Case Report - (2024) Volume 3, Issue 1
Case Report: A Severe Asthma Symptom with Biological Medication
2Institute of Forensic Science Forensic Medicine & Toxicology, Liaquat, University of Medical & Health Sciences, Pakistan
3Jeejal Maau Institute of Physiotherapy and Rehabilitation Sciences, Liaquat University of Medical & Health Sciences, Pakistan
Received Date: Nov 23, 2023 / Accepted Date: Dec 24, 2023 / Published Date: Jan 04, 2024
Copyright: ©Â©2024 Sajjad Ali Chang, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Chang, S. A., Balouch, A., Raja, A. A. (2024). Case Report: A Severe Asthma Symptom with Biological Medication. Archive Clin Med Microbiol, 3(1), 01-04.
Abstract
Research from Sindh Pakistan showed that 15% of patients are in therapy phases, many based on data from the specialized literature. According to the Global Initiative for Asthma, around 9.8% of grownup females and 6.1% of the male population suffer from asthma. In this instance, a 31-year-old patient who has had bronchial asthma for more than ten years and who has never had occupational exposure to the condition is presenting with persistent dyspnea at rest, wheezing, and mechanical ventilation. This patient has also had multiple hospitalizations in the Pneumology Service for severe exacerbations. One of these exacerbations required hospitalization in the Anesthesia and Intensive Care Service, orotracheal intubation, and mechanical ventilation manifesting with chest tightness, wheezing, and persistent dyspnea when at rest and obstructive ventilatory dysfunction with a 37.57% decrease in maximum expiratory volume per second and 156% reversibility. No acute lesions on the X-ray pleuro- pulmonary: 13,550 leukocytes; 5566 eosinophils per milliliter; 4107%. Allergy caused by immunoglobulin E (IgE) confirmed, and IgE total of 513 IU/L, no sensitivities to skin prick testing, according to an allergy consultation and epithelial allergy, herbatum, and fumigatus specific IgE. Bronchoscopic techniques were used to rule out pulmonary aspergillosis, and mycological tests came back negative. Treatment with omalizumab 300 mg per month began, with no need for hospitalizations, exacerbations, or systemic corticosteroid therapy.
Keywords
Pneumonia, Asthma Therapy, Immunoglobulin, Lung Bronchoscopy, Chest Tightness, Omalizumab Injection
Introduction
Approximately 9.8% of grownup females and 6.1% of the male population suffer from pneumonia (asthma), and it is the most prevalent serious illness in kids. There seem to be about 5.1 million youngsters under the age of 18 that are asthmatic. Asthma is almost three times more prevalent in black infants than in white kids [1-5]. A subtype of complicated asthma, exercise-induced asthma is bronchitis that is uncontrollable with the patient's perfect obedience to maximum medication, when threats have been avoided, and after complications have been under care. Reports from the literature show that 24% of subjects in research in Sindh, Pakistan are receiving treatment according to stage five of global initiatives for asthmatic regulation. Among them, 4% have mild asthma, and 17.5% have complicated asthma. Patients who have serious asthma have a bad prognosis because they frequently experience severe symptoms. [1,6-8]. Therefore, further biological treatment may change the outcome in suitable patients.
This development of a focused therapy based on the factors derived from the use of indicators in the identification of acute asthmatic disease is a chronic inflammatory [1,2,8]. The blood eosinophil level, serum IgE valuation, as well as serum periosteal are indeed the diagnostics used to identify patients with acute asthma endotype [8]. Irrespective of eosinophil counts, omalizumab lowers asthma attacks in people with severe asthma, as well as biological anti-interleukin 5 regulators (IL-5R) drugs are recommended for individuals who have serious lung disease, whatever the allergens, and Figure 1 gives you an idea about the normal lung and asthmatic lung [9]. While it is well-recognized that allergic bronchopulmonary abscess is a fungus consequence in asthmatics, exercise-induced asthma combined with fungal sensitization is a distinct condition [10]. Immunoglobulin E (IgE) levels in allergic pulmonary aspergillosis commonly surpass 1000 IU/mL [1,2,9]. There is no correlation between the detection of the fungus in spontaneous phlegm or broncho intake and the detection of allergies determined through prick studies or an array of pulmonary allergies.
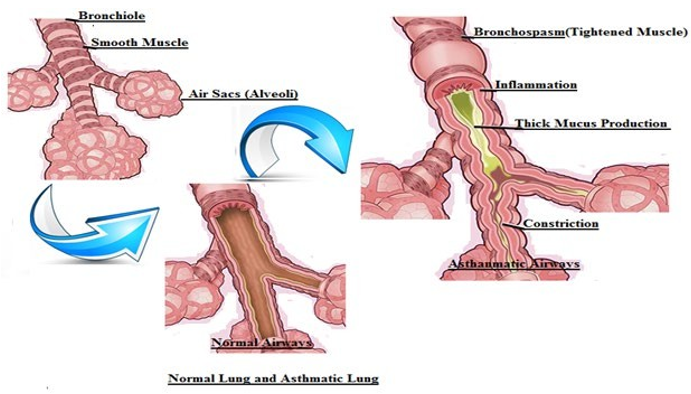
Figure 1: Shows the Diversity between Normal and Asthmatic Airways
Patient/Case Highlight
Consider the situation of a 31-year-old male serene who is receiving outpatient therapy with a long-acting B2 agonist corticosteroid for inhalation arrangement during a higher dosage intended for the respiratory disease after receiving a diagnosis of bronchitis and stage two arterial hypertensions more than ten years ago. A person's presence of persistent breathing dyspnea, whistling, as well as a feeling of chest tightness, and no occupational exposure to pulmonary pollutants was treated with antibiotics, intact corticosteroid medication, breathing therapy, as well as aerosolization amid foster, salbutamol in addition to fluticasone while hospitalized, with a marginally favorable outcome. During lung imaging, no significant cohort abnormalities were shown. Tiotropium respiratory therapy was also connected to LABA-ICS inhalation therapy after evacuation. The patient did, nevertheless, go on to experience multiple mild chronic conditions at home and in stressful environments that required treatment. Salmeterol 50 mg, fluticasone 500 mg, and tiotropium 18 mg (Spiriva 18 mg), as well as beclomethasone 250 mg, were added here to concomitant drug therapies, but the person's progression was still undesirable. After three months, the clinical presentation worsened with a severe exacerbation that necessitated admission to the acute care setting, oxygen therapy, and tracheostomy endotracheal tube.
The person needed recurrent rounds of use of oral corticosteroids (OCS) to aid bouts of respiration distress setting of exacerbation, keeping the same maximal guidelines for aerosol medication in leisure, resulting in the use of decided to reassess holder. The typical obstructive ventilator dysfunction is revealed by the updated pulmonary evaluation from the perspective of functional investigations, together with a significant reactionto bronchodilators reversibility of 158% and a decrease in 36.9% forced expiratory volume, leukocytosis (14,318/mL) and eosinophilia 5841/mL are seen in whole blood work, an immunoglobulin E (IgE) test quantifies the quantity of this particular kind of antibody. To recognize and get rid of microorganisms, the immune system uses antigens, also referred to as antibodies. Samples taken frequently show traces of IgE antibodies. Elevated doses could be a sign of the body overreacting to toxins that could trigger an allergic reaction. If the organism is combating a parasite infestation or some other immune response problem, IgE concentrations may be increased, and the calculated result was 687 IU/L. After a discussion with an allergologist, testing utilizing penicillium led to more accurate, fusarium oxysporum, and fungal fumigates indicating no allergy responses despite the array of pulmonary allergens revealing specific IgE to epithelium and guinea pig. The allergist suspected pulmonary aspergillosis because of this allergy to aspergillus, and therefore bronchoscopy was done to rule out that diagnosis in the absence of a positive bronchial aspirate mycological examination and suggestive endobronchial abnormalities.
Analyzing the inclusion and exclusion criteria is required to establish biological therapy, and injection of omalizumab treatment (not corticosteroid) was initiated for persistent allergic asthma because the patient meets all of the exclusion criteria: age of 6 years, substantiated asthma identification of at least a single year, IgE-mediated allergic response validated by specific IgE, asthma management optimization with at least four months of adoption, and unmanageable asthma. The first level of IgE as assessed earlier than beginning body weight and treatment is used to establish the proper dose and frequency of delivery. Therefore, omalizumab 300 mg, or depending on a particular of 150 mg, and a median time to accomplish a well-controlled outcome in 42 days, case study, was given to a patient who weighed 57 kg and had a total IgE level of 509 IU/L, satisfactory evolution, no local reactions, and no systemic reactions. Once every two weeks, the patient visits pulmonary care for treatment. Considering improved sensations, a significant decrease in systemic corticosteroid dosage, and increased respiratory capacities, as well as Table 1, shows spirometry values, a rise in values one was noted, compared to the baseline value, an act grade of 14, and no significant relapses, the history at 29 days was unquestionably encouraging. In one year, most symptoms of asthma had improved, and systemic corticosteroid medication had been eliminated from the treatment plan. Chronic oxygen treatment (COT) was not used at the residence, nor any exacerbations or hospitalizations. After beginning the bioremediation earlier, the patient is currently receiving nebulized therapy with salmeterol 50 mg, fluticasone 500 mg as prescribed, as well as 5 mg/d of tiotropium and add-on therapy with omalizumab 300 mg per month, and the patient has not used oral corticosteroids in the previous months and has not needed inpatient care for breathing pathogenesis.
|
|
|
July 2021 |
August 2021 |
July 2022 |
|||
|
S. No |
Force |
Pre |
Post |
Pre |
Post |
Pre |
Post |
|
1 |
Percentage of FVC |
83.5 |
97.3 |
87.2 |
102.8 |
81.1 |
98.6 |
|
2 |
FEV1 |
2.1 |
58.7 |
52.4 |
69.1 |
60.5 |
83.9 |
|
3 |
Percentage of FEV1/FVC |
53.93 |
54.4 |
59.2 |
70.13 |
67.4 |
80.2 |
|
4 |
FEF25 |
17.14 |
30.22 |
16.1 |
32.5 |
47.41 |
63.25 |
|
5 |
Percentage of FEF50 |
18.6 |
37.4 |
16.25 |
38.9 |
35.7 |
40.81 |
Table 1: Shows the Spirometry Values, Forced Vital Capacity (FVC), Forced Expiratory Volume (FEV1), Forced Expiratory flow (FEF).
Discussion
Medical studies have recently concentrated on phenotyping and genotyping severe asthma according to epidemiology, immunology, indicators, sensitivity to pharmaceutical therapies, and long¬term outcome [4,9]. These presumptions lead to the conclusion that the patient in the aforementioned case has significant eosinophilia and chronic asthma [2,3,8]. Nevertheless, in the era of individualized medication, numerous phenotypic subgroups can be identified under the clinical entity and indicators [11,13]. Blood eosinophils, as well as IgE levels therefore inpatient, are significantly greater, indicating difficulty breathing as its endotype [4,6,14]. The trial, reported in 2013, showed that omalizumab therapy has a stronger response than other biologic medicines in asthmatic patients who have a Th2 endotype and concomitant elevations in biomarkers, as in our situation [13,15]. However, the case's novelty was the presence of an Aspergillus allergy, which the allergist regarded as a pulmonary respiratory infection and, as a result, as an adverse reaction to therapeutic intervention. The most important aggravation risk for asthma and allergies is fungus sensitization. Several individuals with airway inflammation had fungally extracted in their bacterial isolates, proving that there is a connection between fungus sensitization and fungus colonizing in such people [9,16]. In this case, separated fungal sensitization was shown, but endoscopic growth wasn't.
The choice to provide systemic corticosteroid therapy, whether injectable or sublingual, should consider the risk-benefit ratio because these medications can cause several issues and side effects [2,4]. The patient is experiencing numerous side effects from receiving oral corticosteroids regularly, the use of therapies that have improved this patient's quality of life and prognosis by reducing the need for oral corticosteroid medication and controlling the severity of asthma, as well as biochemical therapy, should not be prohibited by aspergillus allergy; rather, it should serve as a starting point for additional research.
Declaration
The patients consented both written and orally to participate and agree to publish.
The Author declares no competing interest.
References
- GINA committee. (2022). Global Strategy for Asthma Management and Prevention 2022 Update. Global Initiative for Asthma: Fontana, WI, USA, 225.
- Yiltok, E. S., & Akhiwu, H. O. (2021). Asthma monitoring and lung function in children with physician-diagnosed asthma. Journal of Medicine in the Tropics, 23(1), 17.
- Fainardi, V., Caffarelli, C., Deolmi, M., Skenderaj, K., Meoli, A., Morini, R., ... & Esposito, S. (2022). Management of Preschool Wheezing: Guideline from the Emilia-Romagna Asthma (ERA) Study Group. Journal of Clinical Medicine, 11(16), 4763.
- World Health Organization (WHO) (2022). Model list of essential medicines.
- Razzaq, S., Nafees, A. A., Rabbani, U., Irfan, M., Naeem, S., Khan, M. A., ... & Burney, P. (2018). Epidemiology of asthma and associated factors in an urban Pakistani population: adult asthma study-Karachi. BMC pulmonary medicine, 18, 1-13.
- Aslam, R., Sharif, F., Baqar, M., Nizami, A. S., & Ashraf, U. (2023). Role of ambient air pollution in asthma spread among various population groups of Lahore City: a case study. Environmental Science and Pollution Research, 30(4), 8682-8697.
- Farooq, J., Khaliq, S. A., Ghuman, F., Shaikh, J., & Azhar, I. (2022). Study on Allopathic and Alternative Treatments of Asthma and Factors Influencing Treatment Choices. Evidence-Based Complementary and Alternative Medicine, 2022.
- Sultana, R., Ghani, M., Smith, A., Ashraf, S., & Bashir, S. (2022). Factors Influencing the Asthma Control among Children. Medical Research Archives, 10(7).
- Khan, J., Adil, M., Wang, G., Tsheten, T., Zhang, D., Pan, W., ... & Wu, Y. (2022). A cross-sectional study to assess the epidemiological situation and associated risk factors of dengue fever; knowledge, attitudes, and practices about dengue prevention in Khyber Pakhtunkhwa Province, Pakistan. Frontiers in Public Health, 10, 923277.
- Menzella, F., Galeone, C., Formisano, D., Castagnetti, C., Ruggiero, P., Simonazzi, A., & Zucchi, L. (2017). Real-life efficacy of omalizumab after 9 years of follow-up. Allergy, Asthma & Immunology Research, 9(4), 368-372.
- Loureiro, C. C., Amaral, L., Ferreira, J. A., Lima, R., Pardal, C., Fernandes, I., ... & Arrobas, A. (2018). Omalizumab for severe asthma: beyond allergic asthma. BioMed research international, 2018.
- Katsaounou, P., Buhl, R., Brusselle, G., Pfister, P., Martínez, R., Wahn, U., & Bousquet, J. (2019). Omalizumab as alternative to chronic use of oral corticosteroids in severe asthma. Respiratory medicine, 150, 51-62.
- Hogan, C., & Denning, D. W. (2011, December). Allergic bronchopulmonary aspergillosis and related allergic syndromes. In Seminars in respiratory and critical care medicine (Vol. 32, No. 06, pp. 682-692). © Thieme Medical Publishers.
- Lin, C. H., & Cheng, S. L. (2016). A review of omalizumab for the management of severe asthma. Drug design, development and therapy, 2369-2378.
- Froidure, A., Mouthuy, J., Durham, S. R., Chanez, P., Sibille, Y., & Pilette, C. (2016). Asthma phenotypes and IgE responses. European Respiratory Journal, 47(1), 304-319.
- Gauthier, M., Ray, A., & Wenzel, S. E. (2015). Evolving concepts of asthma. American journal of respiratory and critical care medicine, 192(6), 660-668.



