial cell membrane integrity that ensues, if progressive and widespread enough, could trigger an influx of noxious substances from the surroundings, that could lead to a destabilization of the internal milieu of the bacterial cell, and result in bacterial cell lysis and death.
Bioflocculants are made up of natural compounds such as cellulose, nucleic acids, glycoproteins and polysaccharides, that render them readily decomposable and less injurious to the environment; and, this therefore, obviates secondary environmental contamination [6]. Microbial bioflocculants are polymeric substances produced during the microbial growth phase [9]. Plant-derived bioflocculants include mucilage, alginate, starch and cellulose [10-12]. There are, however, different kinds of bacterial species, that also produce cellulose (here known as bacterial or microbial cellulose), that provides mechanical support, and possesses similar bioflocculant characteristics as plant cellulose [13]. Animal- derived bioflocculants include gelatin and chitosan [14].
Bioflocculants composed mainly of proteins, are easily affected by thermal fluctuations because proteins naturally lose their spatial conformations when exposed to external forces such as thermal energy [15]. The nucleic acids (deoxyribonucleic acid (DNA) and ribonucleic acid (RNA)), present in bioflocculants, constitute part of the larger pool of environmental nucleic acids; indeed, their presence in the environment is being utilized, now more than previously, in “molecular biomonitoring” [16]. Glycoprotein bioflocculants are characterized by the presence of oxygen, nitrogen and carbon in their framework; these elements are thought to boost their inherent bioflocculating capacities [9].
Bioflocculants largely composed of polysaccharides, retain their activities at high temperatures [17]. It has been documented that the flocculating capacity of any flocculant is directly proportional to the stretch of monomeric subunits it possesses; therefore, flocculants with higher molar masses are said to possess a commensurate degree of stretch and can even house unbound moieties, that can link flocs, to bring about better floc formation [18,19]. Apart from their use as antibiotics, bioflocculants can be effective against algae and viruses [2]. Bioflocculants that are primarily composed of polysaccharides, can substantially protect cells against free radical injury [20]. Bioflocculants can also be used as stabilizers, thickeners and emulgents, in the food industry and in drug manufacturing [21]. Some exopolysaccharides can demonstrate flocculating properties such as the exopolysaccharide, EPS SM9913, produced from the blue-water, cryophilic bacterium, Pseudoalteromonas sp. SM9913 [22]. Exopolysaccharides that double as bioflocculants, can therefore be utilized as flocculants for wastewater management, in addition to other uses that stem from the properties they possess as exopolysaccharides [22]. The functional moieties of a bioflocculant determine such characteristics as its durability, versatility and flocculating capacity [23]. The hydroxyl, carboxyl and amide functional moieties of a bioflocculant promote the flocculation process, by providing sites of attachment for the bivalent positively-charged ions that principally partake in flocculation [24,25]. Additionally, certain microorganisms can assimilate metals into the bioflocculants they produce, and these metal components affect the intrinsic chemical composition and morphology, and the mechanisms of actions of the bioflocculant [26].
The aim of this study was to characterize a bioflocculant that was produced from Pseudomonas aeruginosa strain F29, accession number OQ734844, that exhibited effective antibacterial activity against two antibiotic resistant bacteria, viz, Staphylococcus aureus SO183, and an identified strain of Pseudomonas aeruginosa, in another study that was part of a Master’s project [27]. This characterization was necessary in order to identify the physical nature and the chemical composition of the bioflocculant. The bacterial DNA sequence was deposited in the National Centre for Biotechnology Information (NCBI) GenBank database under its accession number. FTIR, SEM coupled to EDX, HPLC coupled to MS and the phenolâ??sulfuric acid method, were employed as analytic tools.
Materials and Methods
An aliquot of the bioflocculant was transferred into a sterile bottle and subjected to some analytical procedures to determine its elemental composition, functional groups and physical characteristics. The analytical tools used, were Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM) coupled to energy dispersive X-ray spectroscopy (EDX), high-performance liquid chromatography (HPLC) coupled to mass spectrometry (MS) and the phenolâ??sulfuric acid method.
Fourier Transform Infrared (FTIR) Spectroscopy
The functional groups present in the bioflocculant were characterized using a Perkin Elmer Spectrum 100 Series 3000 MX spectrometer. A set of instructions for FTIR, from the NanoScience Technology Centre (NTSC), was used as a reference point [28]. The bioflocculant sample was thinned between two potassium bromide (KBr) plates for FTIR analysis, and then was placed inside the sample holder of the FTIR device, that was wiped clean with acetone. The addition of relevant computer commands, enabled the recording of the generated infrared spectra. Next, the data from these spectra was analyzed using the spectroscopic software Win-IR Pro Version 3.0, and saved. The interpretation of the infrared (IR) spectra was performed using Infrared and Raman Characteristic Group Frequencies from Socrates: IR Spectra Table and Charts from Sigma Aldrich Company; and FTIR results analyses from Aksnes and Aksnes, Aboul-eneÃÂ?±n et al., Smith, Nandiyanto et al., Ji et al. and Agyapong et al. [29-36].
Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray Analysis/Spectroscopy (EDS/EDX)
The surface topography of the bioflocculant was revealed using a JEOL JSM-7600F scanning electron microscope (SEM) (Tokyo, Japan), while its elemental composition was determined using an in-built energy dispersive X-ray (EDX) spectrometer.
The protocol laid out by Core Facilities for preparing an SEM sample was used as a reference [37]. Primary fixation of a measured volume of the bioflocculant sample was performed using formaldehyde and glutaraldehyde, while secondary fixation was performed with osmium tetroxide. This step was followed by dehydration with ethanol and sample drying. Mounting of the sample, and firm fixation of the sample, on a specimen stub, were performed next, before placement of the sample in the specimen slot, was performed. Sputter coating of the sample with platinum was performed before sectioning of the sample ensued. The bioflocculant surface was observed by SEM.
Analysis of the chemical composition of the bioflocculants was performed via an in-built EDX. The supercharged electron beam from the SEM, hit the bioflocculant sample and caused the release of X-rays from the bioflocculant surface atoms [38]. The X-rays from this interaction, were pooled by the X-ray detector in the EDX, interpreted as elemental-type, and elemental-concentration results, and displayed as an EDX graph [38].
High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS)
The carbohydrate sugars present in the bioflocculant were analyzed using an Agilent 1100 series HPLC system (Agilent Technologies, California, USA) equipped with a diode array detector (DAD) and a reversed-phase C-18 Zorbax Eclipse extradense bundle (XDB-C18) column (250 × 4.6 mm, 5 µm particle size, 300 Å pore size). An Agilent 1100 Series LC/MSD benchtop mass spectrometer connected to the HPLC device was used to determine the massâ??charge ratio of the carbohydrate sugars.
A set of HPLC procedures outlined by, Anumula, Guzzetta, and Sharma et al. were used [39-41]. The Agilent 1100 Series High Value System User’s Guide from Agilent Technologies was also used [42]. The HPLC solvent conduits were degassed by HPLC- grade isopropanol purging prior to the commencement of the analytical technique. Solvent A (HPLC-grade water combined with 0.1% v/v formic acid) and solvent B (HPLC-grade acetonitrile and 0.1% v/v trifluoroacetic acid), composed the HPLC reversed- phase solvent elution setup [40]. Multiple injections of the same samples were enabled through an injector protocol accessed in the HPLC device to separate the monosaccharide moieties [43]. The HPLC procedure involved sorting non-volatile compounds from the analyte to forestall matrix interference [44].
The product of the HPLC analysis was channeled into an Agilent 1100 Series LC/MSD (liquid chromatography/mass selective detector) benchtop mass spectrometer. The ionization of the sample kick-started the analysis; a disaggregation of the sample ions ensued in the electromagnetic field created by the spectrometer on the basis of the differences in ionic mass/charge (m/z) ratios [45]. The disaggregated ions were identified and quantified as they made contact with both ultraviolet and mass selective detectors of the device; this information was combined into an array of graphical results [45]. Consequently, both qualitative and quantitative characterization of the bioflocculant sample, could be performed via mass spectrometry [44]. A total ion chromatogram was the final graphical display of the results of both HPLC and MS.
The Phenol-Sulfuric Acid Method
The phenol-sulfuric acid method was used to measure the concentration of the total sugar content of the bioflocculant (expressed in milligrams/liter); this method employed glucose as the standard [46]. The amount of carbohydrate was determined as described. A quantity of the bioflocculant, 0.1 mL, was mixed with 2 mL of distilled water. To this mixture, 0.1 mL of 6% phenol and 5 mL of 95% sulfuric acid (v/v) were quickly added. This composite mixture was shaken and left to stand for 10 minutes. The optical density was then recorded at 490 nm, as an average of three separate readings. Two milliliters of distilled water mixed with 0.1 mL of 6% phenol and 5 mL of 95% sulfuric acid (v/v) was used as the blank control, and the absorbance was measured. The amount of bioflocculant sugars was determined using a standard glucose curve that had to be prepared. This process was conducted according to the method outlined by DuBois et al. with some modifications [47]. Glucose (0.1 g) was dissolved in 100 mL of distilled water to make a stock solution. From this stock solution, 10 mL was removed and added to 90 mL of distilled water to make 100 mL of diluted stock solution.
The following volumes of the diluted stock solution were poured into five different sterile bottles: 0.1 mL, 0.2 mL, 0.4 mL, 0.8 mL and 1.0 mL. Next, a series of different dilutions of the glucose solution was achieved by making up each volume of stock solution to 3 mL, through the addition of the needed volume of distilled water. For each of these concentrations, 0.1 mL of 6% phenol and 5 mL of 95% (v/v) sulfuric acid were quickly mixed, and the composite mixtures were allowed to stand for 10 minutes. The optical density (OD) at 490 nm was subsequently measured. A standard glucose curve was then plotted with the optical densities of the different glucose dilutions at 490 nm on the y-axis and the corresponding glucose concentration in mg/L on the x-axis. A linear graph of the optical densities of the bioflocculants was also constructed from the best-fit line. From the standard glucose curve, the following mathematical equation was obtained:
y = mx + c
where y represented the optical density (absorbance) at a wavelength of 490 nm; m represented the mass; x represented the concentration of the bioflocculant sugars; and c represented the mathematical (velocity) constant [48].
Substituting values, the exact concentration of bioflocculant sugars was calculated from the following formula:
OD bioflocculant = 0.3492x + 1.2234
where OD bioflocculant represented the average optical density of the bioflocculant sugars
Results
The bioflocculant produced from Pseudomonas aeruginosa strain F29 was characterized using Fourier transform infrared (FTIR) spectroscopy, scanning electron microscopy coupled to energy dispersive X-ray analysis (EDX), high-performance liquid chromatography (HPLC) coupled to mass spectrometry, and the phenolâ??sulfuric acid method. These investigations revealed the typical elemental compositions, the functional groups and the physical characteristics of the bioflocculant.
Fourier Transform Infrared (FTIR) Spectroscopy
The FTIR result of the bioflocculant from Pseudomonas aeruginosa strain F29, at 4000-400 cm-1 frequency range, is shown on Figure 1. The functional groups of the bioflocculant from Pseudomonas aeruginosa strain F29, were determined by interpreting the peaks in both the functional group and the fingerprint regions of the FTIR spectrum. The FTIR showed a strong absorption peak at 3241.10 cm-1 (broad), that correlated with O-H stretching of the hydroxyl group of a carboxylic acid/carboxylate (COOH) or a concentrated aromatic alcohol (-OH). This broad, strong, FTIR absorption peak at 3241.10 cm-1 also fell within the 3300-3200 cm-1 range that correlated with N-H moieties that participated in hydrogen bonding. The absorption at 1635.05 cm-1 was medium and was possibly a result of vibrations from C = C alkene bond stretching in disubstituted (cis) compounds. This absorption at 1635.05 cm-1 also indicated the presence of carbonyl (C = O) groups in sugars or/ and amide/peptide bonds. Furthermore, the absorption at 1635.05 cm-1 fell within the 1660-1500 cm-1 range that is typical for vibrations from saturated nitro compounds. The strong absorption peak at 599.55 cm-1 was attributed to C-I, C-Br or C-Cl stretching (typically 700-600 cm-1) in a halo compound and was also due of the bending of carbonyl (C = O) groups in an amide/peptide bond. Furthermore, this strong FTIR absorption peak at 599.55 cm-1, fell within the 610-390 cm-1 range for P-Cl bonds; 670-550 cm-1 range typical for stretching vibrations from magnesium oxide bonds; and the 600-590 cm-1 range for sulfones. The absorption peak at 474.39 cm-1 was from the Sâ??S stretch correlated with polysulfides, while the FTIR of 418.53 cm-1 was from the vibrations of a metal-chloride (M-Cl) bond.
Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray Analysis/Spectroscopy (EDS/EDX)
Plate 1 shows an SEM image of the bioflocculant from Pseudomonas aeruginosa strain F29, while Figure 2 shows the EDS/EDX result. The SEM image showed a high-resolution three- dimensional image of the scanned surface of a narrow stretch of the surface of the bioflocculant. This surface looked clumped and flaky in topography. The X-ray energy emitted by the SEM electron beam was captured and read by an in-built analyzer (EDX.) The elemental composition of the bioflocculant was recorded as a percentage of its total weight, and the following results were obtained: chlorine (56.00%), carbon (20.50%), sodium (12.50%), oxygen (4.00%), phosphorus (3.00%), magnesium sulfur (2.43%), magnesium (1.06%), potassium (0.32%) and nitrogen (0.30%). The amount of chlorine was slightly greater than half the total weight of the bioflocculant. The presence of carbon and oxygen suggested the presence of carbonyl and carboxylic bonds, as found in sugars; and carbonyl bonds present in proteins and peptides.
High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS)
Figure 3 shows the total ion chromatograph of the HPLC and MS performed on the bioflocculant. The monosaccharide composition of the bioflocculant was revealed by HPLC, and the mass was determined by measuring the ionized atoms of the bioflocculant. The different masses that corresponded to different retention times were compared to a standard for identification of the monosaccharide. There were many peaks for a single simple sugar compound because of the different injection rates used in the HPLC and the existence of stereoisomers. Additionally, the total number of peaks for each type of simple sugar moiety were numbered. The total ion chromatogram showed that the bioflocculant from Pseudomonas aeruginosa strain F29 consisted of monosaccharides; namely, glucose, galactose, inositol and mannose, D-ribose, arabinose, rhamnose and xylose. There was a total of sixteen recorded peaks, with glucose possessing four peaks (1, 2, 9 and 11); galactose with three peaks (5, 14 and 15); inositol with two peaks (3 and 16); rhamnose with two peaks (4 and 12); and mannose with two peaks (6 and 10). Arabinose, xylose and ribose had one peak each (7, 8 and 13). Glucose and galactose are stereoisomers.
The Phenol-Sulfuric Acid Method
This analytic tool was used to determine the concentration of carbohydrate sugars in the bioflocculant from a standard glucose curve. The concentration of sugars in the bioflocculant was 0.0059 g/L.
Plate 1: Scanning Electron Microscopy (Sem) Micrograph of the Bioflocculant Produced from Pseudomonas aeruginosa Strain F29
Key: F29 Bioflocculant from Pseudomonas aeruginosa Strain F29, WD: Working Distance, Mag: Magnification, Hv: High Voltage, HFW: Horizontal Field Switch, MM: Millimeters KV: Kilovolts, µm: Micrometers, PA: Pascals
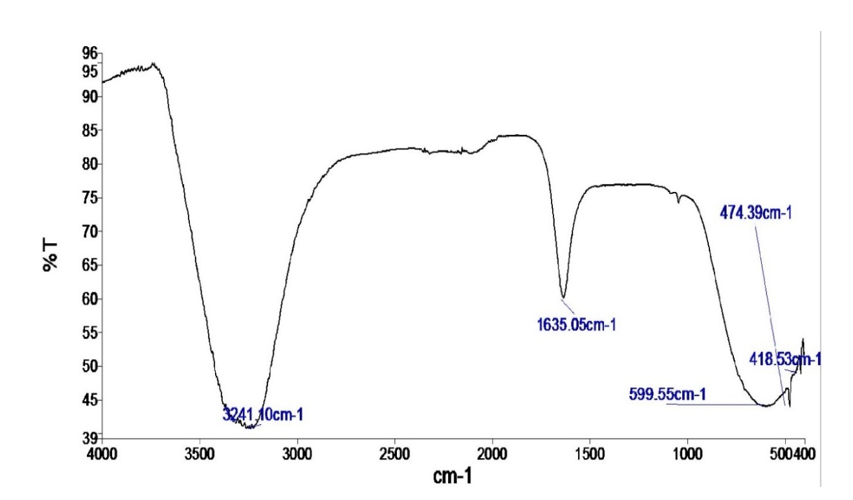
Figure 1: FTIR Spectrum of Bioflocculant from Pseudomonas aeruginosa Strain F29
Key: y-axis- Absorbance (2-log%T), x-axis-Wavelength Measured in cm-1
Figure 2: Energy Dispersive X-Ray Analysis (Edx) Spectrum of the Bioflocculant from Pseudomonas aeruginosa Strain F29
Key- C: Carbon, O: Oxygen, P: Phosphorus, Cl: Chlorine, S: Sulfur, Na: Sodium, Mg: Magnesium, K: Potassium, N: Nitrogen, KeV (x-axis): Energy of the X-rays, y-axis: Peak Intensity (counts), Wt (%): Percentage Weight, F29: Pseudomonas aeruginosa Strain F29
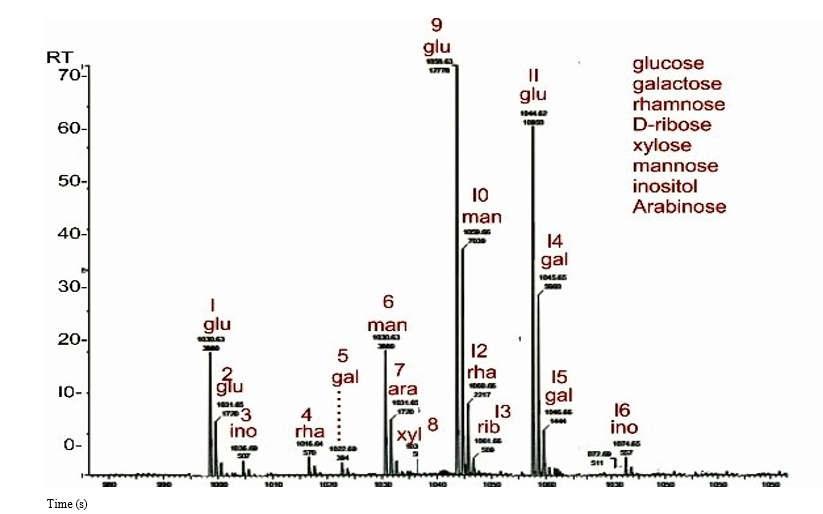
Figure 3: Total Ion Chromatogram of the Bioflocculant from Pseudomonas aeruginosa F29
Key: RT: Retention Time, s: Seconds, glu: Glucose, xyl: Xylose, man: Mannose, rha: Rhamnose, rib: D-Ribose, gal: Galactose, ara: Arabinose, ino: Inositol
Discussion
The physical characteristics, the composition and the relative distribution of the chemical elements, and the functional groups of the bioflocculant that was produced from Pseudomonas aeruginosa strain F29, were determined, using Fourier transform infrared (FTIR) spectroscopy, scanning electron microscopy (SEM) coupled to energy dispersive X-ray spectroscopy (EDS or EDX), high-performance liquid chromatography (HPLC) coupled to mass spectrometry (MS), and the phenol-sulfuric acid method.
Fourier transform infrared (FTIR) spectroscopy of the bioflocculant from Pseudomonas aeruginosa strain F29, revealed the presence of certain functional groups corresponding to the given absorption peaks. Maliehe et al. reported that the functional moieties of a bioflocculant, determine such properties as its resilience, its plasticity; and its flocculating capacity [23]. Furthermore, Okaiyeto et al. reported that the functional groups in bioflocculants, provide dedicated areas for the chemical interactivity of positively-charged ions and colloids suspended in a liquid medium [49].
The presence of a carboxylic acid/carboxylate compound and/ or a concentrated aromatic alcohol, in the bioflocculant from Pseudomonas aeruginosa strain F29, was alluded to from the strong absorption peak at 3241.10 cm-1; this peak was generated from the stretching of O-H (hydroxyl) functional groups. This finding was in partial agreement with the research by Okaiyeto et al. on a purified bioflocculant, who reported that the peak at 3412 cm-1, was attributed to a hydroxyl group [50]. This strong absorption peak at 3241.10 cm-1 was also attributed to the presence of amino groups, and was within the protein amide A band of 3500–3300cm-1 [35].
The medium absorption peak at 1635.05 cm-1 in the bioflocculant from Pseudomonas aeruginosa strain F29, was attributed to C = C alkene bending in compounds, whose two alkyl groups are oriented on the same side of the alkene (cis isomer). In a study by Hurairah et al. on coagulation-flocculation, the seed peel extract from Archidendra jiringa, that was found to be effective as an aid in the elution of lead from man-made residual water, was noted to possibly contain alkene compounds (or aromatic compounds), as indicated by the weak FTIR bands at 3019 cm-1 and 3001 cm-1 [51].
In the FTIR spectrum of the bioflocculant from Pseudomonas aeruginosa strain F29, the absorption at 1635.05 cm-1, also indicated the presence of carbonyl (C = O) groups, typically within the range of 1900–1600 cm-1 that revealed the presence of carbohydrate sugars in the bioflocculant [33,52]. Together with the second functional group commonly found in carbohydrates, and detected at 3412 cm-1, viz, the hydroxyl group, the presence of carbohydrates in the bioflocculant from Pseudomonas aeruginosa strain F29, was confirmed. In another light, the carbonyl (C=O) groups indicated by the medium absorption at 1635.05 cm-1, also fell within the amide I band, documented as typical for peptides or protein macromolecules, while the strong absorption peak, observed at 599.55 cm-1, fell into another typical range for peptides or proteins, the amide VI band, that comprises “out-of-plane” bending of carbonyl (C = O) groups in amide/peptide bonds [53-55]. Additionally, Li documented that the amide I band is the most readily detected, out of the nine amide bands typically characteristic for a protein macromolecule, and thus can be used to study its composition and surroundings [55]. Yatsyna et al. stated, in their research, that amide IV, V and VI bands, can be accurately used to identify the shape of the amide functional groups, and the framework of bound atoms present in a protein macromolecule [56].
In addition, Yatsyna et al. reported that, amide IV, V and VI bands,also reveal that, amide bonds, as may occur in peptides whose building blocks are arranged in rings, possess amino acid molecules oriented on the same side of the amide/peptide bond (“cis- conformation”) [56]. Furthermore, Ji et al. documented that the amide A, I, II and III bands, were the amide bands used to ascertain the conformation of a protein macromolecule [35]. The presence of a protein (or polypeptide) component in the bioflocculant from Pseudomonas aeruginosa strain F29, was therefore, reasonably established, by the amide I, VI and A bands that were detected in it. Additionally, Wang et al. reported that amidation boosted the flocculation of superfine pyrite [57]. Therefore, the inherent amide bonds present in the bioflocculant from Pseudomonas aeruginosa strain F29, were more than likely, partly responsible for its flocculation bioactivity.
The absorption at 1635.05 cm-1 fell within the 1660–1500 cm-1 range that is typical for vibrations from saturated nitro compounds. From an extensive search of documented studies, it would appear that there is no information on the presence of nitro compounds in either conventional flocculants or bioflocculants. While research such as by Smith may have suggested the inherent potential explosivity of the bioflocculant from Pseudomonas aeruginosa strain F29, the study by Noriega et al. on the vast array of the activities of nitro compounds, suggested that the bioflocculant from Pseudomonas aeruginosa strain F29, may exhibit antimicrobial, antiparasitic and antitumour bioactivities in addition to certain other [58,59]. Nonetheless, Noriega et al. also stated that the sum total of the bioactivities of the nitro moiety, whether advantageous or harmful, is completely contingent on its chemical reduction (by receiving as many as 6 electrons) to the amide compound [59]. As stated previously, the amide condensation reaction has been observed to enhance flocculation [57]. This amide bond, characterizes the amide/peptide moiety.
Furthermore, Kurniawan et al. reviewed research that called attention to certain mechanisms by which moieties such as the amide moiety, subserve the coagulation-flocculation brought about by the Moringa plant [4]. Therefore, it is inferable that the nitro moiety may display flocculation via the amide compounds obtained from the nitro-reduction pathway it undergoes, and that the overall flocculation exhibited by a nitro compound-containing polymeric flocculant, such as the bioflocculant from Pseudomonas aeruginosa strain F29, would partly be due to this nitro moiety. Yet again, research such as by Kovacic and Somanathan, and Gupta and Ronen, has documented some nitro compounds as harmful contaminants in the environment and in wastewaters, in need of remediation [60,61].
Ma et al. observed that environmental contaminants such as chromium and nitrobenzene, were flocculated out of the experimental aqueous mixtures of these contaminants that they had created, via activated biomass [62]. Consequently, it would be worthwhile to observe if the flocculation triggered by a nitro compound-containing flocculant such as the bioflocculant from Pseudomonas aeruginosa strain F29, is effective in remediating toxic nitro compounds, from wastewater. If effective, it may be quite instructive to determine how this flocculation compares with effective, non-nitro compound-containing flocculants in common use.
The strong FTIR absorption peak at 599.55 cm-1 was also within the 670–550 cm-1 range, typical for stretching vibrations from magnesium oxide bonds [36]. Although studies such as by Liu et al. and by Das et al. reported on the flocculation property of magnesium oxide, the study by Sofi et al. that reported on some other properties of magnesium oxide, may give better insight into its presence in the bioflocculant produced from Pseudomonas aeruginosa strain F29 [63-65]. It may be that, as reported by Sofi et al. for pigments and zero-resistance materials, magnesium oxide enhanced and improved the properties and performance of the bioflocculant produced from Pseudomonas aeruginosa strain F29 [65]. These enhancements and improvements would no doubt, have boosted the overall qualities of the bioflocculant produced from Pseudomonas aeruginosa strain F29, and not only bioflocculation.
The strong FTIR absorption peak at 599.55 cm-1, fell within the 600–590 cm-1 range for sulfones [29]. There seems to be an apparent lack of documentation on the existence of naturally- occurring sulfonated bioflocculants. However, lignosulfonates, that are bye products in the pulp and paper-making industry, have been observed to possess flocculation properties [66,67]. As such, lignosulfonates are strictly-speaking, man-made compounds. Tang et al. functionalized a bioflocculant, chitosan, with a sulfone [68]. This synthetic, chemically modified, sulfone-containing bioflocculant, CS-g-P(AM-AMPS), was observed to effectively remediate heavy metals from wastewater, by the binding of the sulfone moiety to the heavy metal ions, to form a stable ring structure, in addition to heavy metal-binding to other moieties, and simultaneous sedimentation [68]. The presence of sulfones in the bioflocculant produced from Pseudomonas aeruginosa strain F29, was therefore, very likely, contributory to its overall flocculation capacity. The strong absorption peak at 599.55 cm-1 was additionally, attributed to the presence of halogen compounds (carbon-iodide, carbon-bromide or carbon-chloride bonds), in the bioflocculant from Pseudomonas aeruginosa strain F29 [30]. Maliehe et al. also reported the presence of halo compounds with carbon-chloride bonds at the 599.14 cm-1 absorption peak, and carbon-iodide bonds at the 509.5 cm-1 absorption peak in their research on the bioflocculant TMT1 [23]. BodîrlÄ?u et al. reported the stretching from a carbon-chloride (C-Cl) bond at 700–600 cm-1, in their research on amalgams made from two categories of wood, while Tripathy and De described polyamines as polymeric compounds that possess flocculating activity and are formed by a condensation reaction between halogenated compounds and amines [69,70]. Therefore, halo compounds can be said to possess inherent flocculating properties and their presence in the bioflocculant produced from Pseudomonas aeruginosa strain F29, may be partly responsible for its flocculation capacity.610–390 cm-1 range for P-Cl bonds [29]. However, ascertaining the exact type of phosphorus-chlorine compounds in the bioflocculant from Pseudomonas aeruginosa strain F29, fell outside the scope of this study. Although Roshchin and Molodkina documented that phosphorus chlorides particularly the oxychloride, trichloride and pentachloride, are noxious substances that are considered occupational dangers, Kudzin et al. used gaseous forms of phosphorus trichloride, to formulate a novel antimicrobial compound [71,72].
Additionally, both phosphorus trichloride and phosphorus pentachloride, have been reported as chlorinating substances [73,74]. Zhang et al. reported that crystalline ferric chloride is a moderately strong electron pair-acceptor that is used as both catalytic and chlorinating substances, in the manufacture of carbon- based compounds [75]. Nonetheless, research such as by Ettaloui et al. has reported on the flocculating property of ferric chloride [76]. Therefore, it may be that the phosphorus chlorides present in the bioflocculant from Pseudomonas aeruginosa strain F29, also possess flocculating properties, that synergistically contribute to its total flocculating capacity. Although a phosphorus-chlorine (P- Cl) bond on FTIR, can also emanate from a phosphoryl chloride, the absence of the phosphoryl moiety in the bioflocculant from Pseudomonas aeruginosa strain F29 bond, negated the possibility of the presence of a phosphoryl chloride.
The FTIR of 418.53 cm-1 was from the vibrations of a metal- chloride (M-Cl) bond, in the bioflocculant from Pseudomonas aeruginosa strain F29 [29]. Three metals are naturally present in the bioflocculant, viz sodium, potassium, and magnesium, and any, or all of them, could have formed the corresponding chloride salt. The study by Zhao et al. reported on the use of the chlorides of sodium, potassium and magnesium as aids to maximize the medium for the production of a bioflocculant from Bacillus subtilis [77]. Furthermore, the use of sodium chloride, potassium chloride and magnesium chloride either as flocculants or flocculant aids have been well documented [78-80]. Therefore, any of these chlorides, if present in the bioflocculant from Pseudomonas aeruginosa strain F29, would likely have contributed to its flocculation property.
The bioflocculant from Pseudomonas aeruginosa strain F29 also contained polysulfides. This was alluded to, by the peak at 474.39 cm-1, that was attributed to Sâ??S disulfide bond stretching [29]. Asmare et al. used the coagulation-flocculation induced by a mixture of calcium polysulfide and ferrous sulphate, for remediating waste water from a leather-making industry [81]. They discovered that although the use of a high concentration of calcium polysulfide (4% v/v), mixed with ferrous sulphate, did not eliminate the chemical oxygen demand entirely, it still resulted in a relatively high reduction (86.13%) in waste, and achieved all other indices of proper wastewater treatment [81]. This finding highlights the relative effectiveness of polysulfide compounds in the coagulation-flocculation process.
High-performance liquid chromatography (HPLC) and mass spectrometry (MS) analyses, revealed different monosaccharides,indicating that the bioflocculant produced from Pseudomonas aeruginosa strain F29 possessed a heteropolysaccharide component. Polysaccharides have been observed to possess bioflocculating properties [11]. In a study conducted by Ma et al. the heteropolysaccharide bioflocculant BP50-2, produced from discarded banana skin, demonstrated effective agglomerating activity [82]. Therefore, the polysaccharide component of the bioflocculant produced from Pseudomonas aeruginosa strain F29 would more likely have contributed to the overall coagulation- flocculation capacity of the bioflocculant.
Scanning electron microscopy of the bioflocculant produced from Pseudomonas aeruginosa strain F29, revealed clumped and flaky surfaces. Xiong et al. reported that the nature of the surface of a bioflocculant holds immense significance with regard to its flocculating capacity [83]. The topographic layout of a bioflocculant, thus determines its potency or lack thereof [9]. EDX of the bioflocculant produced from Pseudomonas aeruginosa strain F29, revealed a relative abundance of chlorine (56.00%) and carbon (20.50%). Sodium (12.50%) and oxygen (4.00%) were next in abundance. Phosphorus (3.00%) and sulfur (2.43%) were also present in the bioflocculant produced from Pseudomonas aeruginosa strain F29, with the least abundant elements being magnesium (1.06%), potassium (0.32%) and nitrogen (0.30%). The carbon and oxygen compositions hinted at the presence of polysaccharides and proteins in the bioflocculant produced from Pseudomonas aeruginosa strain F29 [84,85]. The bioflocculant in the research conducted by Tsilo et al., had a much higher oxygen content (43.76%) and phosphorus content (14.44%), and a much lower chlorine content (0.31%), when compared with the same elements in the bioflocculant produced from Pseudomonas aeruginosa strain F29 [9]. The potassium content (0.34%) of the bioflocculant in the research conducted by Tsilo et al., was almost the same as that present in the bioflocculant produced from Pseudomonas aeruginosa strain F29 (0.32%) [9]. Calcium was absent, in the bioflocculant produced from Pseudomonas aeruginosa strain F29, but was relatively abundant (20.35%) in the bioflocculant studied by Tsilo et al. [9]. These differences in bioflocculant composition were probably due to species differences and the type and ratio of the constituents of the bacterial medium that was used in bioflocculant production.
Some metal cations have been observed to aid in floc-formation [86,87]. Sodium alginate, a compound found in nature, was observed to possess good flocculating activity during sewage treatment, in research conducted by Tian et al., while sodium polyacrylate was used to aggregate tiny hematite particles and thereby cause them to drift away from the intermixed quartz particles, in a study by Cheng et al [88,89]. Ozkan and Yekeler, in their research to determine the floc-forming properties of strontium sulphate (“celestite”), by utilizing two classes of flocculants, discovered that magnesium 2+ ions from magnesium chloride, worked best on the strontium sulphate used, than did calcium 2+ (from calcium chloride) and aluminium 3+ (from aluminium chloride) [80]. Also, in research conducted by Hejazi, potassium compounds were combined with sodium dodecyl sulphate, to trigger floc formation in a mixture that contained megestrol acetate particles [90].
The non-metal, chlorine, was utilized as an aid in flocculation, in addition to its use as a deodorizer and a disinfectant, in certain water purification experiments [91]. Ou et al., in an experiment conducted, observed that the chlorine-containing compound, sodium chloride (common salt) displayed good floc-forming activity against small-grained particles of industrial kaolinite, and that this activity was directly proportional to the quantity per unit volume, of both flocculant and industrial kaolinite [92]. Therefore, it may be deduced that the presence of the metals, sodium, potassium, magnesium, and the non-metal, chlorine, in the bioflocculant produced from Pseudomonas aeruginosa strain F29, would have contributed, to some significant degree, to its bioflocculating activity. The non-metal elements, phosphorus, sulfur and nitrogen have been stated as constituents of certain carbohydrates, and the bioflocculant produced from Pseudomonas aeruginosa strain F29, possessed these elements in varying proportions [93].
Nevertheless, these elements- phosphorus, nitrogen and sulfur - have been described by research such as by Kakade et al., as lesser constituents of carbohydrates, and occur in those carbohydrates that have been modified [94]. On the other hand, phosphorus, sulfur and nitrogen have also been documented as constituents of proteins [85]. Although, nitrogen was described by Biswas as a main constituent of a protein macromolecule, with phosphorus and sulfur present in relatively less proportions, the bioflocculant from Pseudomonas aeruginosa strain F29 possessed relatively more abundance of phosphorus (3.00%) and sulfur (2.43%), than nitrogen (0.30%) [85]. In this manner, the protein component of the bioflocculant from Pseudomonas aeruginosa strain F29 may more likely be described as atypical.
The existence of a protein/proteins were alluded to, from the occurrence of phosphorus, nitrogen, sulfur, and amide/peptide bonds within amide I, VI and A bands, in the bioflocculant from Pseudomonas aeruginosa strain F29 [35,55,56,94]. However, the more specific differentiation between a protein and that of a polypeptide, in the bioflocculant, could not be exactly ascertained. This was because the use of a more diagnostic method, such as x-ray crystallography, that can provide a stereographic view of a protein, fell outside the scope of this study [95]. Additionally, the discovery that more than 75% of the bioflocculant from Pseudomonas aeruginosa strain F29, consisted of carbon and chlorine (with chlorine accounting for more than half of the total amount of elements present), strongly indicated that the bioflocculant, was largely an organochlorine compound (organochloride). The sodium, potassium and magnesium ions, accounted for the total metal composition of the bioflocculant from Pseudomonas aeruginosa strain F29. This metal composition was 13.32%, with sodium ion accounting for 90% of all metals present. Therefore, with the confirmed presence of metals, carbohydrate sugars, proteins and organochlorines, in the bioflocculant from Pseudomonas aeruginosa strain F29, it was deduced that this bioflocculant was a metal-containing polymeric compound composed mainly of carbohydrates, proteins/polypeptides and organochlorines; possibly a “metalloglyco-protein/polypeptide- organochlorine” bioflocculant.
Literature is rife with documentation on polyfunctional compounds. However, it was particularly intriguing and puzzling to discover multiple moieties associated with a variety of well-documented bioactivities; and chemical compounds with diverse applications, packed into one macromolecule namely, the bioflocculant from Pseudomonas aeruginosa strain F29. Marks et al. documented that microbial secondary metabolites can be placed into four major categories based on the following biological functions, namely, fostering protection against competitors and disease- causing microorganisms; enhancing adjustability to unfavorable surroundings; regulating interactivity and coexistence with other life forms; and initiating ecological interaction [96]. Therefore, though the bioflocculant produced by Pseudomonas aeruginosa strain F29, is a single secondary metabolite, the presence of multiple functional groups, that provided dedicated reactive zones, that in turn, determined its chemical reactiveness, characteristics and biological functions; and the incorporation of potentially bioactive chemical compounds; suggested an array of biological functions; that was probably, a response of Pseudomonas aeruginosa strain F29, to a plethora of stimuli it had received in its environment [97]. Further studies on this polyfunctional bioflocculant from Pseudomonas aeruginosa strain F29, could provide valuable insight into its range of chemical properties, bioactivities and industrial applications.
An extensive search of documented research, failed to reveal any report of either metal-containing “glyco-protein/polypeptide or- ganochlorines” (“metalloglyco-protein/polypeptide organochlo- rines) or “glyco-protein/polypeptide-organochlorines”. There seemed to be an absence of documentation on the discovery of these categories of glycoconjugates. There was a report by Fuji- mori et al. on Pseudomonas species producing the vaporescent organochloride compound, chloromethane [98]. However, an ex- tensive search of documented studies did not reveal any report of an organochloride bioflocculant, a “glyco-protein /polypeptide or- ganochlorine” bioflocculant, or a “metalloglyco-protein/polypep- tide organochlorine” bioflocculant from Pseudomonas species or any other microorganism. Additionally, there was also an apparent lack of documentation on the natural occurrence of saturated ni- tro compounds, sulfones, polysulfides, phosphorus chlorine com- pounds, magnesium oxide and metal chlorides, in a bioflocculant. From available documentation, this is the first report of a poly- functional “metalloglyco-protein/polypeptide organochlorine” bioflocculant that naturally contains saturated nitro compounds, sulfones, polysulfides. phosphorus chlorine compounds, magne- sium oxide and metal chlorides; and of a bioflocculant produced from porcine feces in Africa. The strong FTIR absorption peak at 599.55 cm-1 was within the
Conclusion
A bioflocculant produced from Pseudomonas aeruginosa strain F29 (accession number OQ734844) was characterized using investigative procedures. These procedures revealed that the bioflocculant was possibly a novel glycoconjugate, a “metalloglyco- protein/polypeptide organochloride”, that contained metals and naturally-occurring saturated nitro compounds, sulfones, polysulfides, phosphorus chlorine compounds, magnesium oxide and metal chlorides. From available documentation, this is the first report of a polyfunctional “metallo-glycoprotein/polypeptide- organochlorine” bioflocculant that naturally contains saturated nitro compounds, sulfones, polysulfides, phosphorus chlorine compounds, magnesium oxide, and metal chlorides; and of a bioflocculant produced from porcine feces in Africa. Further research on this bioflocculant should prove helpful in establishing the ambit of its properties, activities and applications.
Authors Contribution
I, Ikechukwu Kenneth M. Okorie (corresponding author), carried out all the work in this study as my Masters (MSc.) research project. Professor Adeniyi A. Ogunjobi supervised my Master’s research study.
Acknowledgements
The corresponding author would like to thank Professor Adeniyi A. Ogunjobi for supervising this study. Many thanks also go to Professor Tonye G. Okorie for editing and proofreading the article and to Dr. Augustine Akpoka for his useful suggestions. Mr. Gabriel O. Bamidele helped greatly with the laboratory work.
Source of Funding
This study was self-funded by me, Ikechukwu. Kenneth M. Okorie.
No external funding was received.
Competiting Interest
Both authors declare no financial or nonfinancial competing interest.
Conflicting Interest
Both authors declare no conflicting interest.
References
- World Health Organisation (WHO). (2021). Antimicrobial Resistance. Newsroom: Fact sheets. Retrieved Nov. 7, 2021.
- Abu Tawila, Z. M., Ismail, S., Dadrasnia, A., Usman, M. M. (2018). Production and characterization of a bioflocculant produced by Bacillus salmalaya 139SI-7 and its applications in wastewater treatment. Molecules, 23(10), 2689.
- Yang, Y., Guo, W., Ngo, H. H., Zhang, X., Liang, S., et al. (2024). Bioflocculants in anaerobic membrane bioreactors: A review on membrane fouling mitigation strategies. Chemical Engineering Journal, 486, 150260.
- Kurniawan, S. B., Imron, M. F., Chik, C. E. N. C. E., Owodunni, A. A., Ahmad, A., et al. (2022). What compound inside biocoagulants/bioflocculants is contributing the most to the coagulation and flocculation processes?. Science of the Total Environment, 806, 150902.
- Dih, C. C., Jamaluddin, N. A., Zulkeflee, Z. (2019). Removal of Heavy Metals in Lake Water Using Bioflocculant Produced by Bacillus subtilis. Pertanika Journal of Tropical AgriculturalScience 42.1.
- Alias, J., Hasan, H. A., Abdullah, S. R. S., Othman, A. R. (2022). Properties of bioflocculant-producing bacteria for high flocculating activity efficiency. Environmental Technology & Innovation, 27, 102529.
- Wang, Y., Pushiri, H., Looi, L. J., Zulkeflee, Z. (2022). Applications of bioflocculants for heavy metals removal: a systematic review. International Journal of Environmental Research, 16(5), 73.
- Lai, H., Fang, H., Huang, L., He, G., Reible, D. (2018). A review on sediment bioflocculation: Dynamics, influencing factors and modeling. Science of the total environment, 642, 1184-1200.
- Tsilo, P. H., Basson, A. K., Ntombela, Z. G., Maliehe, T. S., Pullabhotla, V. R. (2022). Production and characterization of a bioflocculant from Pichia kudriavzevii MH545928. 1 and Its Application in Wastewater Treatment. International journal of environmental research and public health, 19(6), 3148.
- Das, N., Ojha, N., Mandal, S. K. (2021). Wastewater treatment using plant-derived bioflocculants: green chemistry approach for safe environment. Water Science and Technology, 83(8), 1797-1812.
- Salehizadeh, H., Yan, N., Farnood, R. (2018). Recent advances in polysaccharide bio-based flocculants. Biotechnology advances, 36(1), 92-119.
- Fauzani, D., Notodarmojo, S., Handajani, M., Helmy, Q., Kardiansyah, T. (2021, November). The cellulose of Boehmeria nivea as natural flocculants: synthesis, modification, and flocculation analysis. In IOP Conference Series: Earth and Environmental Science (Vol. 896, No. 1, p. 012056). IOP Publishing.
- Choi, S. M., Rao, K. M., Zo, S. M., Shin, E. J., Han, S. S. (2022).Bacterial cellulose and its applications. Polymers, 14(6), 1080.
- Badawi, A. K., Salama, R. S., Mostafa, M. M. M. (2023). Natural-based coagulants/flocculants as sustainable market- valued products for industrial wastewater treatment: a review of recent developments. RSC advances, 13(28), 19335-19355.
- Bukhari, N. A., Loh, S. K., Nasrin, A. B., Jahim, J. M. (2020). Enzymatic hydrolysate of palm oil mill effluent as potential substrate for bioflocculant BM-8 production. Waste and Biomass Valorization, 11(1), 17-29.
- Littlefair, J. E., Rennie, M. D., & Cristescu, M. E. (2022). Environmental nucleic acids: A field-based comparison for monitoring freshwater habitats using eDNA and eRNA. Molecular Ecology Resources, 22(8), 2928-2940.
- Tsilo, P. H., Basson, A. K., Ntombela, Z. G., Maliehe, T. S., Pullabhotla, R. V. (2021). Isolation and optimization of culture conditions of a bioflocculant-producing fungi from Kombucha tea SCOBY. Microbiology Research, 12(4), 950-966.
- Michaels, A. S. (1954). Aggregation of suspensions by polyelectrolytes. Industrial & Engineering Chemistry, 46(7), 1485-1490.
- Gao, J., Bao, H. Y., Xin, M. X., Liu, Y. X., Li, Q., et al. (2006).Characterization of a bioflocculant from a newly isolated Vagococcus sp. W31. Journal of Zhejiang University Science B, 7(3), 186-192.
- Giri, S. S., Ryu, E., Park, S. C. (2019). Characterizationof the antioxidant and anti-inflammatory properties of a polysaccharide-based bioflocculant from Bacillus subtilis F9. Microbial pathogenesis, 136, 103642.
- Zhong, C., Cao, G., Rong, K., Xia, Z., Peng, T., et al. (2018). Characterization of a microbial polysaccharide-based bioflocculant and its anti-inflammatory and pro-coagulant activity. Colloids and Surfaces B: Biointerfaces, 161, 636-644.
- Li, W. W., Zhou, W. Z., Zhang, Y. Z., Wang, J., Zhu, X. B. (2008).Flocculation behavior and mechanism of an exopolysaccharide from the deep-sea psychrophilic bacterium Pseudoalteromonas sp. SM9913. Bioresource technology, 99(15), 6893-6899.
- Maliehe, T. S., Simonis, J., Basson, A. K., Reve, M., Ngema, S., et al. (2016). Production, characterisation and flocculation mechanism of bioflocculant TMT-1 from marine Bacillus pumilus JX860616. African Journal of Biotechnology, 15(41), 2352-2367.
- Tang, W., Song, L., Li, D., Qiao, J., Zhao, T., et al. (2014). Production, characterization, and flocculation mechanism of cation independent, pH tolerant, and thermally stable bioflocculant from Enterobacter sp. ETH-2. PloS one, 9(12), e114591.
- Vimala, R. T. V. (2019). Role of bacterial bioflocculant on antibiofilm activity and metal removal efficiency. J. Pure Appl. Microbiol, 13(3), 1823-1830.
- Zoaka, M. H., Faruk, A. U., Ibn Abbas, M., Buba, F. M., Adamu, A., et al. (2025). Bioflocculants as sustainable alternatives in water treatment: mechanisms, applications, and future prospects. Arid-zone Journal of Basic & Applied Research. 4. 3: 43-60.
- Okorie, I. K. M. (2023). Biocontrol of selected bacteria using biosurfactant, bioflocculant and exopolysaccharide, produced from Pseudomonas species. MSc. Project. Dept of Microbiology. University of Ibadan. xvii+154pp.
- NanoScience Technology Centre (NTSC). (2008). FTIR Procedure and instructions: uploads: sites. University of Central Florida. Retrieved 31st May, 2023.
- Socrates, G. (2004). Infrared and Raman characteristic group frequencies: tables and charts. John Wiley & Sons.
- Aldrich, S. (2019). IR spectrum table & chart. Sigma Aldrick, 1.
- Aksnes, D. A. G. F. I. N. N., Aksnes, G. U. N. N. A. R. (1963).Intensity Of Infrared Absorption Bands of Phosphoryl Groups in Organic Phosphorus Compounds. Acta Chemica Scandinavica, 17(5), 1262.
- Aboul-ene-n, Y., Bunac-u, A., Flesch-n, S. (2014). Evaluation of the protein secondary structures using Fourier transform infrared spectroscopy. Gazi University Journal of Science, 27(1), 637-644.
- Smith, B. C. (2017). The carbonyl group, part I:introduction. Spectroscopy 32. 9: 31-36.
- Nandiyanto, A. B. D., Oktiani, R., Ragadhita, R. (2019). How to read and interpret FTIR spectroscope of organic material. Indonesian Journal of Science and Technology, 4(1), 97-118.
- Ji, Y., Yang, X., Ji, Z., Zhu, L., Ma, N., et al. (2020). DFT-calculated IR spectrum amide I, II, and III band contributions of N-methylacetamide fine components. ACS omega, 5(15), 8572-8578.
- Agyapong, P. O., Gikunoo, E., Arthur, E. K., Anang, D. A., Agyemang, F. O., et al. (2023). Synthesis of magnesium oxide from waste magnesium-rich Cucurbita pepo (pumpkin) seeds. Sustainable Environment, 9(1), 2258473.
- Facilities, C. (2021). SEM sample preparation techniques.University of Gothenburg. Retrieved, 28, 357-372.
- Thambiratnam, K., Reduan, S. A., Tiu, Z. C., Ahmad, H. (2020). Application of two-dimensional materials in fiber laser systems. In Nano-Optics (pp. 227-264). Elsevier.
- Anumula, K. R. (1994). Quantitative determination of monosaccharides in glycoproteins by high-performance liquid chromatography with highly sensitive fluorescence detection. Analytical biochemistry, 220(2), 275-283.
- Guzzetta, A. (2001). Reverse Phase HPLC Basics for LC/ MS: An IonSource Tutorial. IonSource: Mass Spectrometry Educational Resource. Retrieved 22nd July, 2001.
- Sharma, K., Sharma, N., Handa, S., Pathania, S. (2020). Purification and characterization of novel exopolysaccharides produced from Lactobacillus paraplantarum KM1 isolated from human milk and its cytotoxicity. Journal of Genetic Engineering and Biotechnology, 18(1), 56.
- Agilent Technologies. (1999). Agilent 1100 Series High Value System:User’s Guide. 11/99 ed. Waldbronn: Agilent Technologies.
- Huber, U. (2010). Performing multiple injections in an isocratic purification experiment using the Agilent 1100 Series purification system: Application Note. GiMiTEC. com/file. https://gimitec.com/file/5989-1651EN.pdf
- Ho, C. S., Lam, C. W. K., Chan, M. H., Cheung, R. C. K., Law,L. K., et al. (2003). Electrospray ionisation mass spectrometry: principles and clinical applications. The Clinical Biochemist Reviews, 24(1), 3.
- Reusch, W. (2013). Virtual Text of Organic Chemistry: Lipids.
- Chaplin, M. and Kennedy, J. J. M. S. (1986).Monosaccharides. Mass Spectrom 1.7.
- DuBois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., Smith,F. (1956). Colorimetric method for determination of sugars andrelated substances. Analytical chemistry, 28(3), 350-356.
- Jain, V. M., Karibasappa, G. N., Dodamani, A. S., Mali, G. V. (2017). Estimating the carbohydrate content of various forms of tobacco by phenol-sulfuric acid method. Journal of education and health promotion, 6(1), 90.
- Okaiyeto, K., Nwodo, U. U., Mabinya, L. V., Okoli, A. S., & Okoh, A. I. (2015). Characterization of a bioflocculant (MBF- UFH) produced by Bacillus sp. AEMREG7. International Journal of Molecular Sciences, 16(6), 12986-13003.
- Okaiyeto, K., Nwodo, U. U., Mabinya, L. V., Okoh, A. I. (2013). Characterization of a bioflocculant produced by a consortium of Halomonas sp. Okoh and Micrococcus sp. Leo. International journal of environmental research and public health, 10(10), 5097-5110.
- Hurairah, S. N., Fahimi, N. S. M., Halim, A. A., Hanafiah, M. M., Nordin, N., et al. (2023). Archidendron jiringa seed peel extract in the removal of lead from synthetic residual water using coagulation-flocculation process. ScienceAsia, 49(1).
- Roberts, J. D. and Caserio, M. C. 1977. Basic Principlesof Organic Chemistry. 2nd ed. California: WA Benjamin,Incorporated.
- Krimm, S., Bandekar, J. (1986). Vibrational spectroscopy and conformation of peptides, polypeptides, and proteins. Advances in protein chemistry, 38, 181-364.
- Bandekar, J. (1992). Amide modes and protein conformation. Biochimica et Biophysica Acta (BBA)-Protein Structure and Molecular Enzymology, 1120(2), 123-143.
- Li, C. (2016). Infrared spectroscopy: a tool for protein characterization (Doctoral dissertation, Department of Biochemistry and Biophysics, Stockholm University).
- Yatsyna, V., Bakker, D. J., Feifel, R., Rijs, A. M., Zhaunerchyk,V. (2016). Far-infrared amide IV-VI spectroscopy of isolated 2-and 4-Methylacetanilide. The Journal of chemical physics, 145(10).
- Wang, Z., Liu, N., Zou, D. (2021). Interface adsorption mechanism of the improved flotation of fine pyrite by hydrophobic flocculation. Separation and Purification Technology, 275, 119245.
- Smith, B. C. (2020). Organic nitrogen compounds x: nitrogroups, an explosive proposition. Spectroscopy, 35(9), 27-31.
- Noriega, S., Cardoso-Ortiz, J., López-Luna, A., Cuevas- Flores, M. D. R., Flores De La Torre, J. A. (2022). The diverse biological activity of recently synthesized nitro compounds. Pharmaceuticals, 15(6), 717.
- Kovacic, P., Somanathan, R. (2014). Nitroaromatic compounds: Environmental toxicity, carcinogenicity, mutagenicity, therapy and mechanism. Journal of Applied Toxicology, 34(8), 810-824.
- Gupta, S., Ronen, Z. (2024). Biological treatment of nitroaromatics in wastewater. Water, 16(6), 901.
- Ma, R., Chen, G., Liu, K., Cheng, S. (2020). Colloidal Influence Factor of Bioflocculant in Coagulation of Chromium and Nitrobenzene. Journal of Sensors, 2020(1), 9702147.
- Liu, X., Zhao, J., Liu, L. (2023). Advancements in the evolution of engineering characteristics and reinforcement technologies for subgrade silt. Materials, 16(21), 6965.
- Das, N., Rajput, H., Aly Hassan, A., Kumar, S. (2023). Application of different coagulants and cost evaluation for the treatment of oil and gas produced water. Water, 15(3), 464.
- Sofi, A. H., Akhoon, S. A., Mir, J. F., Rather, M. U. D. (2021).Magnesium oxide (MgO): a viable agent for antimicrobial activity. In Applications of Nanomaterials in Agriculture, Food Science, and Medicine (pp. 98-105). IGI Global Scientific Publishing.
- Anukam, A., Berghel, J., Henrikson, G., Frodeson, S., Ståhl,M. (2021). A review of the mechanism of bonding in densified biomass pellets. Renewable and Sustainable Energy Reviews, 148, 111249.
- Chan, S. L. H., Baker, C. G. J., Beeckmans, J. M. (1976). Flocculating properties of high molecular weight lignosulphonates. Powder Technology, 13(2), 223-230.
- Tang, X., Huang, T., Zhang, S., Wang, W., Zheng, H. (2020). The role of sulfonated chitosan-based flocculant in the treatment of hematite wastewater containing heavy metals. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 585, 124070.
- Bodîrl-u, R., Teac, C. A., Spiridon, I. (2009). Preparation and characterization of composites comprising modified hardwood and wood polymers/poly (vinyl chloride). BioResources, 4(4).
- Tripathy, T., De, B. R. (2006). Flocculation: a new way to treat the waste water, Journal of Physical Sciences.10. 2006:93–127. Retrieved 30th Dec, 2015.
- Roshchin, A. V., Molodkina, N. N. (1977). Chloro compounds of phosphorus as industrial hazards. Journal of Hygiene, Epidemiology, Microbiology, and Immunology, 21(4), 387-394.
- Kudzin, M. H., Mrozinska, Z., Urbaniak, P. (2021). Vapor Phosphorylation of Cellulose by Phosphorus Trichlo-Ride: Selective Phosphorylation of 6-Hydroxyl Function—The Synthesis of New Antimicrobial Cellulose 6-Phosphate (III)- Copper Complexes. Antibiotics, 10(2), 203.
- Dillon, K. B., Reeve, R. N., Waddington, T. C. (1976). Phosphorus pentachloride as a chlorinating agent. Journal of Inorganic and Nuclear Chemistry, 38(8), 1439-1442.
- Xiao, J., Han, L. B. (2019). Atom-efficient chlorination of benzoic acids with PCl3 generating acyl chlorides. Journal of Chemical Research, 43(5-6), 205-210.
- Zhang, M., Buekens, A., Ma, S., Li, X. (2018). Iron chloride catalysed PCDD/F-formation: Experiments and PCDD/F- signatures. Chemosphere, 191, 72-80.
- Ettaloui, Z., Salah, S., Aguelmous, A., Jada, A., Taleb, A. (2021). Use of ferric chloride contained in the rejects from the steel industry as a coagulant for the fuel washing wastewater treatment. Desalination and Water Treatment, 234, 68-76.
- Zhao, C., Yang, Q., Zhang, H. (2017). Optimization of microbial flocculant-producing medium for Bacillus subtilis. Indian journal of microbiology, 57(1), 83-91.
- Pérez, K., Moraga, C., Herrera, N., Salinas-Rodríguez, E., Gálvez, E. (2021). Effect of the addition of flocculants and KCl on sedimentation rate of spodumene tailings. Metals, 11(6), 986.
- Zhou, X., Liu, Z., Wang, W., Miao, Y., Gu, L., et al. (2022). NaCl induces flocculation and lipid oxidation of soybean oil body emulsions recovered by neutral aqueous extraction. Journal of the Science of Food and Agriculture, 102(9), 3752- 3761.
- Ozkan, A., Yekeler, M. E. F. T. U. N. ÃÃÃÂ???°. (2004). Coagulation and flocculation characteristics of celestite with different inorganic salts and polymers. Chemical Engineering and Processing: Process Intensification, 43(7), 873-879.
- Asmare, G., Getachew, B., Kassa, T., Abate, W. (2021). Tannery wastewater treatment by coagulation-flocculation technique using combination of calcium polysulfide and ferrous sulphate. American Journal of Chemical Engineering, 9(4), 79-83.
- Ma, X., Duan, D., Chen, X., Feng, X., Ma, Y. (2022). Apolysaccharide-based bioflocculant BP50-2 from banana peel waste: Purification, structure and flocculation performance. International Journal of Biological Macromolecules, 205, 604- 614.
- Xiong, Y., Wang, Y., Yu, Y., Li, Q., Wang, H., et al. (2010). Production and characterization of a novel bioflocculant from Bacillus licheniformis. Applied and environmental microbiology, 76(9), 2778-2782.
- Awuchi, C. G., Amagwula, I. O. (2021). Biochemistry and nutrition of carbohydrates. Global Journal of Research in Agriculture and Life Sciences, 1(1), 4-12.
- Biswas, S. Ed. 2020. Protein. Basic Food Chemistry. Bharati.K. Ed. Krishna Nagar Delhi: Ocean Publishing House. 25-35
- Kuriyama, H., Umeda, I., Kobayashi, H. (1991). Role of cations in the flocculation of Saccharomyces cerevisiae and discrimination of the corresponding proteins. Canadian journal of microbiology, 37(5), 397-403.
- Tawila, Z. M. A., Ismail, S., Amr, S. S. A., Abou Elkhair, E. K. (2019). A novel efficient bioflocculant QZ-7 for the removal of heavy metals from industrial wastewater. RSC advances, 9(48), 27825-27834.
- Tian, Z., Zhang, L., Sang, X., Shi, G., Ni, C. (2020). Preparation and flocculation performance study of a novel amphoteric alginate flocculant. Journal of Physics and Chemistry of Solids, 141, 109408.
- Cheng, K., Wu, X., Tang, H., Zeng, Y. (2022). The flotation of fine hematite by selective flocculation using sodium polyacrylate. Minerals Engineering, 176, 107273.
- Hejazi, S. M., Erfan, M., Mortazavi, S. A. (2013). Precipitation reaction of SDS and potassium salts in flocculation of a micronized megestrol acetate suspension. Iranian journal of pharmaceutical research: IJPR, 12(3), 239.
- Weston, R. S. (1924). The use of chlorine to assist coagulation.Journal (American Water Works Association), 11(2), 446-450.
- Ou, Y., Li, R., Liang, R. (2016). Experimental study on the impact of NaCl concentration on the flocculating settling of fine sediment in static water. Procedia Engineering, 154, 529-535.
- Gangasani, J. K., Pemmaraju, D. B., Murthy, U. S. N., Rengan,A. K., Naidu, V. G. M. (2022). Chemistry of herbal biomolecules. In Herbal biomolecules in healthcare applications (pp. 63-79). Academic Press.
- Kakade, P., Chatterjee, A., Pandya, A., Disouza, J., Patravale,V. (2022). Carbohydrate anchored lipid nanoparticles.International journal of pharmaceutics, 618, 121681.
- Johnson, A., Lewis, J., ALBERTS, B. (2002). Molecular biology of the cell.
- Marks, B. B., Nogueira, M. A., Hungria, M. (2025). Microbial Secondary Metabolites and Their Use in Achieving Sustainable Agriculture: Present Achievements and Future Challenges. Agronomy, 15(6), 1350.
- Ertl, P., Altmann, E., & McKenna, J. M. (2020). The most common functional groups in bioactive molecules and how their popularity has evolved over time. Journal of medicinal chemistry, 63(15), 8408-841.
- Fujimori, T., Yoneyama, Y., Taniai, G., Kurihara, M., Tamegai, H., et al. (2012). Methyl halide production by cultures of marine proteobacteria Erythrobacter and Pseudomonas and isolated bacteria from brackish water. Limnology and oceanography, 57(1), 154-162.




