Case Report - (2024) Volume 3, Issue 3
Case Report: Arteriovenous Malformation Causing Intra-Oral Bleeding of Non-Dental Origin in a 9 Years Old Female
Received Date: Oct 27, 2024 / Accepted Date: Nov 29, 2024 / Published Date: Dec 04, 2024
Copyright: ©Â©2024 Anum Khan, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Khan, A., Al-Jaddir, G., Maynard, J., Dajani, H., Villanueva, K., et al. (2024). Case Report: Arteriovenous Malformation Causing Intra-Oral Bleeding of Non-Dental Origin in A 9 Years Old Female. J Surg Care, 3(3) 1-10.
Abstract
Profuse and life-threatening haemorrhage may arise from vascular lesions such as Haemangiomas or Vascular malformations (VM) and these differ based on development, endothelial characteristics, aetiological factors, and clinical presentation [1]. Haemangiomas are vascular tumours which arise from rapid and uncontrolled proliferation of endothelial cells at an early stage of embryogenesis, presenting in soft tissues of the oral cavity and rarely involving hard bony structures. Vascular malformations, on the other hand, exhibit a normal endothelial cellular turnover rate but are malformations in the structure of vessels appearing in the late stage of embryogenesis arising from persistent vascular anastomoses. As opposed to the dynamic and active turnover in haemangiomas, VM are present at birth and inherently static in nature, and change in size as body grows, or upon encountering endocrinal changes, trauma or infections. Vascular Malformation can be further subdivided into two categories based on blood flow rate into low-flow lesions (including capillary, venous and lymphatic malformations) or high-flow lesions (such as Arteriovenous malformations [AVM] or fistulae). Approximately half of the cases of Vascular malformations occur in the head and neck region with 50% of the cases involving skull and maxillofacial region [2].
Keywords
Intra-Oral Bleeding, Arteriovenous Malformation, Craniofacial EmbolizationIntroduction
Profuse and life-threatening haemorrhage may arise from vascular lesions such as Haemangiomas or Vascular malformations (VM) and these differ based on development, endothelial characteristics, aetiological factors, and clinical presentation [1]. Haemangiomas are vascular tumours which arise from rapid and uncontrolled proliferation of endothelial cells at an early stage of embryogenesis, presenting in soft tissues of the oral cavity and rarely involving hard bony structures. Vascular malformations, on the other hand, exhibit a normal endothelial cellular turnover rate but are malformations in the structure of vessels appearing in the late stage of embryogenesis arising from persistent vascular anastomoses. As opposed to the dynamic and active turnover in haemangiomas, VM are present at birth and inherently static in nature, and change in size as body grows, or upon encountering endocrinal changes, trauma or infections. Vascular Malformation can be further subdivided into two categories based on blood flow rate into low-flow lesions (including capillary, venous and lymphatic malformations) or high-flow lesions (such as Arteriovenous malformations [AVM] or fistulae). Approximately half of the cases of Vascular malformations occur in the head and neck region with 50% of the cases involving skull and maxillofacial region [2].
AVM in the head and neck region can have various dental manifestations, particularly in paediatric patients [3]. These manifestations can arise due to the complex interplay between the vascular system and the surrounding tissues, including the hard and soft tissues of the oral cavity. The veins emerging into the arteriovenous system become dilated which in turn increases the blood flow rate. This increase in blood flow rate subsequently dilates the arteries, forming a characteristic pulsating mass and bruit. This report presents a case of Arteriovenous Malformation (AVM) and literature review of different presentations and its management.
AVM have the propensity to disrupt normal dental development and may lead to delayed eruption of primary or permanent teeth. This may be due to the altered blood flow and pressure affecting the dental follicle [1,3,4]. AVM may manifest as a slow growing innocuous lesion with sudden onset of gingival bleeding [4,5]. AVM primarily presents as acute or chronic intra-oral bleeding, nose bleeding, toothache, unexplained tooth mobility, facial asymmetry, disfigurement, or paraesthesia [2-4]. Increased blood flow and vascularity associated with AVM can lead to epulis formation or gingival hyperplasia, potentially causing discomfort and difficulty in maintaining oral hygiene [3,5]. Additionally, owing to the vascular nature of AVM, patients may experience episodes of oral bleeding, especially during or after dental procedures or traumatic injury [6].
This can complicate dental treatment and necessitate careful management. AVMs can cause facial asymmetry due to abnormal vascular growth, leading to swelling in the affected area due to soft or hard tissue involvement and expansion of facial bones [5-7]. This can impact the appearance and function of the oral and maxillofacial region including disfigurement and psychosocial impact. Prevalence of dental manifestations in paediatric patients with AVM are mostly concentrated in the posterior region (64.93%) affecting roots of first molar teeth. Peak incidence of AVM is said to be 10 - 20 years of age, however AVM has been encountered in patients 3 months and 74 years old, with a 2:1 female to male ratio of occurrence [2,3,8,9].
AVM in paediatric dental patient often requires a multidisciplinary team (MDT) approach involving paediatrician, paediatric dentists, radiologists, Cranio-Maxillo-facial surgeons, oncologists, and vascular specialists [10]. Clinical examination may reveal hard and soft tissue involvement of the head and neck region; however, AVM are considered slow growing expansile lesions and may have an asymptomatic presentation [11]. Investigations include diagnostic imaging (Orthopantomogram [OPG], Computerised Tomography [CT] scan, Magnetic Resonance Imaging [MRI] and Colour Doppler Ultrasonography [US]). OPG is helpful in identifying bony and dental hard tissue involvement, indication further need for investigation such as CT or MRI scans which have a high diagnostic value due to 3-dimensional imaging, however, they come with high radiation exposure risk to patients[4,6,7]. Doppler Ultrasound is less invasive than CT and MRI scans and may help in directing towards the imaging technique more suitable and tailored for individual cases.
MRI scans can help in assessing degree and extent of soft tissue lesions, particularly vascular growths. Doppler US can reveal large draining vessels or feeding arteries, along with fistulous communications. Another advantage of Doppler US is that it can evaluate the velocity of blood flow, strengthening the diagnostic predilection towards a slow or fast flowing vascular lesions [11].Treatment plans and techniques should be tailored and individualized based on the severity of the AVM, the specific dental manifestations, and the overall health of the patient [12]. Surgical management of AVM in paediatric patients is a critical and complex area of care that requires a comprehensive understanding of the anatomy, available surgical techniques, and the specific needs of the patient including medical history and comorbidities. Indications of surgery include acute symptoms such as pain, swelling, facial asymmetry, profuse active bleeding, and functional impairment [12-14].
Surgical treatments are available depending on the extent of growth and clinical presentation, including intravascular embolization, devascularization, sclerosing solutions, surgical resection [15,16]. Resection of AVM is possible in cases of superficial lesions in which complete surgical resection is a feasible option and damage to surrounding structures can be avoided. Due to its highly vascularized and proliferative, infiltrative, and invasive nature, profuse bleeding and haemorrhage risk can complicate the surgery. Hence an embolization procedure can occlude and reduce the blood flow to the AVM making the surgical resection procedure safer by aiding in access and vision. The risk of complications and intra-operative bleeding is high in the paediatric age group, particularly because of the anatomy of the vessels in this population [17,18].
Embolisation is a minimally invasive procedure which involves angiogram-based occlusion of abnormal AVM blood vessels within the nidus. An embolization blocking agent is used to block the abnormal blood vessels. Embolisation is a pre-surgical procedure and not curative in itself. The procedure helps to block the AVM vascular shunts, preventing blood loss during the surgical resection surgery. The procedure involved introduction of a catheter into the connecting blood vessels and blocking the abnormal vessels using embolisation agents such as medical glue, plugs or metal coils. For AVM considered inoperable, unsafe for surgery or embedded into deeper tissues, radiotherapy can be undertaken to induce thrombosis over a period of time, which may help in reducing the size of the AVM [13,15,16].
Management and treatment of AVM requires extensive surgical treatment and close monitoring for complications such as bleeding, infection, or neurological deficits is necessary. Other complications include scar formation at surgical site, dysphagia, paraesthesia, wound dehiscence, or relapse of disease. Rehabilitation may be required depending on the extent of the surgery and the location of the surgical defect. Regular recall and follow-up is important to address and manage complications, and to monitor for recurrence especially in cases where complete resection may not have been possible [10,12,15]. Rehabilitation teams involve school, social workers, occupational therapist, speech and language therapists, dieticians, paediatric psychology support [19]. These are monumental as the sudden transition for the child requires support in managing all aspects of recovery such as emotional trauma, physical pain and functional changes, physiotherapists, and dietary support. Speech and language therapists are instrumental in recovery and rehabilitation and aid in evaluation of swallowing patterns, muscle movements and oral feeding with the aim of recovering as much of function as possible to aid with dietary requirements and speech improvement. A close-knit MDT rehabilitation team and family support with active engagement in the road to recovery has proven to encourage successful and quick rehabilitation for children when family and caregivers [19].
Case Presentation
A 9-year-old female was referred to the Paediatric Dental Unit by the Accident & Emergency (A&E) for unexplained atraumatic profuse intra-oral bleeding. Medical history revealed a single isolated episode of Haematemesis 2 years ago following which patient was seen by the hospital Paediatricians and Gastroenterology team, with no concerns and subsequent discharge. At the time of presentation at our unit, patient was medically fit and healthy. Patient presented to the A&E with mother in the morning (who was working a night shift and was not with the patient at home). As per mother, intra-oral bleeding started at home at night-time, waking the patient up from sleep. There was no history of pain or trauma prior to the onset of bleeding. Father was not present at this time of consultation. Mother showed photographs shared by patient’s father (who took photographs for reference at the onset of bleeding at night) of blood in buckets. As per emergency services advice over the telephone, pressure was applied with gauze on the intra¬oral site of bleeding which controlled the bleeding intermittently at home.
At admission, patient was afebrile, alert, exhibiting no rash or respiratory distress. There was no evidence of bleeding from skin, no blood in stool or urine. However, due to intermittent but profuse bleeding, patient suffered vasovagal syncope secondary to postural hypotension while inpatient. Haemoglobin (Hb) following admission was 87 g/L. Acute bleeding was controlled with tranexamic acid (15 mg/kg) bolus and mouthwash. 10 ml/kg saline bolus, 380 mls of red blood cells and Fresh Frozen Plasma (FFP) transfusion were administered in the A&E twice to combat hypovolemia, along with Ondansetron (IV 2.5 mg), Omeprazole (10mg) due to ‘coffee ground’ vomiting episode.
Patient presented to the Paediatric Dentistry Unit along with parents in a wheelchair due to previous episodes of vasovagal collapse and lethargy. There was no extra-oral facial asymmetry or swelling. Intra-oral examination revealed erythematous buccal gingiva adjacent to LLE and LL6. Lower left first permanent molar (LL6) was lingually tilted exhibiting grade 2 mobility, along with mobile second primary molar (LLE), as shown in Figure 1. Otherwise, dentition was caries free with no other soft tissue abnormality detected. There was no paraesthesia or numbness indicating nerve involvement. Intraorally, palpation revealed expansion of buccal and lingual cortical plates.
Figure 1: Blanching and Erythema of Attached Gingivae Adjacent to LLE and Lingually Tilted LL6
A) Lingually tilted LL6 and B) Blanching of attached gingiva and erythematous marginal gingiva on LLE and LL6
A Orthopantomogram (OPG) radiograph showed 'soap bubble/ honey-comb' radiolucent lesion on the left mandible extending between unerupted LL5 to the angle of mandible, extending upwards towards the ramus, root resorption of LL6 and LLE, displacement of LL7, and thinning of cortex, as shown in Figure 2.
Figure 2: OPG Showed Diffuse Radiolucency Across Angle and Ramus of left Side of the Mandible
A) Diffuse lower left mandible radiolucency with soap bubble appearance, loss of trabeculation of bone. B) Thinning of bony cortex. C) Displacement of LL7 and obliteration of the left side Inferior Dental (ID) canal. D) Loss of lamina dura or teeth LLE and LL6 with root resorption.
Following this, both parents were consulted by the MDT and consented for further investigations including additional imaging and diagnostic Esophagogastroduodenoscopy (OGD) procedure to rule out gastric origin of bleeding. An emergency OGD procedure was carried out under general anaesthesia which revealed no abnormal findings or bleeding source identified within the upper Gastrointestinal (GI) tract. While admitted, intermittent episodes of intraoral bleeding occurred with an average duration of 5-8 minutes of bleeding and up to 200 ml of amount of blood collected into a sick bowl per episode.
Paediatric Dental and Gastroenterology teams approached parents to discuss the findings from the OGD procedure undertaken, at which point father recalled the events of the night before and stated bleeding started as an intra-oral pulsating ‘fountain like’ spurt of blood, appearing to arise from the lower left posterior region (lower left first permanent molar [LL6] site). A pulsating, spurting of blood flow is characteristic of fast flowing VM indicating an AVM and all clinicians agreed on the possible diagnosis of AVM based on this important piece of information given by patient’s father.
The rationale behind performing a CT scan over MRI in the first instance was the radiolucent lesion on the left mandible suggesting a lesion with bone involvement rather than solely a soft tissue lesion. Figure 3 shows a coronal view of the head and neck CT scan which revealed buccolingual expansion of mandible, cortical erosion at the mandibular ramus, lingual tilt of LL6, and displacement of LL7 distally towards the angle of the mandible.
Figure 3: Coronal view of the CT Scan Revealed Asymmetrical Pattern of Bone Expansion on the Left Side of the Mandible (anterior to posterior from A - C)
A) Shortened roots of LL6 with buccal expansion of mandibular bone. B) Lingual bony expansion in contrast to the right side. C) Arrow showing left sided bony expansion with loss of trabeculation.
CT scan showed unremarkable appearance of the remainder of the soft tissues of the neck, with a patent oropharynx and well-aerated paranasal sinuses and mastoid air cells. Figure 4 shows an axial view of CT head and neck which also confirms bony expansion, tooth displacement.
Figure 4: Axial View of CT Scan Head and Neck
A) axial view showed buccolingual expansion of left mandible as compared to right side and displacement of LL7 distally. B) bone expansion and radio-opaque vascular structures buccally (feeding vessels).
Figure 5 shows the vascular view from the CT scan vessel analysis and revealed medullary expansion of the left mandibular body and ramus with intraosseous vessels entering by the mandibular foramen, supplied by arterial collateral feeding vessels from the superficial temporal artery/ terminating vessels of the external carotid artery and venous draining vessel from the left retromandibular vein. Externally, the internal jugular vein (1.2 cm) and external carotid artery (0.6 cm) appeared dilated, suggesting a high flow lesion. This view showed multiple serpiginous and dilated vessels at the left para-pharyngeal space with feeding vessels extending from the left internal jugular vein and internal carotid artery, extending into the left mandible. This view also showed further dilated vessel extending left masticator space and lateral left neck.
Figure 5: CT Scan Vascular Analysis
A) Right side of the mandible (for comparison). B) Dilated arterial collateral feeding vessels from the superficial temporal artery/ terminating vessels of the external carotid artery, feeding vessels extending from the left internal jugular vein and internal carotid artery, extending into the left mandible (as shown by arrows). C) Mandibular bony expansion and feeding vessels in sagittal view. D) Medullary expansion of the left mandibular body with intraosseous vessels entering by the mandibular foramen, venous draining vessel from the left retromandibular vein, multiple serpiginous and dilated vessels at the left para-pharyngeal space with feeding vessels.
CT scan images were deemed sufficient for a formal diagnosis and an urgent referral to Craniofacial unit was made following confirmation of AVM diagnosis. MRI was taken at a later stage as mentioned in the proceeding section, as a diagnostic and therapeutic intervention.
Differential diagnoses included Differential diagnosis of ruptured aneurysmal bone cyst, Langerhans cell histiocytosis, haemangioma, Ewing’s sarcoma, aneurysmal bone cyst, ameloblastoma, osteosarcoma, odontogenic myxoma, ameloblastic fibroma, central giant cell granuloma was considered [3,5]. However, parents’ detailed recall of ‘pulsating, spurting fountain-like blood’ flowing from LL6 site strongly suggested a vascular origin of bleeding [1,2,7,8].
Cerebral Angiography as shown in Figure 6, followed by embolization of left mandibular AVM was undertaken as shown in Figure 7. At the end of the procedure, immediate brisk bleeding ensued requiring major haemorrhage protocol activation with infusion of 270 ml of red blood cells, 400 ml of octaplas (a form of FFP) and 500 ml of Hartmann’s solution, in theatre. Following, approximately 1 litre of blood loss, bleeding was controlled with gauze soaked with adrenaline and tranexamic acid and patient was transferred to Paediatric Intensive Care Unit (PICU) post-procedure.
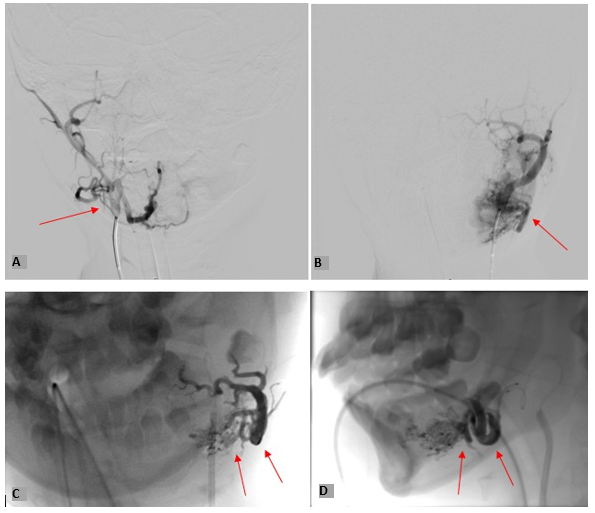
Figure 6: Showing Selective Angiography of
A) Right Internal Carotid Artery, B) Left Internal Carotid Artery. C) Coronal view and, D) Sagittal view of large left mandibular AVM with a massive varix within the bone.
Figure 7: Embolisation of Mandibular AVM (Transarterial Embolization Through Facial, Lingual and Mandibular Branches)
Two days following the embolization procedure, surgical excision of congenital Craniofacial AVM was undertaken under general anaesthesia with successful extubation and no post-surgical complications. Resection of craniofacial AVM left mandible, complete disc articulation resection to Lower right lateral incisor (LR2) was undertaken. Following surgical treatment, patient was admitted in Paediatric Intensive Care Unit (PICU), and Percutaneous Endoscopic Gastrostomy (PEG) fed for 2 weeks to aid with functional adjustment, nutrition and rehabilitation. Patient was managed by the Maxillofacial team as the primary team and other teams such as intensive care, plastic surgery, pain management were involved. Patient transitioned to soft food in 4 weeks’ time, however, struggled with drooling occasionally and experienced frequent panic attacks. Comprehensive paediatric psychologist support was arranged to address the shock and panic associated with the diagnosis and surgical treatment, along with functional changes. One of the complications of resection is scar formation at the surgical site. Dermatex and Kelo-cote was prescribed to help with the appearance of the surgical scar. Furthermore, dieticians were involved, physiotherapy, and speech and language therapy were arranged to help with nutrition and speech following resection of more than half of the mandible.
Follow up with Maxillofacial team revealed good intra and extra oral healing with no evidence of discharge or dehiscence. Facial, hypoglossal, and lingual nerves appeared intact. There was movement of jaw towards the left side on opening. Post-operative CT scans were taken showing, as shown in Figure 7.
Figure 8: Weeks Post-Operative Facial Bones CT Scan Showing Healing Following AVM and Left Mandibular Reception up to LR2 A) Coronal view, B) Axial view,, C) Sagittal view showing complete left mandibular resection up to articular disk and D: Coronal facial bones profile
A) Coronal view, B) Axial view, C) Sagittal view showing complete left mandibular resection up to articular disk and D: Coronal facial bones profile.
Discussion
Vascular malformations are abnormal congenital endothelial connections between arterial and venous systems that grow exponentially with age [1,2]. These malformations are usually asymptomatic as opposed to Haemangiomas which are fast growing in the early years of childhood [3,11]. Approximately 50% of all VM occur in the head and neck region owing to high vascularization in the area. Amongst the vascular malformations, high flow AVM are most commonly found and mostly encountered in head, and neck regions, limbs, and lungs. AVM in the head and neck region are rare and over half of the cases are exhibited in mandible more than maxilla. As opposed to Haemangiomas, AVM may manifest as slow growing, painless lesions, and may or may not cause facial asymmetry due to soft tissue or hard tissue expansion [1,3,4,6]. The patient in this case report had a similar presentation as the main complaint was profuse bleeding of intra¬oral origin. There was no associated pain, tenderness on percussion of teeth or tenderness on palpation of the soft tissues.
There was also the characteristic presentation of tooth mobility due to bone loss and root resorption, which are characteristic clinical and radiographical findings of mandibular AVM [2-4]. Gingival involvement in AVM is a misleading presentation as clinicians may focus on childhood periodontal issues, trauma, or foreign body, while there is a life threatening underlying vascular cause. This case report was similar as patient presented with excellent oral hygiene, caries free dentition and periodontal causes were excluded. AVM can be felt as hyperthermic lesions with pulsation and bruit, however, this was not found in our patient.1,3,11. The most indicative presentation of high-flow lesion such as AVM is the pulsating or bruit felt due to the velocity of arterial blood flow.11 This case would have been diagnosed faster if this small but significant information would have been given at the time of initial presentation by parents.
Although parents were comprehensive in documenting the extent of bleeding with reference photographs, the ‘pulsating, fountain-like spurt’ of blood was not mentioned by patient’s mother and came into light only following the OGD procedureRadiographic imaging is essential in diagnosing any head and neck lesions and can prove diagnostically effective, ranging from 2 to 3-dimensional imaging available with intricate delivery of details [4,6-8]. However, AVM are considered ‘radiographic imitators’ and may mimic various other soft and hard, odontogenic and non-odontogenic lesions of the head and neck [11]. Mandibular AVM appears as a diffused, poorly localized, multilocular, punched out, uncorticated radiolucency with loss of trabeculation, displaced teeth, root resorption and ID canal obliteration [4,6,11].
These correlated to the findings of OPG in this case report as it showed a diffused, poorly localized radiolucency without septae, loss of trabeculation, loss of lamina dura of teeth and displaced LL7. The findings in OPG can also mimic other radiolucent or mixed radiolucent/ radio-opaque lesions of the jaw such as ameloblastoma, myxoma, Central giant cell granuloma (CGCG) [3,5]. OPG in ameloblastoma and CGCG lesions show the characteristic honeycomb or soap bubble appearance which are also present in AVM. However, they are differentiated by perforation of cortex. In this case, there appeared to be cortical thinning and erosion, but perforation was not present [11]. Epithelial cysts such as traumatic bone cysts, haemorrhagic or aneurysmal bone cysts (ABC) were considered based on clinical presentation and initial OPG findings [1,3,4,7,11]. However, the characteristic pulsating spurting high flow bleeding ruled out the presence of cyst, along with the lack of multilocular appearance, sclerotic rimmed margin, discrete lining of endothelial cysts visible in OPG [6-8].
To combat the limitations of a 2-dimensional image of a OPG, CT scan and MRI scans prove useful due to high resolution, intricate detail delivery and 3 dimensional reporting. However, they pose risks associated with high radiation exposure [4,11]. CT scans indicate osteolytic expansive bony changes, bone marrow expansion and trabecular obliteration, which was consistent with the CT scan taken for our patient, revealing ipsilateral buccolingual expansion of left mandibular body, LL7 tooth displacement and ID canal obliteration. MRI scans are an invaluable diagnostic tool for vascular lesions as the use of MRI can determine the origin and course of abnormal vascular channels and branches and ascertain the flow characteristics (high or low flow lesion) [11]. MRI, albeit an excellent diagnostic tool for Vascular lesions, also expose patients (particularly young patients) to additional radiation exposure. As MRI can be used as a diagnostic tool by itself or prior to angiography before embolization procedure, it was decided to defer undertaking an MRI scan for diagnostic purposes only, especially as CT scan was sufficient to reach a diagnosis of AVM and provided intricate details and clarity in the vascular view. In our case, as Angiography and embolization was undertaken by Craniofacial team 2 days following presentation to our dental unit, double radiation exposure to MRI was deemed unnecessary.
The diagnostic tool, which could have been of great value to reach to an initial diagnosis (whilst waiting for CT scan results and reporting) and which was not undertaken was Doppler Ultrasonography. This tool is non-invasive, quick, and would have further cemented the diagnosis at very early stages by confirming a high-flow lesion by measuring velocity of blood flow and resistance of vessels. For reference, normal velocity for arteries and veins are 80 cm s-1 and is 30 cm s-1 respectively [11]. Overall, existence of atraumatic bleeding (sufficient to cause hypovolemic collapse) and expansile radiolucent radiographic changes warrants strong suspicion towards Vascular malformation and the typical ‘pulsatile, fountain-like, spurting’ blood flow creates a unidirectional path towards diagnosis of an AVM [1,3,4,11]. The critical part of management of AVM is early diagnosis (detailed and accurate history), investigations and prompt surgical treatment [3,4]. Nevertheless, all diagnostic tools available should be and were utilised to reach to a definitive diagnosis, however, parents recall of this characteristic of blood flow pattern at home would have made the time taken to provide a formal diagnosis much shorter in this case.
Newer treatment modalities of management of AVM include embolization of feeding vessels which was undertaken as the first surgical step in this case [1,9,10,13]. Resection of the jaw may be followed by partial Embolization due to better haemorrhagic control following this procedure [12,13]. The management of AVM needs to be planned as a phased approach as was undertaken in this case and overall treatment requires comprehensive consideration including risks of embolization (occlusion of cerebral or pulmonary vessels) and recurrence [2,4-6].Specifically, paediatric patients with craniofacial AVM, have seen major attention directed at the effectiveness of embolization in the recent years of AVM treatment. Embolization has become an essential component in the treatment of AVM being employed as definitive as well as supportive approach, as it can aid in haemorrhagic control and in minimizing the size of the malformation, by blocking or occluding the feeding vessels, for purposes of minimizing intra-operative haemorrhage and assisting in the identification of the VM margins [6,11].
The procedure, therefore, has been used prior to surgical resection to improve safety and efficacy. As the risk of complications is high in the paediatric age group, particularly because of the anatomy of the vessels in this population, embolization is a useful option to treat these high flow vascular lesions. These findings are echoed in the case report described herein because embolization was used in tandem with surgery, thus supporting the need for phased treatment to reduce danger associated with resection of highly vascular lesions.17 The management of the mandibular AVM in this case required cerebral angiography and embolization as an initial method of managing the extravasation associated with the lesion. Using the technique, the flow to the lesion was effectively minimized and safe resection was undertaken two days later.
However, intra-operatively, the procedure was complicated due to acute haemorrhage and approximately 1 litre of blood loss. This is the classic example of uncontrolled bleeding during or after a procedure with VM. However, as the AVM was embolized, the next ladder of the surgical procedure could be undertaken safely. This is in concordance with current research studies that reveal that preoperative embolization may help in reducing the risk of intraoperative blood loss and procedural complications [17-19]. Furthermore, more recent publications indicate that embolization is more effective at preventing relapse of AVM in the long term. In paediatric patient, haemorrhagic cerebrovascular pathologies were reviewed, and the investigation presented that embolization combined with surgery have low recurrence of AVM as compared to resection surgery only [18]. This is in line with phases of treatment discussed in this case report whereby embolization formed the background alongside providing a temporizing measure to surgery that aimed at resection of the AVM.
Block resection of the affected area has been suggested and carried out in this case in view of the extent of the vascular lesion and bone involvement (bony expansion) [11-13]. However, it comes with major functional damage to hard and soft tissues, growth, and psychological trauma. In this case, temporary reconstruction with alloplastic bone plates have been planned in the short term, depending on recovery and rehabilitation progress. Interim and long-term reconstruction options were briefly discussed with a rib graft or plate; however, immediate focus was on healing and psychological support for the child [13-15,19].
Post-surgical procedure recovery and rehabilitation requires MDT agencies and support. Owing to the extensive surgery and functional changes (mandibular resection, complications such as muscular collapse causing respiratory distress or dysphagia), patients are monitored in acute or critical care units, with analgesic and sedative support, as paediatric patients are less likely to comply with immediate post-operative advice and restrictions [19]. This was similar to the patient in our case study as patient was admitted for 2 weeks, sedated for 72 hours, and followed a soft diet for 4 weeks. Immediate paediatric psychologist support was arranged due to panic attacks and speech and language therapy initiated to address drooling and speech. Speech and language therapy addresses changes in musculature and hard tissues and aims to recover speech, swallowing and muscle tone, as was undertaken in this case. However, the patient is in the early stage of recovery and long-term challenges, complications or side effects cannot be determined yet[19]. The need to share this case report at this early stage of recovery and prior to other rehabilitation and reconstruction efforts was to disseminate information to clinicians to aid in faster diagnosis and eliminate unnecessary diagnostic procedures.
Conclusion
The dental manifestations of arteriovenous malformations in paediatric patients can significantly impact oral health and development, necessitating an urgent multidisciplinary approach for effective management. These manifestations may include delayed tooth eruption or displacement, tooth mobility, malocclusion, intra-oral bleeding gingival hyperplasia. Early diagnosis and intervention are crucial to mitigate complications and improve the quality of life for affected children. Regular dental evaluations and tailored treatment plans are essential to address the challenges posed by arteriovenous malformations in the dental context. Every stage of treatment, starting from history taking to post procedure rehabilitation and recovery, is important for patient care and well-being. An accurate and detailed history can aid in early diagnosis and intervention.References
- Rudagi, B. M., Patil, S., Hammannavar, R., & Jaiswal, T. (2016). Mandibular Arteriovenous Malformation in an 8-year-old Child: A Case Report. The Journal of Contemporary Dental Practice, 17(1), 85-89.
- Manjunath, S. M., Shetty, S., Moon, N. J., Sharma, B., Metta, K. K., Gupta, N., & Singh, S. (2014). Arteriovenous malformation of the oral cavity. Case reports in dentistry, 2014(1), 353580.
- Ramakrishnan, K., Palanivel, I., Narayanan, V., Chandran, S., &Gurram, P. (2021). Management of vascular malformations in the oral and maxillofacial region: a systematic review. Journal of Stomatology, Oral and Maxillofacial Surgery, 122(6), 588-599.
- Behravesh, S., Yakes, W., Gupta, N., Naidu, S., Chong, B. W., Khademhosseini,A., &Oklu, R. (2016). Venous malformations: clinical diagnosis and treatment. Cardiovascular diagnosis and therapy, 6(6), 557.
- Sham, M. E., & Sultana, N. (2012). Vascular anomalies in maxillofacial region. Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology, 24(3), 137-146.
- El-Ghanem, M., Kass-Hout, T., Kass-Hout, O., Alderazi, Y. J., Amuluru, K., Al-Mufti, F., ... & Gandhi, C. D. (2016). Arteriovenous malformations in the pediatric population: review of the existing literature. Interventional neurology, 5(3-4), 218-225.
- Li, X., Su, L., Wang, D., Gui, Z., Jiang, M., Yang, X., ... & Fan, X. (2020). Clinical and imaging features of intraosseous arteriovenous malformations in jaws: a 15-year experience of single centre. Scientific Reports, 10(1), 12046.
- Shamloo, N., Mashhadiabbas, F., &Safarpour, R. (2023). Mandibular Arteriovenous Malformation with Unusual Radiographic Appearance. Journal of Dentistry, 24(1), 66.
- Narang, S., Gupta, R., Narang, A., &Nema, R. N. (2012). Periodontal manifestations and management of a patient with AV malformation. Journal of Indian Society of Periodontology, 16(2), 266-270.
- Mitra, S., Shen, D., & Afshar, S. (2022). Assessing the Dental and Oral Health Needs at Vascular Anomalies Centers across the United States. Journal of Vascular Anomalies, 3(1), e031.
- Manjula, M., & Kumar, L. V. (2012). Arteriovenous malformation of the mandible and parotid gland: report of a case with a note on imaging findings. Dentomaxillofacial Radiology, dmfr_pap10279.
- Tanoue, S., Tanaka, N., Koganemaru, M., Kuhara, A., Kugiyama, T., Sawano, M., & Abe, T. (2023). Head and neck arteriovenous malformations: clinical manifestations and endovascular treatments. Interventional Radiology, 8(2), 23-35.
- Guerrero, W. R., Dandapat, S., & Ortega-Gutierrez, S. (2020). Hemorrhagic cerebrovascular pathology in the pediatric population. Frontiers in neurology, 11, 1055.
- Ota, Y., Lee, E., Sella, E., & Agarwal, P. (2023). Vascular malformations and tumors: a review of classification and imaging features for cardiothoracic radiologists. Radiology: Cardiothoracic Imaging, 5(4), e220328.
- Brahmbhatt, A. N., Skalski, K. A., & Bhatt, A. A. (2020). Vascular lesions of the head and neck: an update on classification and imaging review. Insights into imaging, 11, 1-18.
- Rosenberg, T. L., Suen, J. Y., & Richter, G. T. (2018). Arteriovenous malformations of the head and neck. Otolaryngologic Clinics of North America, 51(1), 185-195.
- Gennaro, P., Cioni, S., Bracco, S., Cascino, F., Latini, L., Abbagnato, M., ... & Gabriele, G. (2023). A Single-Center Retrospective Analysis of 14 Head and Neck AVMs Cases Treated with a Single-Day Combined Endovascular and Surgical Approach. Journal of Personalized Medicine, 13(5), 757.
- Jha, V. C., Alam, M. S., & Sinha, V. S. (2023). Comparative outcome of endovascular embolization with microsurgery in managing acute spontaneous cerebral hemorrhage in pediatric patients, an institutional experience. Child's Nervous System, 39(4), 963-974.
- Dimachkieh, A. L., &Chelius, D. C. (2022). Complex head and neck resection, reconstruction, and rehabilitation in children. Otolaryngol. Clin. N. Am, 55, 1205-1214.