Review Article - (2019) Volume 1, Issue 1
Biomarkers for Early Diagnosis of Acute Kidney Injury
2Department of Nephrology, Theodor Bilharz Research Institute, Ministry of Scientific Research, Egypt
Received Date: Dec 07, 2019 / Accepted Date: Dec 12, 2019 / Published Date: Dec 19, 2019
Copyright: ©Bothina S Madkour, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Acute kidney injury (AKI) is a common and potentially life-threatening conditionassociated with morbidity and mortality. Currently, the standard diagnostic tools for the detection of AKI are monitoring of urinary output and serum creatinine (sCr), both of which are markers of kidney function but not kidney injury. Novel AKI biomarkers have made significant contributions to our understanding of the molecular under pinnings of AKI, they could also have use as molecular phenotyping tools that facilitate the identification of patients who could benefit from a specific intervention, even a biomarker-targeted intervention and they were proved to be useful in facilitating early diagnosis, guiding targeted interventions and monitoring the disease progression and resolution.
Keywords
Acute Kidney Injury, Renal Biomarkers
Introduction
Acute kidney injury (AKI) is a common and potentially life-threatening condition associated with morbidity and mortality as well as with unfavorable long-term outcomes caused by the development of chronic kidney disease (CKD) or the occurrence of cardiovascular events [1].
At the time of AKI diagnosis, a number of diagnostic and therapeutic measures are needed for determination of the underlying cause of AKI and the initiation of specific and supportive therapeutic measures, such as antibiotic therapy for sepsis, immunosuppression for autoimmune disease, an adjustment of nephrotoxic drugs and directed fluid management [2].
As early institution of these measures is critical for their effectiveness, efforts have been made to identify subtle insults to the kidney that do not cause measurable functional decline, that is subclinical forms of AKI, and to identify indicators of a particular risk of AKI [3].
Despite these efforts, there is still little implementation of this knowledge in daily clinical practice [4]. Currently, the standard diagnostic tools for the detection of AKI are monitoring of urinary output and serum creatinine concentration (sCr), both of which are markers of kidney function but not kidney injury [5].
Accordingly, AKI is defined by an increase of sCr by ≥0.3 mg/dl in 48 h or an increase by ≥1.5 fold from a known or assumed baseline or by a decrease of urinary output to less than 0.5 ml/kg/h for 6 h [6].
In clinical reality, however, serial measurements of sCrare often unavailable complicate the differentiation between AKI and CKD. In addition, sCr and urinary output when measured at presentation do not always predict adverse outcomes, such as hospital mortality or a requirement for renal replacement therapy [6].
Furthermore, all these measures does not account for the etiology of AKI. Most importantly, it does not differentiate between quickly reversible, volume-sensitive reductions in glomerular filtration rate “pre-renal AKI” and primary structural injury to the kidney “intrinsic AKI” [4].
One major advantage to detect AKI at an earlier stage would be the implementations of new reliable biomarkers that identify AKI earlier than conventional tests or that detect subclinical AKI [1].
Based on sCr, kidney injury diagnosis occurs only at the stage of reduction of GFR and increased sCr, after a greater degree of renal injury, with a reduction of at least 30% in GFR. After an abrupt decrease in GFR, there is a delay of days until sCrrises [7].
New Biomarkers of AKI
Novel AKI biomarkers have made significant contributions to our understanding of the molecular underpinnings of AKI, they could also have use as molecular phenotyping tools that facilitate the identification of patients who could benefit from a specific intervention, even a biomarker-targeted intervention [8].
They were proved to be useful in facilitating early diagnosis, guiding targeted interventions and monitoring the disease progression and resolution [9].
According to Nguyen and Devarajan, Characters of clinically applicable AKI biomarkers could be summarized [10].
- Non-invasive, Rapidly measurable.
- Using easily accessible samples.
- Sensitive and using standardized clinical assay platforms.
- Have a wide dynamic range and cut-off values that allow for risk stratification.
- Specific for AKI, to differentiate intrinsic AKI from pre-renal azotemia & CKD.
- Predictive of outcomes e.g. dialysis, length of hospital stay, and mortality.
- Able to guide initiation of therapies and facilitate monitoring the response.
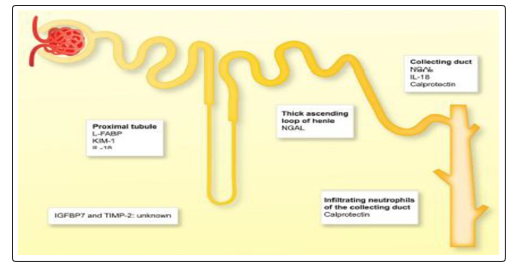
Figure 1: Sites of origin of AKI biomarkers. L-LABP: Liver-Fatty Acid Binding Protein, KIM-1: Kidney Injury Molecule-1, NGAL: Neutrophi Gelatinase Associated Lipocalin, IL-18: Interleukin-18, IGFBP7: Insulin-like Growth Factor-Binding Protein7, TIMP2: Tissue Inhibitor of Metalloproteinases 2.
Table 1: Overview of the most recent and promising biomarkers for early detection of AKI (42). EO: endogenous ouabain, ER: emergency room, FABPs: fatty acid-binding proteins, ICU: intensive care unit, IGFBP7: insulin-like growth factorbinding protein 7, IL-18: interleukin-18, KIM-1: kidney injury molecule-1, NGAL: neutrophil gelatinase-associated lipocalin, ROC: receiver operating characteristic curve, TIMP2: tissue inhibitor of metalloproteinases 2, Tx: transplantation.
|
Biomarker |
Settings Studied |
Source |
Measured from |
Used for |
Diagnostic accuracy (ROC) |
|
NGAL |
Cardiac surgery, ER, hospitalized patients, kindly Tx, sepsis, critically-ill patients |
Leukocytes, loop of henle and collecting ducts |
Serum plasma Urine |
Detection of established AKI, early diagnosis, prognosis |
0.53-0.96 |
|
Cystatin-C |
Hospitalized patients, cardiac surgery |
Nucleated cells |
Serum plasma Urine |
Detection of established AKI, early diagnosis, prognosis |
0.79-0.89 |
|
KIM-1 |
Hospitalized patients, cardiac surgery |
Proximal tubular cells |
Urine |
Increased risk of AKI, established AKI, prognosis |
0.61-0.78 |
|
IL-18 |
Cardiac surgery, ICU, hospitalized patients, Tx |
Monocytes, dendritic cells, macrophages |
Urine |
Detection of established AKI, early diagnosis, prognosis |
0.70-0.95 |
|
FABPs |
Contrast nephropathy, Sepsis, cardiac surgery, ischemic/ reperfusion injury |
Hepatocytes, proximal tubular cells |
Urine |
Detection of established AKI, progression to CKD |
0.84-0.96 |
|
TIMP-2 and IGFBP7 |
Major surgery, sepsis, shock, trauma |
Tubular epithelial cells |
Urine |
Detection of established AKI, prognosis |
0.79-0.85 |
|
EO |
Cardiac surgery |
Adrenal cells |
Plasma |
Identification of patients with increased risk of AKI |
0.73-0.80 |
Neutrophil Gelatinase-Associated Lipocalin (NGAL)
NGAL is a 25-kDa protein of the lipocalin family. Initially identified bound to gelatinase in specific granules of the neutrophils (Figure 2). It is synthesized during a narrow window of granulocyte maturation in the bone marrow, but it may also be induced in epithelial cells in the setting of inflammation or malignancy [9].
NGAL should be considered as a marker of tubular damage. NGAL (also known as lipocalin 2 or lcn2) to be one of the most upregulated genes in the kidney very early after acute injury especially after ischemic or nephrotoxic AKI [11].
The elevation of NGAL is detectable as early as 3 h after the injury and it peaks at approx. 6–12 h after injury depending on the severity of injury. The elevation can persist up to 5 days after the initial injury when the injury is severe [12].
NGAL is the most widely studied biomarker of AKI. It has shown its performance in various settings such as the prediction of AKI in pediatric and adult cardiac surgery patients, in critically ill patients, in patients in the emergency roomas well as in the kidney transplant setting [13].
Elevated urinary NGAL levels at AKI diagnosis predicted long-term adverse outcomes of ESRD or death [4].
Plasma NGAL measurements may be influenced by a number of coexisting variables such as CKD, chronic hypertension, systemic infections, and inflammatory conditions. However, the increase in plasma NGAL in these situations is generally much less than those typically encountered in intrinsic AKI [4].
Although it is not clear which test (urine vs. plasma sample) provides the best diagnostic performance for AKI. Some authors have suggested that a combination of the two tests might be the best option [14].
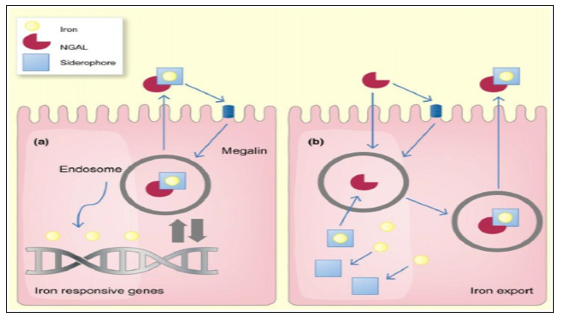
Figure 2: Schematic model of the functions of neutrophil gelatinase associated lipocalin. NGAL: Neutrophil Gelatinase Associated Lipocalin [2].
Cystatin-C (Cys-C)
A 13-kDa protein and is believed to be one of the most important extracellular inhibitors of cysteine proteases which is freely filtered by the glomerulus, reabsorbed and catabolized, but not secreted, by the tubules [9].
Urinary Cys-C levels have been found to be elevated in individuals with known tubular dysfunction with high predictive value of poor outcome [15].
Koyner et al, (2008) stated that within the first 6 h of renal injury, urinary values of Cys-C rise predicting AKI, but no change was observed in plasma levels, suggesting that the urinary test might be superior to the plasma assay for the early detection of AKI and when it is compared withsCr, Cys-C seems to be less affected by age, gender, and body weight [16]. Serum levels of Cys-C are a more precise indicator of kidney function than s.Cr levels but seem to be influenced by large doses of corticosteroids, hyperthyroidism, inflammation, hyperbilirubinemia and hypertriglyceridemia [17].
Kidney Injury Molecule-1 (KIM-1)
Is a type I cell membrane glycoprotein containing a unique six-cysteine immunoglobulin-like domain and a mucin domain in its extracellular region. Urinary Kim-1 has been used as an early and promising diagnostic biomarker of kidney injury and its clinical outcomes in hospitalized patients. Its concentrations were significantly increased within 12 hours. The recent availability of a rapid urine dipstick test for KIM-1 will facilitate its further evaluation in preclinical and clinical studies [18].
Han et al, (2004) demonstrated marked expression of KIM-1 in kidney biopsy specimens with acute tubular necrosis (ATN), and elevated levels within 12 h after an initial ischemic renal insult, prior to the appearance of casts in the urine [19].
In sum, a soluble form of human KIM-1 can be detected in the urine of patients with ATN and can serve as a useful biomarker in kidney proximal tubular damage, facilitating the early diagnosis and serving as a differential diagnosis of renal injury [20].
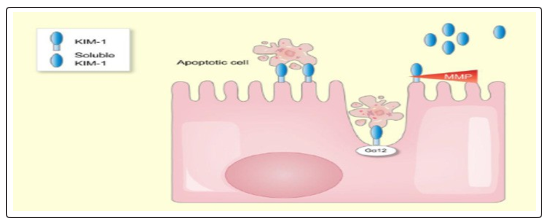
Figure 3: Schematic model of the functions kidney injury molecule-1. KIM-1: Kidney injury molecule-1
Liver-Type Fatty Acid Binding Protein (FABPs)
Small (15 kDa) cytoplasmic proteins abundantly expressed in all tissues with active fatty acid metabolism [21]. Two types of FABP have been identified in the human kidney: liver-type FABP (L-FABP) in the proximal tubule and heart-type FABP (H-FABP) in the distal tubule. Free fatty acids (FFAs) in proximal tubules are bound to cytoplasmic FABPs and transported to mitochondria or peroxisomes, where they are metabolized by β-oxidation. Besides its transport function, L-FABP also protects cells from oxidative stress induced by H2O2 [22].
In children undergoing cardiac surgery who subsequently developed AKI, urine L-FABP concentrations were significantly increased within 4 hours of the surgery with higher levels in patients with poor outcome [23].
In patients with septic shock and AKI, urinary L-FABP measured at admission was significantly higher in the non survivors than in the survivors, thus, emerging data point to L-FABP as a promising urinary biomarker of AKI and its outcomes. However, the urinary excretion of L-FABP is also increased in the setting of CKD [24].
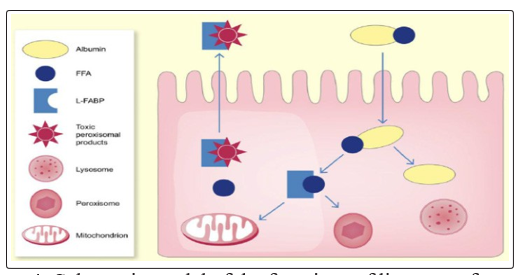
Figure 4: Schematic model of the functions of liver-type fatty acid-binding protein (L-FABP). FFAs: Free fatty acids [4].
Interleukin-18 (Interleukin-18)
Also known as interferon-gamma inducing factor is a 24-kDa cytokine that belongs to the interleukin-1 superfamily. First synthesized as an inactive precursor without a signal peptide and it remains intracellular until its cleavage by caspase-1 and its subsequent secretion by monocytes/macrophages [25].
Caspase-1 acts as a component of inflammasome that mediates cleavage and release of interleukins in response to extrinsic stimuli. Several cell surface receptors such as TLRs, retinoic acid-induced gene-like receptors, nucleotide-binding domain-leucine-rich repeat, scavenger receptors and C-type lectins can start the cascade [26].
Cleaved IL-18 exerts a prion flammatory effect by signal transduction through the IL-18 receptor. IL-18 is also produced by the intercalated cells of the collecting ducts in the healthy kidney [27].
Because of the pathophysiological plausibility of IL-18 in the development and progression of AKI, which was reported in different rodent animal models, IL-18 was suggested to be a new biomarker in AKI. IL-18- deficient mice are protected from ischemia/reperfusion-induced AKI [28].
The administration of this IL-18 binding protein just before ischaemia/reperfusion injury ameliorated kidney damage in rats. Renal IL-18 mRNA levels have been shown to be significantly upregulated following ischemia–reperfusion injury, inflammatory/ autoimmune nephritis, and cisplatin-induced nephrotoxicity [29]. IL-18 has been shown to be more elevated in patients with established acute tubular necrosis AKI than in those with prerenal azotemia, urinary tract infection, or chronic kidney disease (CKD) [30].
In sum, even if the prognostic and diagnostic value of IL-18 is limited, the use of anti-IL-18 treatment may be a potential future AKI treatment option.
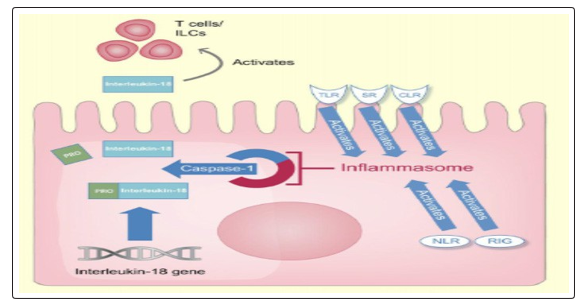
Figure 5: Schematic model of the functions of IL-18. RIG: Retinoic acid-induced gene-like receptor, TLRs: Toll-like receptors, SR: Scavenger receptors, CLR: Cell-type lectin receptor, NLR: nucleotide-binding domain-leucine-rich repeat [4].
Tissue Inhibitor of Metalloproteinase-2 (TIMP-2)&Insulin-like Growth Factor-Binding Protein 7 (IGFBP7)
TIMP-2 Is a member of the tissue inhibitor of metalloproteinase (TIMP) family, which is endogenous inhibitors of metalloproteinase activities. IGFBP7, a 29-kDa 14 secreted protein, is known to bind to and inhibit signaling through IGF-1 receptors [31].
They are markers of cellular stress in the early phase of tubular cell injury caused by a wide variety of insults as inflammation, ischemia, oxidative stress, drugs, and toxins. Therefore, both markers are involved in the process of G1 cell-cycle arrest that prevents cells from dividing in the case of damage to the DNA until such damage can be repaired [32].
The finding that IGFBP7 and TIMP-2 are enriched in the urine of patients at risk of AKI, the site of synthesis of these molecules in the setting of AKI is unknown [4]. While Kashani speculated that IGFBP7 and TIMP-2 are synthesized by renal tubular cells there is no scientific evidence to support this [33].
It was found that the product of TIMP-2 and 15 IGFBP7 was superior to other biomarkers (NGAL, plasma Cys- C, urine KIM-1, urine IL-18, urine pi-GST, urine L-FABP given the promising performance of these markers in some clinical studies, research into their pathophysiology will be a major priority [4].
Endogenous Ouabain (EO)
EO is a neuroendocrine hormone synthesized in the adrenal cortex that modulates the activity of Na, K-ATPase and induces signal transduction via sodium-calcium exchange [34]. The hypertensive effect of EO is well established in both animal and human models as well as its association with organ damage [35]. Furthermore, a rat model of ouabain-induced hypertension exhibited reduced creatinine clearance, proteinuria, and impaired podocytenephrin expression; thus, elevated EO per se maybe a direct cause of podocyte damage [9].
A significant association has been reported of preoperative EO levels with adverse renal outcomes in cardiac surgery patients and with mortality in critically-ill patients. In one study, elevated preoperative EO levels were associated with a higher incidence of postoperative AKI and ICU stay in a second study [36]. Finally, post-operative EO levels were also associated with a higher mortality rate after cardiac surgery [37].
Calprotectin
Is a 24 kDa heterodimer formed from the two monomers S100A8 (10 835 Da) and S100A9 (13 242 Da) [38]. Intracellular calprotectin’s main function is to interact with the cytoskeleton whereas when is secreted by activated immune cells it acts as a danger-associated molecular pattern protein [39].
It has been shown that renal collecting duct epithelial cells produce S100A8 and S100A9 in a model of kidney injury in response to unilateral ureteral obstruction (UUO) [40].
S100A8 and S100A9 are also induced in response to ischaemia reperfusion injury in mice.Infiltrating kidney neutrophils are the main source of S100A8/9 in the ischaemic kidney. S100A9-knockout mice, which lack active calprotectin, show an increased transition to renal fibrosis in response to ischaemia reperfusion injury, while the initial renal injury is similar to wild-type mice [41].
Ebbing et al, (2016) investigated time-dependent changes of calprotectin in patients undergoing nephron-sparing surgery for kidney tumours, which leads to iatrogenic renal ischaemia reperfusion injury due to transient clamping of the renal artery. Calprotectin concentrations started to be significantly increased at the end of the operation, approximately 2 h after ischaemia, and reached maximal levels 48 h post-surgery, with a 69-fold increase over baseline in calprotectin levels with significantly increased its level 5 days after surgery [42].
In the clinical interpretation of urinary calprotectin findings, one should be aware that there are two clinical settings other than AKI that lead to an increase of calprotectin: as calprotectin is predominantly derived from neutrophils and monocytes, pyuria substantially increases urinary calprotectin. Moreover, urothelial carcinoma is associated with increased concentrations [43].
In sum, the diagnostic accuracy of calprotectin in its ability of distinguishing pre-renal from intrinsic AKI showed a very high accuracy in predicting intrinsic AKI [44].
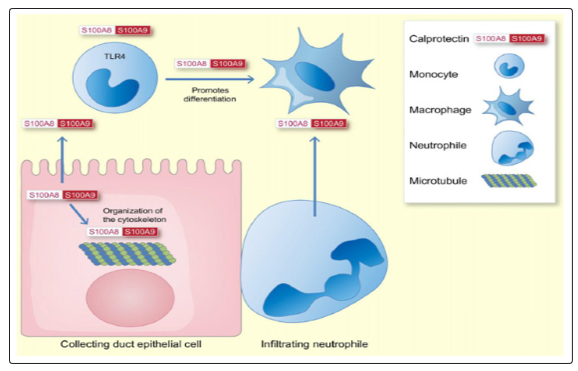
N-Acetyl-β-d-Glucosaminidase (NAG)
Lysosomal enzyme predominantly found in proximal tubules, so that the increased activity of this enzyme in urine suggests tubular cell injury and, therefore, it can serve as a specific urinary marker for these tubular cells [7].
The increase in urinary NAG activity indicates damage to the tubular cells, although it may also reflect an increased lysosomal activity without cell damage. Increased urinary excretion of NAG was reported in acute kidney disease of various etiologies, induced by toxic agents, after cardiac surgery and after kidney transplantation [45].
It is to be noted that the use of NAG remains limited by the fact that the urinary excretion of the enzyme is also high in diseases such as diabetic nephropathy, hyperthyroidism and rheumatic diseases [46].
Netrin-1
One of the kidney injury biomarkers, a laminina-related molecule little expressed in tubular epithelial cells of normal kidneys, however, it is highly expressed and excreted in the urine after AKI in animals [7].
Furthermore, significantly higher levels of netrin-1 were found in urine samples from patients with ischemic AKI induced by radio-contrast agents, sepsis and drugs compared with healthy controls [7].
Therefore netrin-1 is a promising urinary biomarker that rises early on for the detection of renal injury and can also serve as a universal biomarker for AKI [47].
Vanin-1
An epithelial ectoenzyme participates in the response to oxidative stress in vivo and catalyzes the conversion of pantetheine to pantothenic acid (vitamin B5) and cysteamine [7].
It is discovered the existence of increased levels of kidney vanin-1 mRNA in rats with ischemia-reperfusion type of lesion. In addition it showed increased levels of renal vanin-1 in rats with streptozotocin-induced diabetic nephropathy and in patients with diabetic nephropathy [48]. It has been found that elevated urinary concentration of vanin-1 occurs before conventional markers in rats with nephrotoxin-induced lesions, Therefore, it appears that urinary vanin-1 may be a potential biomarker for early detection of AKI [49].
To address this issue, it has been found that the urinary vanin-1 was detected prior to elevation of s.Cr, NAG, NGAL and KIM-1 in two well established animal models of drug-induced AKI [50].
Angiotensinogen
Recently described as a novel prognostic biomarker of AKI, although it has not yet been investigated as an early diagnostic biom arker [8].
The findings regarding the prognostic significance of angiotensinogen have strong implications for the molecular and cellular mechanisms underlying renal injury in AKI and could lead to novel interventional approaches [8].
Activation of the renin-angiotensin system (RAS) has long been recognized as an important contributor to chronic renal injury. Urinary angiotensinogen has been proposed as a marker of intrarenal RAS activity, and it is predictive of progression of CKD [51].
The prognostic significance of urinary angiotensinogen as an AKI biomarker is strongly suggestive of a role for RAS activation in modulating the severity of AKI. Given the clearly defined role for the RAS in CKD progression, it is plausible that RAS activation not only modulates the severity of an episode of AKI but also mediates progression to chronic disease [8].
Other New Potential Biomarkers
Recently, some new potential biomarkers have been proposed for early determination of AKI in specific conditions. Clusterin, osteopontin, glutathione-S-transferase (GST) and pyruvate kinase M2 [52-55]. All were associated with the development of drug-induced nephrotoxicity [55]. However, further investigations are needed to confirm these relationships and the potential benefits of these new molecules.
Conclusion
AKI is a very dangerous complication. It is associated with an increased risk of mortality and morbidity, and longer hospital stay, requires additional treatment, and increases the costs of the heath care. Unfortunately, the heterogeneity of AKI subtypes poses a great limit for large population studies in human subjects. In this setting, the use of classic clinical predictive models associated with novel renal biomarkers may well be the only way to refine the methods of treatment and improve the prognosis of patients. Introduction of novel biomarkers of AKI into the clinical setting is crucial for earlier diagnosis and improved risk assessment. The purpose of this review was to help clarify the biological basis of new AKI biomarkers that might contribute to improving the early detection or diagnosis of this pathology. But before biomarkers can be advocated for the diagnosis of AKI, further research is needed. Our understanding of how to prevent and manage AKI in an optimal way requires additional effort.
Recommendations
The discovery of new markers of glomerular and tubular function, tubular damage and inflammation allows a much better description and characterization of AKI than traditional markers of renal function can offer. It is therefore very likely that they will be incorporated into future definitions and classifications of AKI. As indicators of specific pathophysiological processes within the kidney, some of the new biomarkers also offer the opportunity to be used as diagnostic tools to identify the etiology of AKI. However, a single biomarker is unlikely to be useful. Instead, a panel of functional and damage biomarkers in combination with traditional markers of renal function and clinical judgment will provide best results. Some of these biomarkers also have the potential to facilitate the development of new drugs by indicating renal injury earlier than conventional methods.
References
- Chawla LS, Eggers PW, Star RA, Paul L Kimmel (2014) Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med 371: 58-66.
- Murugan R, Karajala-Subramanyam V, Lee M, Sachin Yende, Lan Kong, et al. (2010) Genetic and Inflammatory Markers of Sepsis (GenIMS) Investigators. Acute kidney injury in non-severe pneumonia is associated with an increased immune response and lower survival. Kidney Int 77: 527-535.
- Chawla LS, Goldstein SL, Kellum JA, Claudio Ronco (2015) Renal angina: concept and development of pretest probability assessment in acute kidney injury. Crit Care 19: 93.
- Schrezenmeier EV, Barasch J, Budde K, Westhoff T, Schmidt-Ott KM (2017) Biomarkers in acute kidney injury–patho physiological basis and clinical performance. Acta Physiol (Oxf) 3: 554-572.
- Waikar SS, Betensky RA, Bonventre JV (2009) Creatinine as the gold standard for kidney injury biomarker studies? Nephrol Dial Transplant 24: 3263-3265.
- Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120: c179-c184.
- Luis ABP, AdemarDdJ, Alex JS (2013) Biomarkers of acute kidney injury. J Bras Nefrol 3: 229-236.
- Joseph LA, John MA (2015) Biomarkers of AKI: A Review of Mechanistic Relevance and Potential Therapeutic Implications. Clin J Am SocNephrol 10: 147-155.
- Simona P, Marco S, Paolo M (2018) Predicting acute kidney injury: current status and future challenges. J Nephrol 31: 209-223.
- Nguyen MT, Devarajan P (2008) Biomarkers for the early detection of acute kidney injury. Pediatr Nephrol 23: 2151-2157.
- Cruz DN, de Cal M, Garzotto F, Perazella MA, Lentini P, et al. (2010) Plasma neutrophil gelatinase-associated lipocalin is an early biomarker for acute kidney injury in an adult ICU population. Intensive Care Med 36: 444-451.
- Parikh CR, Coca SG, Thiessen-Philbrook H, Shlipak MG, Koyner JL, et al. (2011) Postoperative biomarkers predict acute kidney injury and poor outcomes after adult cardiac surgery. J Am SocNephrol 22: 1748-1757.
- Zhou F, Luo Q, Wang L, Han L (2016) Diagnostic value of neutrophil gelatinase-associated lipocalin for early diagnosis of cardiac surgery-associated acute kidney injury: a meta-analysis. Eur J CardiothoracSurg 49: 746-755.
- Shaw A (2012) Update on acute kidney injury after cardiac surgery. J Thorac Cardiovasc Surg 143: 676-681.
- Conti M, Moutereau S, Zater M, Lallali K, Durrbach A, et al. (2006) Urinary cystatin C as a specific marker of tubular dysfunction. ClinChem Lab Med 44: 288-291.
- Koyner JL, Bennett MR, Worcester EM, Ma Q, Raman J, et al. (2008) Urinary cystatin C as an early biomarker of acute kidney injury following adult cardiothoracic surgery. Kidney Int 74: 1059-1069.
- Charlton JR, Portilla D, Okusa MD (2014) A basic science view of acute kidney injury biomarkers. Nephrol Dial Transplant 29: 1301-1311.
- Vaidya VS, Ozer JS, Dieterle F, Collings FB, Ramirez V, et al. (2010) Kidney injury molecule-1 outperforms traditional biomarkers of kidney injury in preclinical biomarker qualification studies. Nat Biotechnol 28: 478-485.
- Han WK, Bailly V, Abichandani R, Thadhani R, Bonventre JV (2002) Kidney Injury Molecule-1 (KIM-1): a novel biomarker for human renal proximal tubule injury. Kidney Int 62: 237-244.
- Ichimura T, Asseldonk EJ, Humphreys BD, Gunaratnam L, Duffield JS, et al. (2008) Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J Clin Invest 118: 1657-1668.
- ChmurzyÅ?ska A (2006) The multigene family of fatty acid-binding proteins (FABPs) function, structure and polymorphism. J Appl Genet 47: 39-48.
- Wang G, Gong Y, Anderson J, Sun D, Minuk G, et al. (2005) Antioxidative function of L-FABP in L-FABP stably transfected change liver cells. Hepatology 42: 871-879.
- Portilla D, Dent C, Sugaya T, Nagothu KK, Kundi I, et al. (2008) Liver fatty acid-binding protein as a biomarker of acute kidney injury after cardiac surgery. Kidney Int 73: 465-472.
- Kamijo A, Sugaya T, Hikawa A, Yamanouchi M, Hirata Y, et al. (2006) Urinary liver-type fatty acid binding protein as a useful biomarker in chronic kidney disease. Mol Cell Biochem 284: 175-182.
- Novick D, Kim S, Kaplanski G, Dinarello CA (2013) Interleukin-18, more than a Th1 cytokine. Semin Immunol 25: 439-448.
- Chang A, Ko K, Clark MR (2014) The emerging role of the inflammasome in kidney diseases. Curr Opin Nephrol Hyperten 23: 204-210.
- Franke EI, Vanderbrink BA, Hile KL, Zhang H, Cain A, et al. (2012) Renal IL-18 production is macrophage independent during obstructive injury. PLoS One 7: e47417.
- Wu H, Craft ML, Wang P, Kate R Wyburn, Gang Chen, et al. (2008) IL-18 contributes to renal damage after ischemia-reperfusion. J Am Soc Nephrol 19: 2331-2341.
- Wang J, Long Q, Zhang W, Chen N (2012) Protective effects of exogenous interleukin 18- binding protein in a rat model of acute renal ischemia-reperfusion injury. Shock 37: 333-340.
- Parikh CR, Mishra J, Thiessen-Philbrook H, Dursun B, Ma Q, et al (2006) Urinary IL-18 is an early predictive biomarker of acute kidney injury after cardiac surgery. Kidney Int 70: 199-203.
- Evdokimova V, Tognon CE, Benatar T, Yang W, Krutikov K, et al. (2012) IGFBP7 binds to the IGF-1 receptor and blocks its activation by insulin-like growth factors. Sci Signal 5: ra92.
- Yang QH, Liu DW, Long Y, Liu HZ, Chai WZ, et al. (2009) Acute renal failure during sepsis: potential role of cell cycle regulation. J Infect 58: 459-464.
- Kashani K, Al-Khafaji A, Ardiles T, Artigas A, Bagshaw SM, et al. (2013) Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit Care 17: R25.
- Dostanic-Larson I, Van Huysse JW, Lorenz JN, Lingrel JB (2005) The highly conserved cardiac glycoside binding site of Na, K-ATPase plays a role in blood pressure regulation. ProcNatlAcadSci 102: 15845-15850.
- Villa L, Buono R, Ferrandi M, Molinari I, Benigni F, et al. (2016) Ouabain contributes to kidney damage in a rat model of renal ischemia-reperfusion injury. Int J MolSci 17: 1728.
- Bignami E, Casamassima N, Frati E, Chiara Lanzani, Laura Corno, et al. (2013) Preoperative endogenous ouabain predicts acute kidney injury in cardiac surgery patients. Crit Care Med 41: 744-755.
- Simonini M, Pozzoli S, Bignami E, Nunzia Casamassima, Elisabetta Messaggio, et al. (2015) Endogenous ouabain: an old cardiotonic steroid as a new biomarker of heart failure and a predictor of mortality after cardiac surgery. Biomed Res Int 2015: 714710-714793.
- Stríz I, Trebichavský I (2004) Calprotectin – a pleiotropic molecule in acute and chronic inflammation. Physiol Res 53: 245-253.
- Ehrchen JM, Sunderkötter C, Foell D, Vogl T, Roth J (2009) The endogenous Toll-like receptor 4 agonist S100A8/S100A9 (calprotectin) as innate amplifier of infection, autoimmunity, and cancer. J Leukoc Biol 86: 557-566.
- Fujiu K, Manabe I, Nagai R (2011) Renal collecting duct epithelial cells regulate inflammation in tubulointerstitial damage in mice. J Clin Invest 121: 3425-3441.
- Dessing MC, Tammaro A, Pulskens WP, Teske GJ, Butter LM, et al. (2015) The calcium-binding protein complex S100A8/ A9 has a crucial role in controlling macrophage-mediated renal repair following ischemia/reperfusion. Kidney Int 87: 85-94.
- Ebbing J, Seibert FS, Pagonas N, Frederic Bauer, Kurt Miller, et al. (2016) Dynamics of urinary calprotectin after renal is chaemia. PLoS One 11: e0146395.
- Ebbing J, Mathia S, Seibert FS, Pagonas N, Bauer F, et al. (2014) Urinary calprotectin: a new diagnostic marker in urothelial carcinoma of the bladder. World J Urol 32: 1485-1492.
- Chang CH, Yang CH, Yang HY, Chen TH, Lin CY, et al. (2015) Urinary biomarkers improve the diagnosis of intrinsic acute kidney injury in coronary care units. Medicine (Baltimore) 94: e1703.
- Bazzi C, Petrini C, Rizza V, Arrigo G, Napodano P, et al. (2002) Urinary N-acetyl-beta-glucosaminidase excretion is a marker of tubular cell dysfunction and a predictor of outcome in primary glomerulonephritis. Nephrol Dial Transplant 17: 1890-1896.
- Katagiri D, Doi K, Honda K, Negishi K, Fujita T, et al. (2012) Combination of two urinary biomarkers predicts acute kidney injury after adult cardiac surgery. Ann Thorac Surg 93: 577-583.
- Ramesh G, Krawczeski CD, Woo JG, Wang Y, Devarajan P (2010) Urinary netrin-1 is an early predictive biomarker of acute kidney injury after cardiac surgery. Clin J Am SocNephrol 5: 395-401.
- Fugmann T, Borgia B, Révész C, Godó M, Forsblom C, et al. (2011) Proteomic identification of vanin-1 as a ma¬rker of kidney damage in a rat model of type 1 diabetic ne¬phropathy. Kidney Int 80: 272-281.
- Jansen PA, Kamsteeg M, Rodijk-Olthuis D, Van Vlijmen-Willems IM, de Jongh GJ, et al. (2009) Expression of the vanin gene family in normal and inflamed human skin: induction by proinflammatory cytokines. J Invest Dermatol 129: 2167-2174.
- Hosohata K, Ando H, Fujimura A (2012) Urinary vanin-1 as a novel biomarker for early detection of drug-induced acute kidney injury. J Pharmacol Exp Ther 341: 656-662.
- Kobori H, Ohashi N, Katsurada A, Miyata K, Satou R, et al. (2008) Urinary angiotensinogen as a potential biomarker of severity of chronic kidney diseases. J Am Soc Hypertens 2: 349-354.
- Vaidya VS, Ferguson MA, Bonventre JV (2008) Biomarkers of acute kidney injury. Annu Rev PharmacolToxicol 48: 463-493.
- Xie Y, Sakatsume M, Nishi S, Narita I, Arakawa M, et al. (2001) Expression, roles, receptors, and regulation of osteopontin in the kidney. Kidney Int 60: 1645-657.
- Shu K-H, Wang C-H, Wu C-H, Huang T-M, Wu P-C, et al. (2016) Urinary π-glutathione S-transferase predicts advanced acute kidney injury following cardiovascular surgery. Sci Rep 6: 26335.
- Cheon JH, Kim SY, Son JY, Kang YR, An JH, et al. (2016) Pyruvate kinase M2: a novel biomarker for the early detection of acute kidney injury. Toxicol Res 32: 47-56.



