Research Article - (2025) Volume 8, Issue 1
Bacterial Reverse Mutation Test of 1-[[(3-hydroxy-1- adamantyl) Amino] Acetyl] Pyralidine-(s)-2-Carboxamide (vildagliptin amide impurity) using Salmonella Typhimurium and Escherichia Coli Tester Strains
Received Date: Jan 17, 2025 / Accepted Date: Feb 14, 2025 / Published Date: Feb 20, 2025
Copyright: ©Â©2025 T. Pavan Pradeep, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Pradeep, T. P., Nithyanandam, R. K., Padhy, D. (2025). Bacterial Reverse Mutation Test of 1-[[(3-hydroxy-1- adamantyl) Amino] Acetyl] Pyralidine-(s)-2-Carboxamide (vildagliptin amide impurity) using Salmonella Typhimurium and Escherichia Coli Tester Strains. Adv Bioeng Biomed Sci Res, 8(1), 01-14.
Abstract
The purpose of the Ames short-term bacterial reverse mutation experiment is to identify a variety of chemicals that can cause genetic harm that results in gene mutations. In this study, we investigated the mutagenic potential of 1-[[(3-hydroxy- 1-adamantyl) amino] acetyl] pyralidine-(S)-2-carboxamide (Vildagliptin amide impurity) using Salmonella typhimurium and Escherichia coli tester strains. The evaluation utilized plate incorporation methods to assess point mutations at the histidine locus in four strains of Salmonella and at the tryptophan locus in E. coli WP2uvrA, both with and without a metabolic activation system (S9). A series of concentrations (15.8 to 5000 μg/plate) were tested, revealing no significant decreases in revertant colony counts or cytotoxicity, as evidenced by the integrity of the bacterial lawn and lack of precipitation. Confirmatory assays at varying concentrations indicated that treatment did not induce notable increases in revertant counts across the tester strains, further supporting the absence of mutagenic activity. Positive controls demonstrated expected responses, confirming the reliability of the test conditions and the functionality of the metabolic activation system. Overall, the findings suggest that Vildagliptin amide impurity does not exhibit mutagenic properties under the tested conditions.
Keywords
AMES Assay, Bacterial Strains, Revertant colony counts, Preliminary Cytotoxicity, Mutagenic Assay, Confirmatory Assay
Introduction
The bacterial reverse mutation test, sometimes known as the Ames test because it was created by Bruce Ames in the early 1970s, is the most widely used assay to evaluate chemically induced gene mutation. A variety of Salmonella typhimurium and Escherichia coli bacterial strains are used in this short-term assay, which is especially intended to detect a wide range of deoxyribonucleic acid (DNA) reactive compounds that cause fixed gene mutation. In order to determine the mutagenic potential of novel chemical entities, active pharmaceutical components, or possible genotoxic contaminants in pharmaceutical products, as well as the potential for genotoxic carcinogenicity, the Ames test is widely used throughout the world as a hazard screen because there is a high predictive value for rodent carcinogenicity when a mutagenic response is obtainedIn order to screen for potential carcinogens and mutagens in mammals, the bacterial reverse mutation assay, which uses strains of bacteria that require amino acids, is frequently used as a first screening technique for identifying the mutagenic activity of chemicals. This assay's basic idea is that bacterial strains' mutations can be identified by their reversion. In the absence of the amino acid needed by the parent type strains, this results in the bacteria's functional ability to synthesis the crucial amino acid being restored, allowing them to proliferate. The majority of the compounds merely undergo metabolic change, which is mostly catalyzed in vivo by the liver's enzyme systems, and are neither mutagenic nor carcinogenic in and of themselves. As a result, the tests are conducted both directly and with the aid of an exogenous metabolic activation system. The most often used method is a co-factor supplemented post mitochondrial fraction (S9) that is taken from the livers of rats that have been exposed to an enzyme-inducing drug, such as β-Naphthoflavone and sodium phenobarbital. The his+/trp+ revertants are readily discernible as colonies against the limited background growth of his-/ trp- cells. (histidine auxotroph’s) and one strain of tryptophan dependent Escherichia coli (tryptophan auxotroph’s) obtained from Molecular Toxicology, Inc. 157, Industrial Park, Dr. Boone, NC 28607. Working stocks prepared from master vials (frozen permanents prepared from culture disc) maintained as frozen stocks in deep freezer (-70 ± 10°C), were used in the assay.
Materials and Methods
Characterization of the Bacterial Strains
Four tester strains of histidine dependent Salmonella typhimurium
The revertants were identified as colonies that grew in trace quantities of histidine & biotin (Salmonella typhimurium strains) and tryptophan (E. coli WP2uvrA strain). Frame shift and base pair substitution mutations are represented to identify mutagens of both types. Additional genetic markers (uvrB, rfa mutation, pKM101 plasmid) enhance the sensitivity of the strains to certain types of mutagens.
<img src="https://www.opastpublishers.com/scholarly-images/8875-68bec6cc5ad3b-bacterial-reverse-mutation-test-of-hydroxy-adamantyl-amino-a.png" width="670" height="500">
Preparation of Overnight Culture
Frozen stock cultures were inoculated in a flask containing 25 ml of Oxoid nutrient broth No. 2. Following inoculation, the flasks were placed in shaking water bath at 37 ± 2 °C shaking at 120 ± 10 rpm for 15:15 and 14:23 (hh:mm) in the preliminary cytotoxicity and mutagenicity assay respectively. Bacterial cultures removed from the shaker water bath at the end of the incubation period were held at 2 - 8°C (cold room) until use on the same day Bacterial Cell Viability
The bacterial cell viability
was determined by measuring the optical density of the nutrient broth containing cells at 670 nm using a spectrophotometer. The OD value of all the tester strains were in the acceptable range i.e., between 0.2 and 0.6 thus ensuring the cell density of at least 1 x 109 CFU/ml.
Details of Media
Culture Broth
Oxoid Nutrient Broth No. 2 (2.5 % w/v) was used to grow overnight cultures of the tester strains Minimal Glucose Agar Plates Vogel-Bonner minimal medium E supplemented with 1.5% w/v bacto agar and 1.0 % (w/v) glucose was used as bottom agar/
Minimal Glucose Agar (MGA).
Vogel-Bonner minimal medium E supplemented with 1.5% w/v bacto agar and 1.0 % (w/v) glucose was used as bottom agar/ Minimal Glucose Agar (MGA).
Top Agar for Selection of Revertants
Top (overlay) agar was prepared with 0.6 % (w/v) bacto agar and 0.6 % (w/v) sodium chloride and supplemented with 10 ml of 0.5 mM histidine/biotin (for Salmonella typhimurium strains) or 0.1 mM tryptophan solution (for Escherichia coli strains) per 100 ml of top agar for selection of histidine/tryptophan revertants.
Preparation of S9 Mix S9 mix
(cofactors and liver homogenate) was prepared freshly prior to use. In the preliminary cytotoxicity assay 2.5 mL of liver homogenate was added to 22.5 mL of cofactor mix to achieve 10 % v/v. In the Mutagenicity Assay, 6mL of liver homogenate was added to 54 mL of cofactor mix to achieve 10 % v/v. In the Confirmatory Assay, 14 mL of liver homogenate was added to 56ml of cofactor mix to achieve the concentration of 20 % v/v.
Method of formulation preparation
In the preliminary cytotoxicity assay, 322.5 mg of test item was dissolved in DMSO and made up to 5 mL and mixed with aid of vortex mixer which was equivalent to the concentration of 50 mg/ mL. From this, subsequent dilutions were made with DMSO to achieve concentrations of 15.8, 5.0, 1.58, 0.5 and 0.158 mg/ml and each dose spaced by dilution factor of 3.16. For mutagenicity and confirmatory assays, 774.0 mg of test item was dissolved in DMSO mixed with the aid of vortex mixer for complete dissolution and then final volume was made upto 10 ml using volumetric flask and additional 2 ml was added using micropipette to get the final volume of 12mL. This was equivalent to a concentration of 50 mg/mL. Subsequently, serial dilutions were made with spacing factor 2 using vehicle- DMSO from its immediate higher dose to achieve concentrations of 25, 12.5, 6.25, and 3.125 mg/mL. Formulations were mixed thoroughly with vortex mixer, during stock preparation, dilutions and treatment.
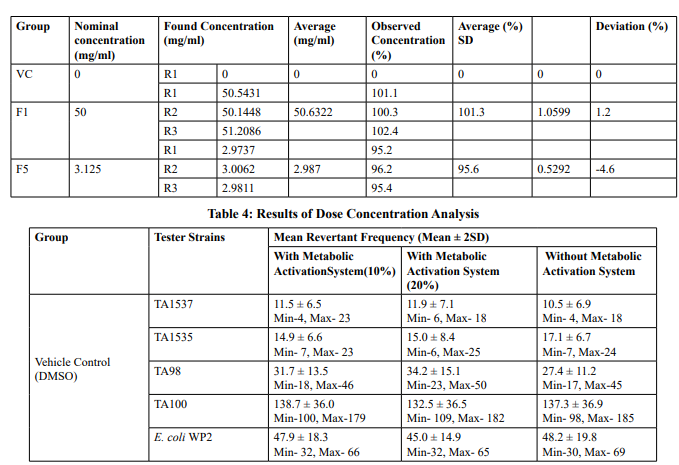
Dose concentration analysis
0.5 ml of samples each from 50 mg/ml, 3.125 mg/ml, and vehicle were subjected to dose formulation analysis. Formulations were analysed to determine the test item concentration in the selected concentrations as per Analytical method validation study no: VLL/0123/G/A002. The formulations were found to be stable up to 48 hours at room temperature as per Analytical method validation study no: VLL/0123/G/A002. Results of dose concentration analysis were tabulated in Table 4
Preparation of Dilute Sodium Hydroxide Solution
Weighed accurately and transferred 4.00155 g of Sodium hydroxide (NaOH) in 100 mL water, sonicated to dissolve and diluted to volume with water and mixed well.
Preparation of Buffer Solution
Weighed and transferred 5.52204 g of Sodium dihydrogen phosphate monohydrate into a 5 liters Mobile Phase containing 4000 mL water, dissolved and adjusted pH of the solution to 7.006 was diluted sodium hydroxide solution. Filtered the solution through 0.45 µm membrane filter and degassed.
Preparation of Mobile Phase-A
Mixed 1900 mL of Buffer and 100 mL of methanol in the ratio of 95:5 (v/v) respectively, sonicated to degas.
Preparation of Mobile Phase-B
Mixed 1400 mL of Buffer and 600 mL of Acetonitrile in the ratio of 70:30 (v/v) respectively, sonicated to degas.
Preparation of Diluent and Blank solution
Acetonitrile was used as Diluent and Blank solution. •
Preparation of Vildagliptin Amide Impurity (VPCD/ TC/22/570) Standard stock solution
A 13.44 mg of Vildagliptin Amide Impurity standard wasweighed and transferred into a 50 ml volumetric flask. Added diluent, sonicated to dissolve and made upto volume with diluent and mix well. Further 1.0 ml of Vildagliptin Amide Impurity standard was transferred a 10.0 ml volumetric flask and volume was made up to volume with diluent
Preparation of Sample Solution for VC - 0.0 mg/ml
Dose formulation solution 0.17 ml of was transferred into a 20 ml volumetric flask and volume was made up to volume with diluent.
Preparation of Sample Solution for F1-50.0 mg/ml
Dose formulation solution 0.10 ml was transferred into a 100.0 ml volumetric flask and volume was made up to volume with diluent. Further 2.6 ml was transferred into a 5.0 ml volumetric flask and volume was made up to volume with diluent. •
Preparation of Sample Solution for F5-3.125 mg/m
Dose formulation solution 0.17 ml of was transferred into a 20 ml volumetric flask and volume was made up to volume with diluent.
Preliminary cytotoxicity Assay
The preliminary cytotoxicity assay was performed by plate incorporation method, in tester strains TA98 and TA100 both in the presence (10%) and absence of metabolic activation system. The tester strains were exposed to the test item concentrations of 15.8, 50, 158, 500, 1580 and 5000 µg/plate both in the presence (10%) and absence of metabolic activation system. During the treatment, 2 ml of molten top agar (maintained at 47 °C) containing trace quantity of histidine and biotin (Salmonella strains), 500 µl of S9 mix for with-S9 or 0.1 mM sodium phosphate buffer for without-S9, 100 µl of respective tester strain along with either the vehicle or test item or positive control formulations were added in sterile tubes. The contents were mixed and overlaid onto the surface of minimal glucose agar plates. No Precipitation of test item was observed both in the presence and absence of metabolic activation system in both the strains. Following treatment, all treated plates were incubated at 37 ºC for 68:18 (hh:mm). Following incubation, all the plates were observed under microscope for background bacterial lawn inhibition and presence of precipitation on the plates. The bacterial background lawn was evaluated for the evidence of cytotoxicity. Also, cytotoxicity was scored relative to the vehicle control and recorded along with the mean revertant counts for each treatment. Revertant colonies were counted by automated colony counter.
Mutagenicity Assay
The mutagenicity assay was performed using the tester strains TA1537, TA1535, TA98, TA100 and E. coli WP2uvrA both in the presence (10% v/v S9) and absence of metabolic activation system. Based on the solubility, precipitation and preliminary cytotoxicity assay results, the mutagenicity assay was performed using test item concentrations of 5000, 2500, 1250, 625 and 312.5 µg/plate both in the presence (10% v/v) and absence of metabolic activation system. In mutagenicity assay, the tester strains were exposed to the test item via plate incorporation method, in which, 2 ml of molten top agar (maintained at 47°C) containing trace quantity of histidine and biotin (for Salmonella typhimurium strains)/ tryptophan (for Escherichia coli strains), 500 µl of S9 mix (10% S9) for with S9 or 0.1 mM sodium phosphate buffer for without S9, 100 µl of respective tester strain along with either the vehicle or test item or positive control formulations were added in sterile tubes. The contents were mixed and overlaid onto the surface of minimal glucose agar plates. No Precipitation of test item was observed from both in the presence and absence of metabolic activation system in all five tester strains.
Confirmatory Assay
Confirmatory assay was performed using test item concentrations of 5000, 2500, 1250, 625 and 312.5 µg/plate in the presence (20% v/v) of metabolic activation system. Tester strains were exposed to the test item via plate incorporation method, in which, 2 ml of molten top agar (maintained at 47°C) with trace quantity of histidine and biotin (for Salmonella typhimurium strains)/ tryptophan (for Escherichia coli strains), 500 µl of S9 mix (10% S9), 100 µl each of the tester strain and either the vehicle or test item or positive control formulations were added in sterile tubes, the contents were mixed and overlaid onto the surface of minimal glucose agar plates. No Precipitation of test item was observed at 312.5 to 5000 µg/plate concentrations. All the mutagenicity and confirmatory assay treated plates were incubated at 37 ºC for 64:12 (hh:mm). Following incubation, all the plates were observed under microscope for background lawn inhibition, presence of precipitation on the plates and were recorded along with the individual revertant counts for each test concentration. Revertant colonies were counted by automated colony counter.
Assay Evaluation Criteria
No formal hypothesis testing was performed to analyse the data. The responses observed in the assay were evaluated as per the criteria mentioned below. For the test item to be considered positive, the test item must produce at least a 2- fold increase in the mean revertants per plate of at least one of these tester strains; TA98, TA100 and E. coli WP2uvrA over the mean revertants per plate of respective vehicle and; at least a 3-fold increase in the mean revertants per plate in one or both of the tester strains TA1535 and TA1537 in comparison with appropriate vehicle control. This increase in the mean number of revertants colony units per plate can be accompanied by a dose response (in at least 2-3 consecutive concentrations) to increasing concentrations of the test item.
Results and Discussion
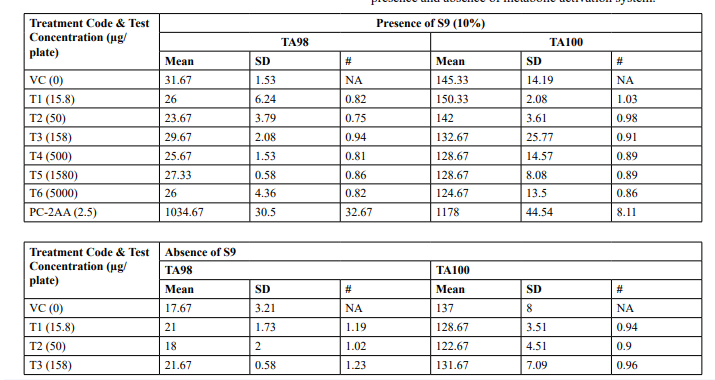
Preliminary Cytotoxicity Assay (Refer Table 1; Appendix 1, 4 & Figure 1,2)
Cytotoxicity was measured in terms of thinning of background bacterial lawn and reduction of revertant colony counts when compared to vehicle control. Test item treated at 15.8, 50, 158, 500, 1580 and 5000 µg/plate concentrations did not elicit considerable reduction in revertant colony counts both in the presence (10%) and absence of metabolic activation system in both tester strains TA98 and TA100 in comparison to respective vehicle control. Based on the preliminary cytotoxicity assay results, 5000 µg/ plate was selected as highest concentration for mutagenicity assay both in the presence (10%) and absence of metabolic activation system and confirmatory assay with (20%) metabolic activation. Subsequent lower concentrations selected were 2500, 1250, 625 and 312.5 µg /plate. No precipitation was observed at 15.8 µg/ plate to 5000 µg/plate in both TA98 and TA100 strains in the presence and absence of metabolic activation system
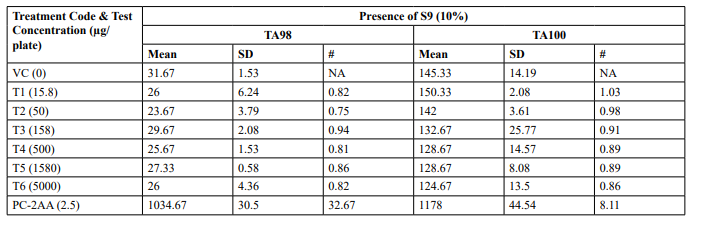
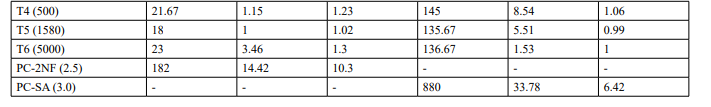
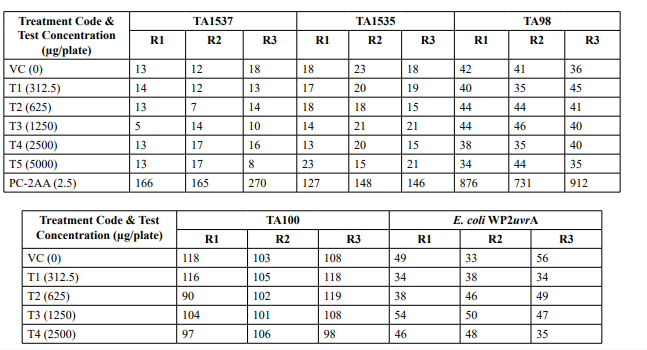
Table 1: Preliminary Cytotoxicity Assay - Mean Revertant Frequency
Appendix 1: Preliminary Cytotoxicity Assay - Precipitation and Background Lawn Evaluation
Appendix 4: Preliminary Cytotoxicity Assay - Individual Revertant Colony Counts
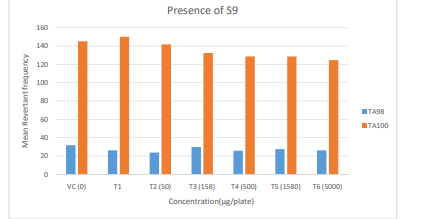
Figure 1: Diagrammatic representation of Preliminary Cytotoxicity Assay - Mean Revertant Frequency (Presence of S9)
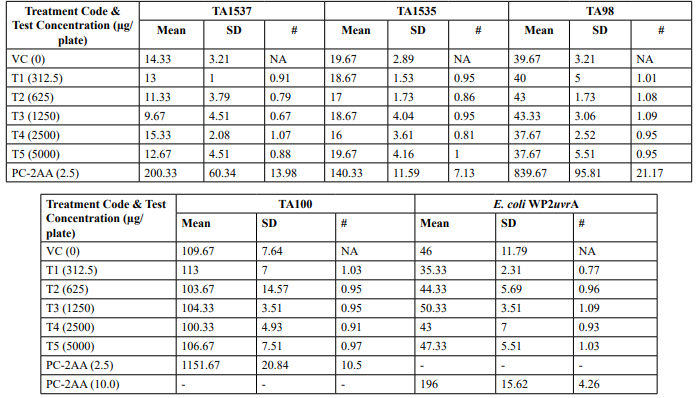
Table 2: Mutagenicity Assay - Mean Revertant Frequency Metabolic Activation: Presence of S9 (10%)
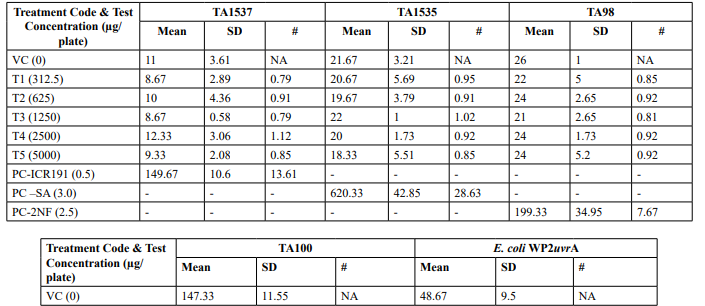
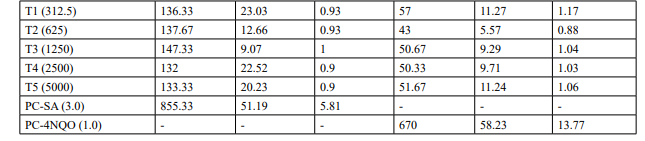
Table 2 (Continued): Mutagenicity Assay Mean Revertant Frequency Metabolic Activation: Absence of S9
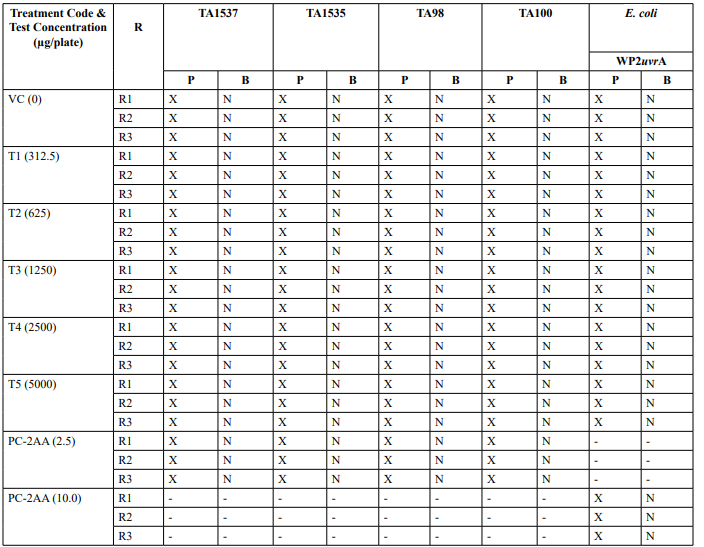
Appendix 2: Mutagenicity Assay - Precipitation and Background Lawn Evaluation Metabolic Activation: Presence of S9 (10%)
Appendix 2 continued: Mutagenicity Assay - Precipitation and Background Lawn Evaluation Metabolic Activation: Absence of S9
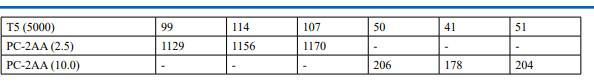
Appendix 5: Mutagenicity Assay - Individual Revertant Colony Counts Metabolic Activation: Presence of S9 (10%)
Appendix 5: Continued: Mutagenicity Assay - Individual Revertant Colony Counts Metabolic Activation: Absence of S9
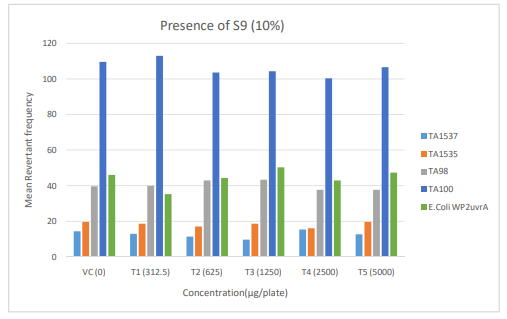
Figure 3: Diagrammatic representation of Mutagenicity Assay - Mean Revertant Frequency Metabolic Activation: Presence of S9 (10%)
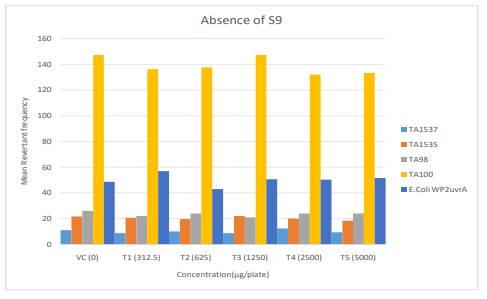
Figure 4: Diagrammatic representation of Mutagenicity Assay Mean Revertant Frequency Metabolic Activation: Absence of S9
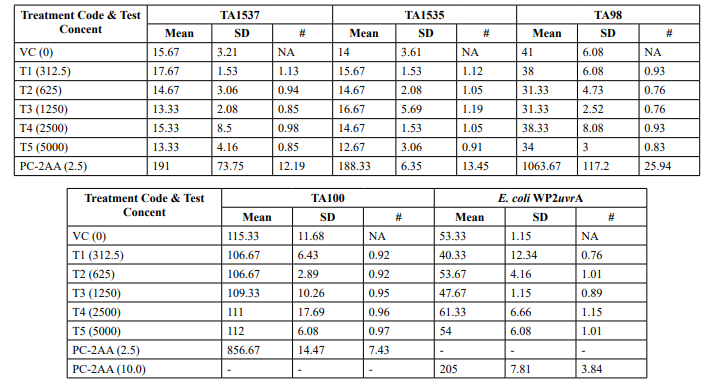
Table 3: Confirmatory Assay - Mean Revertant Frequency Metabolic Activation: Presence of S9 (20%)
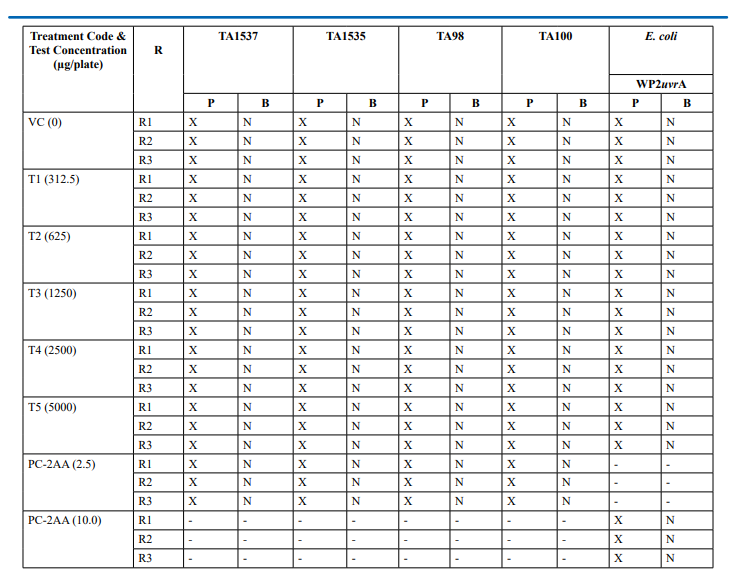
Appendix 3: Confirmatory Assay - Precipitation and Background Lawn Evaluation Metabolic Activation: Presence of S9 (20%)
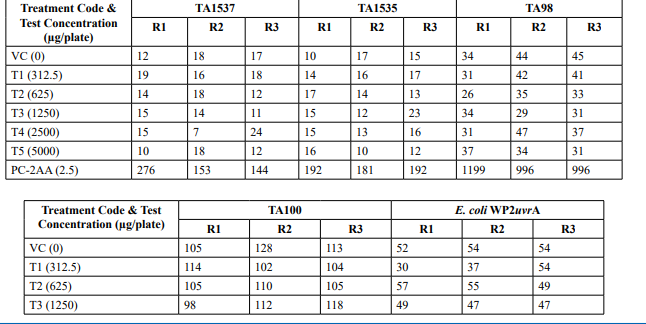
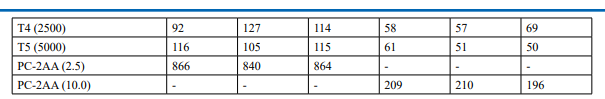
Appendix 6: Confirmatory Assay - Individual Revertant Colony Counts Metabolic Activation: Presence of S9 (20%)
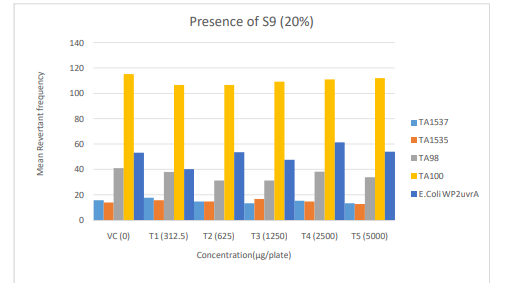
Figure 5: Diagrammatic representation of Confirmatory Assay - Mean Revertant Frequency Metabolic Activation: Presence of S9 (20%)
Mutagenicity Assay (Refer Table 2; Appendix 2, 5 & Figure
The mean concentration of the test item found in the highest and lowest tested dose formulations prepared for treatment were 50.6322 mg/mL and 2.9870 mg/mL against the nominal concentrations of 50 mg/mL and 3.125 mg/mL respectively. The results fell within 100 ± 10 % of respective nominal concentrations, thereby meeting the acceptance criteria. No precipitation of test item was observed on plates tested at 312.5 to 5000 µg/plate in all five tester strains Test item treated at concentrations from 312.5 to 5000 µg/plate did not elicit any considerable increase in the mean revertant colony counts both in the presence (10%) and absence of metabolic activation system in TA1537, TA1535, TA98, TA100 and E. coli WP2uvrA tester strains in comparison to the vehicle control. No toxicity in the form of thinning of background bacterial lawn was observed in TA1537, TA1535, TA98, TA100 and E. coli WP2uvrA treated at 312.5 to 5000 µg/plate in presence (10%) and absence of metabolic activation system.
Confirmatory Assay (Refer Table 3; Appendix 3, 6 & Figure 5)
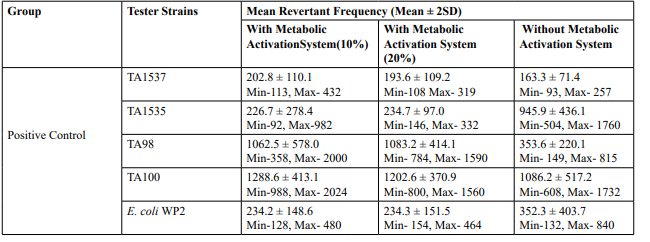
No precipitation of test item was observed on plates tested at 312.5 to 5000 µg/plate in all five tester strains. Test item treated at concentrations from 312.5 to 5000 µg/plate, in all five tester strains did not elicit any considerable increase in the mean revertant colony counts in the presence (20%) of metabolic activation system in comparison to the vehicle control. No toxicity in the form of thinning of background bacterial lawn was observed in TA1537, TA1535, TA98, TA100 and E. coli WP2uvrA treated at312.5 to 5000 µg/plate in presence (20%) of metabolic activation system. The mean spontaneous revertant colony count frequency of vehicle controls fell within the range of the respective in-house historical control data. The positive control treated plates exhibited multi-fold increase (>3 fold) in the mean revertant colonies (his+/ trp+) respective to their strains indicating the sensitivity of the test system towards their specific mutagens and confirmed that the test conditions adopted were appropriate; and that the metabolic activation system functioned properly
Conclusion
Based on the results, it is concluded that 1-[[(3-hydroxy-1- adamantyl) amino] acetyl] pyralidine- (S)-2-carboxamide (Vildagliptin amide impurity), tested up to 5000 µg/plate under the tested conditions, is non-mutagenic in Bacterial Reverse Mutation Test.
References
1. Ames, B. N., McCann, J., & Yamasaki, E. (1975). Methods for detecting carcinogens and mutagens with the Salmonella/mammalian-microsome mutagenicity test. Mutat. Res.;(Netherlands), 31.
2. Teasdale, A. (2017). ICH M7: Assessment and control of DNA reactive (mutagenic) impurities in pharmaceuticals to limit potential carcinogenic risk. ICH Quality Guidelines: An Implementation Guide, 667-699.
3. Guideline, I. C. H. (2006). Q3A (R2), Impurities in New Drug Substances. ICH Steering Committee.
4. Mortelmans, K., & Zeiger, E. (2000). The Ames Salmonella/ microsome mutagenicity assay.Mutation research/fundamental and molecular mechanisms of mutagenesis, 455(1-2), 29-60.
5. Maron, D. M., & Ames, B. N. (1983). Revised methods for the Salmonella mutagenicity test. Mutation Research/ Environmental Mutagenesis and Related Subjects, 113(3-4), 173-215.
6. McCann, J., & Ames, B. N. (1976). Detection of carcinogens as mutagens in the Salmonella/microsome test: assay of 300 chemicals: discussion. Proceedings of the National Academy of Sciences, 73(3), 950-954.
7. No, O. T. (1997). 471: Bacterial reverse mutation test. OECD guidelines for the testing of chemicals, section, 4, 1-11.
8. Thomas, D. N., Wills, J. W., Tracey, H., Baldwin, S. J., Burman, M., Williams, A. N., ... & Lynch, A. M. (2024). Ames test study designs for nitrosamine mutagenicity testing: qualitative and quantitative analysis of key assay parameters. Mutagenesis, 39(2), 78-95.


