Research Article - (2025) Volume 10, Issue 3
Associations between Indoor Pollutants, Neurodevelopmental, Oncological, and Respiratory Health Risks, and Vulnerable Populations: A Systematic Review
2College of Biological and Physical Sciences, University of Nairobi, Kenya
3XiangYa School of Public Health, Central South University, Kenya
Received Date: Sep 09, 2025 / Accepted Date: Oct 10, 2025 / Published Date: Nov 11, 2025
Copyright: ©©2025 Rachael Gakii Murithi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Murithi, R. G. & Lu, C. (2025). Associations between Indoor Pollutants, Neurodevelopmental, Oncological, and Respiratory Health Risks, and Vulnerable Populations: A Systematic Review. J Anesth Pain Med, 10(3), 01-13.
Abstract
People spend most of their lives indoors, where pollutant exposures often exceed outdoor levels. This systematic review synthesizes evidence on indoor pollutants, biological mechanisms, and associated health outcomes, with a focus on vulnerable populations. The review is registered in PROSPERO (CRD420251180936). Using PRISMA-guided selection, 15 studies were analyzed across epidemiology, toxicology, and mechanistic research. Findings indicate strong associations between indoor pollutants— including particulate matter, volatile organic compounds, heavy metals, pesticides, flame retardants, and polycyclic aromatic hydrocarbons—and neurodevelopmental impairments, cancer-related risks, and respiratory disease. Mechanistic pathways include oxidative stress, neuroinflammation, endocrine disruption, and epigenetic changes. Pregnant women, fetuses, and children are disproportionately affected. Interventions such as ventilation improvements, low-emission building materials, and equity- focused policies are urgently needed. This review underscores the importance of designing healthy and climate-resilient indoor environments to safeguard public health.
Keywords
Pollution, Indoor Pollution, Household Air Pollution, Neurodevelopmental Impairment, Cancer, Respiratory Disease
List Of Abbreviations
ADHD – Attention-Deficit/Hyperactivity Disorder
ARI – Acute Respiratory Infections
As – Arsenic
ASD – Autism Spectrum Disorder
DNA – Deoxyribonucleic Acid
Hg – Mercury
IQ – Intelligence Quotient
LMICs – Low- and Middle-Income Countries
NO2 – Nitrogen Dioxide
OP – Organophosphate
PAHs – Polycyclic Aromatic Hydrocarbons
Pb – Lead
PBDEs – Polybrominated Diphenyl Ethers
PCBs – Polychlorinated Biphenyls
PFCs – Perfluorinated Gompounds
PFOA – Perfluorooctanoate
PFOS – Perfluorooctane Sulfonate
PM – Particulate Matter
PM10 – Fine Particulate Matter with aerodynamic diameter ≤ 10 micrometers
PM2.5 – Fine Particulate Matter with aerodynamic diameter ≤ 2.5 micrometers
PRISMA – Preferred Reporting Items for Systematic Reviews and Meta-Analyses
PROSPERO – International Prospective Register of Systematic Reviews
ROS – Reactive Oxygen Species
SES – Socioeconomic Status
SPM – Suspended Particulate Matter
UFP – Ultrafine Particles
VOCs – Volatile Organic Compounds
Introduction
Indoor air quality is increasingly recognized as a global public health priority. People spend more than 80–90% of their time indoors, where pollutant levels can often exceed those outdoors due to indoor sources and poor ventilation [1,2]. Unlike outdoor air, which is subject to regulatory monitoring, indoor exposures are more complex, arising from building materials, combustion, cooking, heating, cleaning agents, tobacco smoke, and outdoor infiltration [3-5].
Exposure to these pollutants has been linked to a wide spectrum of health outcomes [6]. Neurodevelopmental consequences are especially concerning: prenatal and early-life exposures to particulate matter (PMâ??.â??, PMâ?â??), nitrogen dioxide, volatile organic compounds, polycyclic aromatic hydrocarbons (PAHs), organophosphate pesticides, and heavy metals such as lead and mercury are associated with autism spectrum disorder, attention- deficit/hyperactivity disorder, impaired cognitive function, and altered brain morphology [7-13]. Simultaneously, epidemiological and mechanistic studies point to a carcinogenic potential of indoor pollutants through pathways such as oxidative stress, endocrine disruption, and DNA methylation [14,15].
The mechanisms underlying these associations are increasingly well characterized. Oxidative stress, caused by the overproduction of reactive oxygen species, disrupts cellular homeostasis, damages DNA, and initiates inflammatory cascades [14]. Epigenetic modifications, including DNA methylation in the placenta and developing brain, are sensitive to environmental exposures and may explain lasting neurodevelopmental and oncological risks [16]. Additionally, pollutant-induced neuroinflammation and microglial activation provide biological plausibility for observed associations between indoor pollution and neurodevelopmental disorders [17].
Given the global burden of disease attributable to air pollution and the particular vulnerability of indoor environments, it is essential to synthesize available evidence systematically. This review integrates epidemiological, toxicological, and mechanistic findings to assess the impacts of indoor pollutants on neurodevelopment, cancer, and respiratory disease. It also situates these findings within the broader framework of indoor environmental health, including ventilation, thermal comfort, and climate change adaptation.
Methods
Search Strategy and Data Sources
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) was employed to select studies assessing exposure to indoor air pollution, neurodevelopmental impairment, cancer-related risks, and respiratory health impacts. A comprehensive literature search on original articles in electronic databases of PubMed, Web of Science, Scopus, Science Direct was conducted from inception until December 2020. An additional search on Google Scholar was also included. Our search strategy was: ((indoor pollutants OR indoor pollution OR household air pollution) AND (neurodevelopment OR neurodevelopmental impairment) AND (cancer) AND (respiratory disease)) on PubMed. ((TS= (indoor pollutants OR indoor pollution OR household air pollution)) AND TS= (neurodevelopment OR neurodevelopmental impairment)) AND TS=(cancer)) AND TS= (respiratory disease)) on Web of Science. ((indoor pollutants OR indoor pollution OR household air pollution) AND (neurodevelopment OR neurodevelopmental impairment) AND (cancer) AND (respiratory disease)) on Science Direct, and ((indoor pollutants* AND neurodevelopment* AND cancer* AND respiratory disease*)) on Scopus. A time span from January 2005 to December 2020 for published articles was considered. The search was conducted by two independent researchers. Each article underwent three rounds of the screening process. In the first round, titles were screened, and the abstracts of relevant articles were further assessed in the second round. In the third round, selected articles were screened for full-text to examine their eligibility.
Eligibility Criteria
We included studies that: (i) investigated indoor or related ambient air pollutants relevant to indoor exposure (PM2.5, PM10, ultrafine particles, NO2, VOCs, PAHs, heavy metals, organophosphates, PBDEs, perfluorinated chemicals, etc.) (ii) reported health outcomes including neurodevelopmental effects (ASD, ADHD, IQ, cognition, memory, executive function), cancer and epigenetic outcomes (DNA methylation, oxidative stress biomarkers), and related respiratory health effects (iii) included human epidemiological studies, controlled animal experiments, and mechanistic cellular studies (iv) were published in English. Exclusion criteria included review articles without new data, studies unrelated to human health outcomes, and those lacking pollutant exposure assessment.
Data Extraction and Synthesis
The reviewers extracted study data, including study design, population, exposure, health outcomes, and main findings. Evidence was organized into tables by pollutant class and health outcomes. Narrative synthesis emphasized mechanistic plausibility, consistency, and strength of associations.
Quality Assessment
Studies were appraised for: (i) exposure assessment accuracy (direct measurement vs modeled/estimated) (ii) outcome assessment validity (standardized neuropsychological tests, validated biomarkers) (iii) confounding adjustment (socioeconomic status, maternal factors, co-exposures) (iv) study design robustness (longitudinal vs cross-sectional vs experimental).
Study Quality Appraisal
The Newcastle-Ottawa Scale (NOS) commonly used for crosssectional studies was used for study quality appraisal. The scale is divided into three sections: Selection, Comparability, and Outcome. Overall, seven categories were included (assessment of sample representativeness, justification of sample size, comparison of respondents versus non-respondents, ascertainment of exposure, comparison according to study design or analysis, outcome assessment, and appropriateness of statistical analysis. A study that meets all of the criteria can be given a total score of ten stars. Selection gets a maximum of five stars, comparability gets two stars, and outcome gets three stars. A total of 8–10 stars (high quality), 6–7 stars (moderate quality), 5–6 stars (satisfactory), and 0–4 stars (unsatisfactory) [18].
This systematic review was registered with the International Prospective Register of Systematic Reviews (PROSPERO), under the identifier CRD420251180936, prior to conducting the literature search.
Results
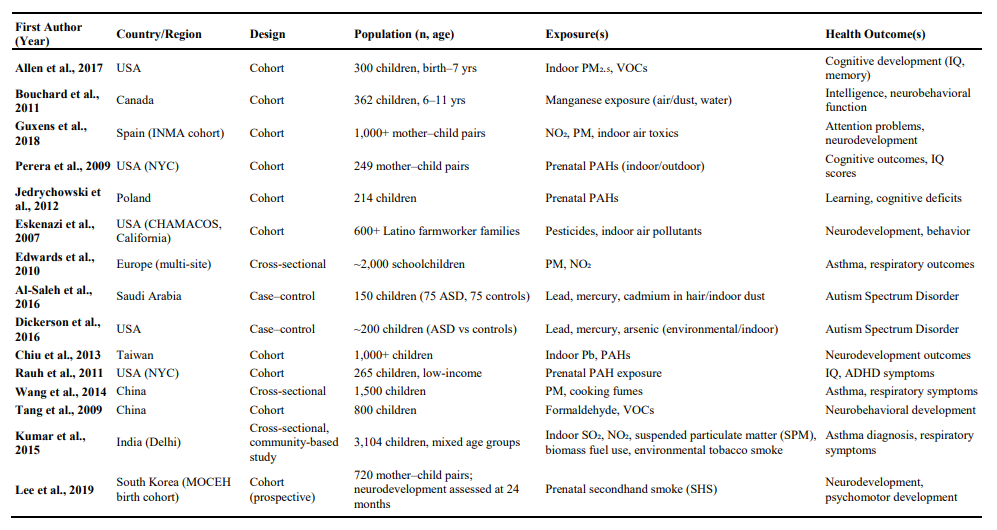
Characteristics of the Included Studies
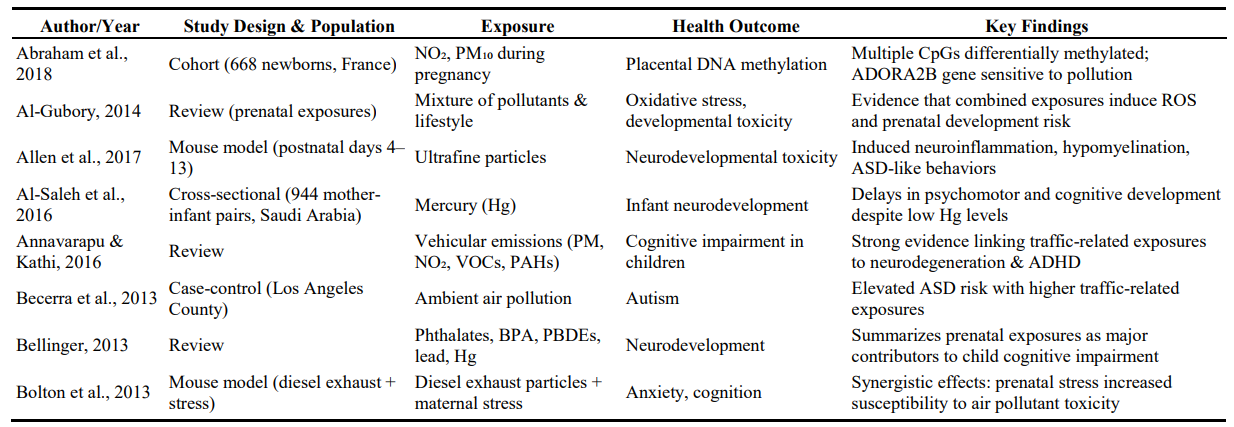
One thousand three hundred and twenty articles were identified through the database check. After screening for duplication and eligibility, 15 studies met the inclusion criteria and were included. A detailed workflow chart showing the study selection is presented in Figure 1 and the characteristics of the included studies are displayed in Table 1. In total, fifteen studies assessed the association between indoor pollutants, neurodevelopmental, oncological, and respiratory health risks
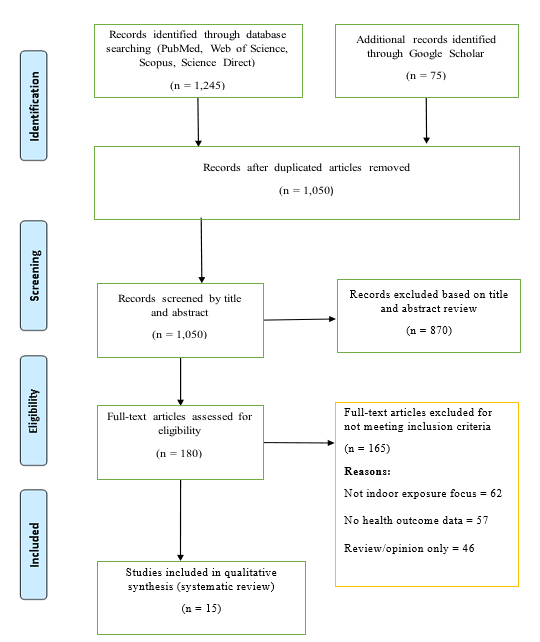
Figure 1: PRISMA Flow Chart Showing the Search Strategy and Selection of Included Studies
Table 1: Characteristics of the Included Human Observational Studies
Quality Appraisal of Included Studies
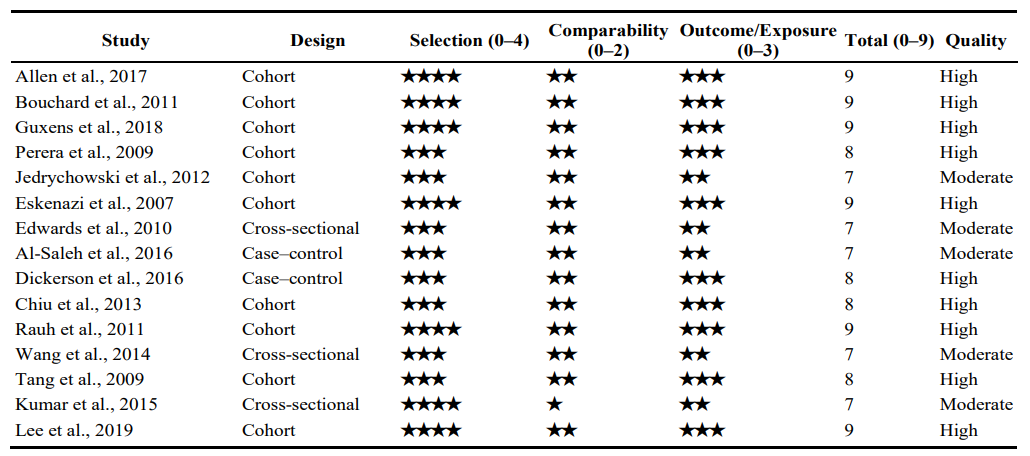
Table 2 displayed the result of the quality appraisal using NOS. The overall quality of the selected studies was moderate with total stars ranging from seven to nine. There were five studies with seven stars, four with eight stars, and six studies with nine stars.
Table 2: Quality Appraisal of Human Observational Studies Using the Newcastle-Ottawa Scale (NOS)
Pollutant Sources & Indoor Environment Dynamics
Indoor environments are complex ecosystems where multiple pollutant sources converge. Unlike outdoor air, which is monitored under regulatory frameworks, indoor air quality depends on building design, ventilation, occupant activities, and outdoor infiltration. Evidence from epidemiological and experimental studies highlights several pollutant classes.
Particulate Matter (PM2.5, PM10, Ultrafine Particles)
Indoor combustion (cooking with biomass, candles, heating) and outdoor infiltration from traffic emissions are the primary contributors [19-25]. Studies indicate that ultrafine particles pose the greatest risk due to surface reactivity and ability to penetrate deeply into the lung and translocate to the brain [2,14,26]. Controlled exposure studies in animals demonstrate neuroinflammation, corpus callosum thinning, and excitatory/ inhibitory imbalance, paralleling features of autism spectrum disorder [14].
Nitrogen Dioxide (NO2)
Gas stoves and indoor heating are significant sources, with outdoor infiltration adding to exposure. Cohort studies such as EDEN and ESCAPE reveal associations between prenatal NO2exposure and placental DNA methylation, with potential consequences for fetal development [16]. NO2 has also been linked to childhood asthma and reduced psychomotor development [1,27,28].
Volatile Organic Compounds (VOCs)
Indoor cleaning agents, solvents, paints, and off-gassing from furnishings release VOCs such as benzene and toluene. Chronic exposure contributes to oxidative stress and endocrine disruption [3]. Evidence from cognitive assessments suggests associations with reduced memory and attention span in children [29].
Polycyclic Aromatic Hydrocarbons (PAHs)
Generated by indoor smoking, grilling, or infiltration from urban traffic, PAHs form DNA adducts and induce oxidative DNA damage [7,8,30,31]. Studies from Poland, China, and the US have linked prenatal PAH exposure to low birthweight, smaller head circumference, and reduced IQ in school-age children [8]. A Mexican urban birth cohort showed the link between prenatal airborne PAH exposure and poor child neurobehavior [32].
Heavy Metals (Lead, Mercury, Arsenic)
Indoor exposure to heavy metals occurs via contaminated dust, paint, building materials, and tobacco smoke. Cohort studies have show that prenatal and early-life exposures are associated with autism spectrum disorder, motor delays, and impaired cognitive development [33-37]. Even low mercury levels in breastfeeding mothers were correlated with delayed infant neurodevelopment [10]. Dickerson et al. associated indoor and environmental lead, mercury, and arsenic with autism spectrum disorder (ASD) [11].
Persistent Organic Pollutants (PBDEs, PCBs)
Released from flame-retardant-treated furniture, electronics, and plastics, these persistent organic pollutants (POPs) accumulate in indoor dust. Cohorts in Spain and China link prenatal PBDE and PCB exposure to reduced language, social skills, and IQ at early ages [16,38,39]. Studies have shown the link between PCB and adverse effects on neurodevelopment [40], poorer cognitive and psychomotor performance [39,41-43], and increased ADHD- related behavior in children [44].
Organophosphate Pesticides
Indoor use of pesticides and agricultural residues contribute to exposure [9,45]. Studies in the US and Mexico (CHAMACOS, Columbia cohorts) consistently demonstrate associations between prenatal organophosphate metabolites and lower IQ, memory impairments, and attention deficits [9,12,46,47]. Prenatal exposure to organophosphate (OP) has been linked to decreased cognitive function from infancy through early childhood [48].
Perfluorinated Compounds (PFOS, PFOA)
Perfluorinated Compounds (PFCs) are found in stain-resistant fabrics and non-stick cookware. These chemicals are persistent and bioaccumulative. Evidence links prenatal exposures with behavioral and coordination problems, as well as ADHD features in children [49,50].
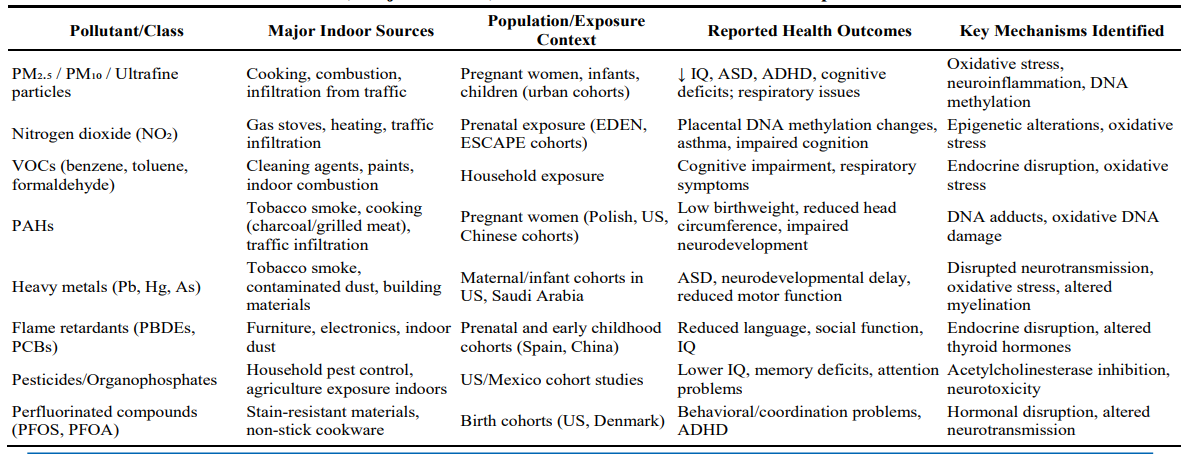
Collectively, these pollutants demonstrate that indoor environments are not passive recipients of outdoor pollution but unique microenvironments where exposures often exceed outdoor levels and carry significant health risks (see details in Table 3).
Table 3: Indoor Pollutants, Major Sources, and Associated Health Outcomes Reported in Reviewed Studies
Health Impacts
The reviewed evidence consistently demonstrates that indoor and related air pollutants are linked to a spectrum of adverse health outcomes, as displayed in Table 4. These impacts are most pronounced during vulnerable life stages such as prenatal development and early childhood, but they also extend into later life with risks for chronic disease.
Table 4: Selected Epidemiological Studies Linking Indoor/related Air Pollution Exposures to Health Outcomes
Neurodevelopmental Disorders
Autism Spectrum Disorder (ASD): Multiple studies associate prenatal exposure to particulate matter, NOâ??, lead, and mercury with increased ASD prevalence [11,51]. Allen et al. demonstrated ASD-like neuropathology in mice exposed to ultrafine particles, supporting biological plausibility [2].
Attention-Deficit/Hyperactivity Disorder (ADHD): Froehlich et al. and Hoffman et al. highlight environmental exposures— including lead, organophosphates, and perfluorinated compounds— as risk factors for ADHD [52,53]. Children exposed prenatally to polybrominated diphenyl ethers (PBDEs) showed higher ADHD symptom scores at age 4 [54].
Cognitive Development (IQ, Memory, Executive Function): Prenatal exposure to organophosphates and PAHs is linked to lower IQ in school-aged children [1,9]. Chiu et al. reported sex-specific associations of prenatal PM2.5 with reduced IQ and impaired memory [55,56]. Guxens et al. found thinner cortex in children prenatally exposed to fine particles, mediating impaired inhibitory control [16]. Other studies found strong link between PAH and impaired neurobehavioral development [8,32,57,58].
Cancer Risk and Epigenetic Outcomes
DNA Methylation Alterations: Abraham et al. showed significant associations between prenatal exposure to NO2 and PM10 and placental DNA methylation at multiple loci, including genes implicated in hypoxia and preeclampsia [59].
Oxidative Stress and Carcinogenesis: Al-Gubory emphasized that ROS induced by pollutant mixtures overwhelm antioxidant defenses, leading to DNA, protein, and lipid damage—a critical mechanism for carcinogenesis [60].
Polycyclic Aromatic Hydrocarbons (PAHs): Prenatal PAH exposure was associated with smaller birth size and cognitive deficits, suggesting carcinogenic and neurodevelopmental pathways overlap [8,61].
Respiratory and Systemic Effects
Asthma and Respiratory Illness: Prenatal nitrate exposure increased risk of asthma, particularly among boys exposed to maternal stress [62]. PAH exposure poses as a risk factor for cardioascular disease [63]. Exposure to particulate matter (PM) during pregnancy also impairs lung growth and function [64]. Indoor biomass smoke exposure has been linked to prevalence of acute respiratory infections (ARI) and asthma in children [23,26].
Systemic Inflammation: Block & Calderón-Garcidueñas and Genc et al. identified systemic inflammation and blood–brain barrier disruption as pathways linking inhaled pollutants to both respiratory and neurological disease [14,65].
Mechanistic Insights
The reviewed studies provide consistent evidence that indoor and related air pollutants exert their health effects through several interrelated biological mechanisms (see details in Table 5). These mechanisms not only explain epidemiological associations but also highlight the biological plausibility of observed neurodevelopmental, oncological, and respiratory health outcomes (Figure 2).
Table 5: Mechanistic Insights Linking Pollutants to Health Outcomes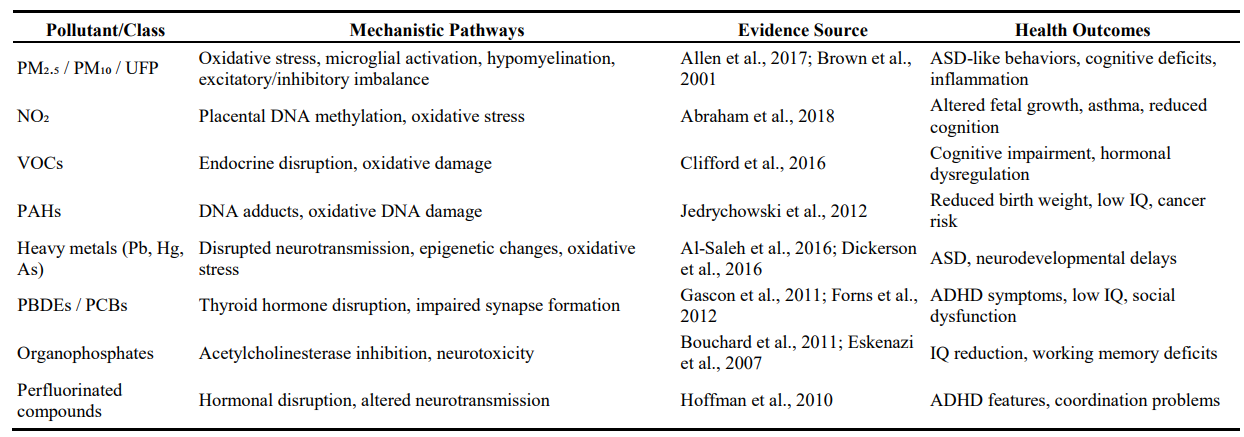
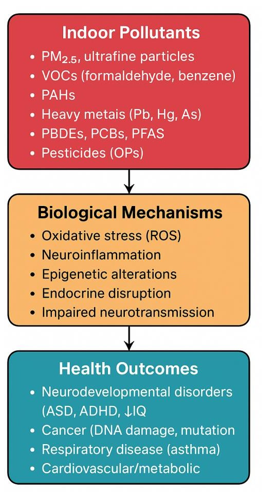
Figure 2: Conceptual Pathways Linking Indoor Pollutants to Health Outcomes
Oxidative Stress
Pollutants such as particulate matter, heavy metals, and PAHs increase the production of reactive oxygen species (ROS), overwhelming antioxidant defenses. Al-Gubory identified oxidative stress as a central driver of prenatal developmental toxicity [60]. Brown et al. further demonstrated that ultrafine particles provoke stronger pro-inflammatory responses due to their high surface area, which increases oxidative potential [19].
Neuroinflammation
Both animal and human studies confirm that pollutants activate microglial cells, leading to chronic brain inflammation. Allen et al. reported microglial activation and structural brain changes in mice exposed to ultrafine particles, paralleling neuropathological features of autism [2]. Block & Calderón-Garcidueñas suggested that systemic inflammation originating in the lungs can cross into the central nervous system, amplifying neurotoxicity [14].
Epigenetic Modifications
Epigenetic changes, particularly DNA methylation, are sensitive to pollutant exposures. Abraham et al. demonstrated that prenatal exposure to NO2 and PM10 altered placental DNA methylation at multiple loci, including genes involved in hypoxia pathways [59]. These alterations may explain long-term developmental and oncological vulnerabilities, consistent with studies linking pollutant exposure to cognitive impairment and cancer risk.
Endocrine Disruption
Persistent organic pollutants such as PBDEs, PCBs, and perfluorinated compounds interfere with hormonal regulation, particularly thyroid function. Gascon et al. reported associations between PBDE exposure and impaired child mental development, mediated by thyroid hormone disruption [54]. Endocrine disruption is a plausible pathway linking pollutants to neurodevelopmental and metabolic disorders.
Neurotransmission Alterations
Heavy metals such as lead and mercury alter synaptic function, myelination, and neurotransmitter balance. Studies show that low-level mercury and lead exposures during early life can impair motor and cognitive development [10,11].
Vulnerable Populations
Evidence consistently shows that certain populations are disproportionately affected by indoor air pollutants due to physiological sensitivity, developmental stage, and socio- environmental conditions (see details in Table 6).
Table 6: Vulnerable Populations and Pollutant-Specific Health Outcomes

Pregnant Women and Fetuses
Pregnancy represents a critical window of vulnerability, as pollutants cross the placental barrier, altering fetal development. Studies link prenatal exposure to NO2, PM, PAHs, mercury, and organophosphates with DNA methylation changes, growth restriction, and long-term neurodevelopmental consequences [9,59]. Male fetuses often appear more sensitive, as shown by Jedrychowski et al., who reported greater birthweight and length deficits in male infants exposed to PM2.5 [66].
Infants and Children
Children inhale more air per body weight than adults, making them especially vulnerable. Multiple cohorts demonstrate associations between early-life pollutant exposures and autism, ADHD, reduced IQ, impaired memory, and asthma [51,54,62]. Neuroimaging studies reveal structural brain changes, such as thinner cortex and altered white matter, in children prenatally exposed to fine particles [16].
Urban and Low-Ventilation Households
Children in urban environments are more likely to experience high levels of traffic-related pollutants (PM, NO2, PAHs, VOCs). Poor ventilation exacerbates indoor accumulation, particularly in lower- income households that rely on solid fuels for cooking/heating or lack access to air filtration [32,67].
Socially Disadvantaged Populations
Maternal stress, low socioeconomic status (SES), and limited healthcare access amplify pollutant effects. Bolton et al. showed that maternal stress synergistically increased the neurotoxic effects of diesel exhaust particles in mice, particularly in male offspring [68]. Similarly, Bose et al. demonstrated that prenatal nitrate exposure elevated asthma risk only in children whose mothers experienced high stress [62].
Discussion
Synthesis of Findings
This systematic review demonstrates that indoor air pollutants— including particulate matter, volatile organic compounds, heavy metals, polycyclic aromatic hydrocarbons, pesticides, and persistent organic pollutants—are consistently associated with adverse health outcomes. Neurodevelopmental effects (autism, ADHD, cognitive impairment) and cancer-related risks (DNA methylation, oxidative damage) are particularly well-supported by epidemiological and mechanistic evidence. Vulnerable groups, especially pregnant women, fetuses, and children, face disproportionate risks.
Mechanistic studies confirm biological plausibility: pollutants induce oxidative stress, neuroinflammation, epigenetic modifications, endocrine disruption, and neurotransmission alterations. These pathways converge to impair brain development, trigger systemic inflammation, and promote carcinogenic processes. Importantly, interactions with psychosocial stress and socioeconomic disadvantage amplify these risks, highlighting the multifactorial nature of indoor environmental health.
Comparison with Previous Reviews
Our findings align with earlier reviews emphasizing the neurodevelopmental toxicity of air pollution [69,70]. However, this review extends the scope by focusing specifically on indoor environments, where exposures often exceed outdoor levels. Moreover, we highlight epigenetic and mechanistic pathways that connect pollution not only to cognitive impairment but also to oncological and respiratory health outcomes, an area underrepresented in prior reviews.
Implications for the Study
The implications of these findings are highly relevant to public health and policy. Improved ventilation, low-emission materials, and indoor air cleaning technologies are critical to reduce pollutant exposure. Regulatory frameworks should expand from outdoor to indoor environments, especially schools, childcare centers, and residential buildings in high-traffic areas. As rising global temperatures increase reliance on indoor cooling, pollutant accumulation may worsen unless sustainable and heat-resilient ventilation systems are implemented. Interventions must prioritize low-income and high-stress populations, who face compounded risks from poor housing, limited resources, and higher pollutant exposure.
Strengths and Limitations of the Review
Strengths of this review include the integration of diverse study types (epidemiological, toxicological, mechanistic) and focus on pollutant-specific pathways. The consistent findings across multiple pollutants strengthen causal inference. Limitations include; (i) reliance on existing datasets, (ii) heterogeneity in exposure measurement, and (iii) the lack of standardized outcome assessments across studies. In addition, most studies originated from high-income countries, limiting generalizability to low- and middle-income (LMIC) contexts where indoor pollution from solid fuels remains widespread.
Research Gaps and Future Directions
Several gaps require urgent attention. Few studies account for cumulative and interactive effects of pollutant mixtures, despite real-world co-exposures. More longitudinal and multi-exposure studies are required. Research disproportionately focuses on Western populations; more data from LMIC settings with biomass fuel use and poor ventilation are needed. Further work is needed to unravel how epigenetic and endocrine pathways mediate pollutant effects across the lifespan. Rigorous evaluations of ventilation systems, filtration technologies, and sustainable building interventions should be prioritized. Research should assess how climate-related factors (heat, humidity, energy use) modulate indoor pollution and health outcomes.
Indoor environments are critical determinants of human health, particularly for vulnerable populations. Pollutants encountered indoors contribute to neurodevelopmental disorders, cancer risk, and respiratory disease through well-characterized biological pathways. Addressing these challenges requires multidisciplinary approaches that integrate engineering, architecture, public health, toxicology, and social sciences. Future policies must prioritize healthy, comfortable, and climate-resilient indoor environments as a cornerstone of public health protection.
Conclusions and Recommendations
Conclusions
This systematic review highlights that indoor environments are not neutral spaces, but dynamic sources of pollutant exposures that significantly impact human health. Pollutants such as particulate matter, volatile organic compounds, heavy metals, polycyclic aromatic hydrocarbons, flame retardants, and pesticides are consistently linked to neurodevelopmental impairments, cancer- related risks, respiratory disease, and systemic inflammation.
The biological plausibility of these outcomes is supported by strong mechanistic evidence, including oxidative stress, neuroinflammation, epigenetic modifications, endocrine disruption, and altered neurotransmission. These pathways converge to explain epidemiological observations of autism spectrum disorder, ADHD, cognitive deficits, asthma, and cancer in exposed populations.
Importantly, the burden of disease is disproportionately borne by pregnant women, fetuses, children, and socially disadvantaged groups, underscoring equity considerations in both research and interventions.
Recommendations
Policymakers should extend air quality regulations to indoor settings, including schools, childcare centers, and residential housing. Investments in ventilation, filtration, and low-emission building materials are essential to mitigate exposure. With climate change increasing reliance on indoor spaces, sustainable and heat-resilient ventilation systems are needed to avoid exacerbating pollutant accumulation. Targeted public health interventions and programs should focus on protecting vulnerable populations—particularly children in urban, low-ventilation, and socioeconomically disadvantaged households.
Future research should focus on: longitudinal studies of multi- pollutant exposures; expansion of research in LMICs; mechanistic studies clarifying epigenetic and endocrine pathways, and intervention trials testing the effectiveness of air cleaning technologies and building modifications. Indoor environmental health must bring together experts in engineering, architecture, toxicology, epidemiology, and social sciences to design and implement holistic solutions.
Consent for Publication
Not applicable.
Funding
This review received no external funding.
Credit Authorship Contribution Statement
Rachael Gakii Murithi: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Visualization, Validation, Writing - original draft.
Chan Lu: Supervision, Writing - review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgement
Not applicable.
References
- Edwards, S. C., Jedrychowski, W., Butscher, M., Camann, D., Kieltyka, A., Mroz, E., ... & Perera, F. (2010). Prenatal exposure to airborne polycyclic aromatic hydrocarbons and children’s intelligence at 5 years of age in a prospective cohort study in Poland. Environmental health perspectives, 118(9), 1326-1331.
- Allen, J. L., Oberdorster, G., Morris-Schaffer, K., Wong, C., Klocke, C., Sobolewski, M., ... & Cory-Slechta, D. A. (2017). Developmental neurotoxicity of inhaled ambient ultrafine particle air pollution: Parallels with neuropathological and behavioral features of autism and other neurodevelopmental disorders. Neurotoxicology, 59, 140-154.
- Tang, X., Bai, Y., Duong, A., Smith, M. T., Li, L., & Zhang,L. (2009). Formaldehyde in China: production, consumption, exposure levels, and health effects. Environment international, 35(8), 1210-1224.
- Wang, J., Lai, S., Ke, Z., Zhang, Y., Yin, S., & Zheng, J. (2014). Exposure assessment, chemical characterization and source identification of PM2. 5 for school children and industrialdownwind residents in Guangzhou, China. Environmental geochemistry and health, 36(3), 385-397.
- Wang, S., Hu, C., Lu, A., Wang, Y., Cao, L., Wu, W., ... &Yan, C. (2021). Association between prenatal exposure to persistent organic pollutants and neurodevelopment in early life: A mother-child cohort (Shanghai, China). Ecotoxicology and Environmental Safety, 208, 111479.
- Murithi, R. G., Lu, C., & Tang, K. (2025). Prenatal Exposure to Airborne and Indoor Pollutants and the Risk of Childhood Neurodevelopmental Disorders. J Anesth Pain Med, 10(3), 01-13.
- Perera, F. P., Li, Z., Whyatt, R., Hoepner, L., Wang, S., Camann, D., & Rauh, V. (2009). Prenatal airborne polycyclic aromatic hydrocarbon exposure and child IQ at age 5 years. Pediatrics, 124(2), e195-e202.
- Jedrychowski, W., Perera, F. P., Tang, D., Stigter, L., Mroz, E., Flak, E., ... & Jacek, R. (2012). Impact of barbecued meat consumed in pregnancy on birth outcomes accounting for personal prenatal exposure to airborne polycyclic aromatic hydrocarbons: birth cohort study in Poland. Nutrition, 28(4), 372-377.
- Bouchard, M. F., Chevrier, J., Harley, K. G., Kogut, K., Vedar, M., Calderon, N., ... & Eskenazi, B. (2011). Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environmental health perspectives, 119(8), 1189-1195.
- Al-Saleh, I., Nester, M., Abduljabbar, M., Al-Rouqi, R., Eltabache, C., Al-Rajudi, T., & Elkhatib, R. (2016). Mercury (Hg) exposure and its effects on Saudi breastfed infant's neurodevelopment. International journal of hygiene and environmental health, 219(1), 129-141.
- Dickerson, A. S., Rahbar, M. H., Bakian, A. V., Bilder, D. A., Harrington, R. A., Pettygrove, S., ... & Zahorodny, W. M. (2016). Autism spectrum disorder prevalence and associations with air concentrations of lead, mercury, and arsenic. Environmental monitoring and assessment, 188(7), 407.
- Rauh, V. A., Perera, F. P., Horton, M. K., Whyatt, R. M., Bansal, R., Hao, X., ... & Peterson, B. S. (2012). Brain anomalies in children exposed prenatally to a common organophosphate pesticide. Proceedings of the National Academy of Sciences, 109(20), 7871-7876.
- Liao, H., Murithi, R. G., Lu, C., Yang, W., Liu, Z., & Cao, L. (2023). Long-term exposure to traffic-related air pollution and temperature increases gynecological cancers. Building and Environment, 230, 109989.
- Block, M. L., & Calderón-Garcidueñas, L. (2009). Air pollution: mechanisms of neuroinflammation and CNS disease. Trends in neurosciences, 32(9), 506-516.
- Calderón-Garcidueñas, L., Engle, R., Mora-Tiscareño, A., Styner, M., Gómez-Garza, G., Zhu, H., ... & D’Angiulli, A. (2011). Exposure to severe urban air pollution influences cognitive outcomes, brain volume and systemic inflammation in clinically healthy children. Brain and cognition, 77(3), 345-355.
- Guxens, M., Lubczyska, M. J., Muetzel, R. L., Dalmau-Bueno, A., Jaddoe, V. W., Hoek, G., ... & El Marroun,H. (2018). Air pollution exposure during fetal life, brainmorphology, and cognitive function in school-age children.Biological psychiatry, 84(4), 295-303.
- Calderón-Garcidueñas, L., Mora-Tiscareño, A., Ontiveros, E., Gómez-Garza, G., Barragán-Mejía, G., Broadway, J., ... & Engle, R. W. (2008). Air pollution, cognitive deficits and brain abnormalities: a pilot study with children and dogs. Brain and cognition, 68(2), 117-127.
- Modesti, P. A., Reboldi, G., Cappuccio, F. P., Agyemang, C., Remuzzi, G., Rapi, S., ... & ESH Working Group on CV Risk in Low Resource Settings. (2016). Panethnic differences in blood pressure in Europe: a systematic review and meta- analysis. PloS one, 11(1), e0147601.
- Brown, D. M., Wilson, M. R., MacNee, W., Stone, V., & Donaldson, K. (2001). Size-dependent proinflammatory effects of ultrafine polystyrene particles: a role for surface area and oxidative stress in the enhanced activity of ultrafines. Toxicology and applied pharmacology, 175(3), 191-199.
- Oberdörster, G., Ferin, J., & Lehnert, B. E. (1994). Correlation between particle size, in vivo particle persistence, and lung injury. Environmental health perspectives, 102(suppl 5), 173- 179.
- Lippmann, M., Chen, L. C., Gordon, T., Ito, K., & Thurston,G. D. (2013). National Particle Component Toxicity (NPACT) Initiative: integrated epidemiologic and toxicologic studies of the health effects of particulate matter components. Research Report (Health Effects Institute), (177), 5-13.
- Harris MH, Gold DR, Rifas-Shiman SL, Melly SJ, Zanobetti A, Coull BA, Schwartz JD, Gryparis A, Kloog I, Koutrakis P et al: Prenatal and childhood traffic-related air pollution exposure and childhood executive function and behavior. Neurotoxicology and Teratology 2016, 57:60-70.
- Mohammadi M, Mohammadi P: Indoor air pollution and acute respiratory infection among children: an update biomass smoke. Journal of Air Pollution and Health 2018, 3(1):49-62.
- Parikh R, Rao SR, Kukde R, O’Connor GT, Patel A, Hibberd PL: Assessing the respiratory effects of air pollution from biomass Cookstoves on pregnant women in rural India. International journal of environmental research and public health 2021, 18(1):183.
- Sinha, D., & Ray, M. R. (2015). Health effects of indoor air pollution due to cooking with biomass fuel. In Studies on Experimental Toxicology and Pharmacology (pp. 267-302). Cham: Springer International Publishing.
- Kumar, R., Nagar, J. K., Goel, N., Kumar, P., Kushwah, A. S., & Gaur, S. N. (2015). Indoor air pollution and asthma in children at Delhi, India. Advances in Respiratory Medicine, 83(4), 275-282.
- Guxens, M.,Aguilera, I., Ballester, F., Estarlich, M., Fernández- Somoano, A., Lertxundi, A., ... & INMA (INfancia y Medio Ambiente) Project. (2012). Prenatal exposure to residential air pollution and infant mental development: modulation by antioxidants and detoxification factors. Environmental health perspectives, 120(1), 144-149.
- Lertxundi, A., Andiarena, A., Martínez, M. D., Ayerdi, M., Murcia, M., Estarlich, M., ... & Ibarluzea, J. (2019). Prenatal exposure to PM2. 5 and NO2 and sex-dependent infant cognitive and motor development. Environmental research, 174, 114-121.
- Lee, M., Ha, M., Hong, Y. C., Park, H., Kim, Y., Kim, E.J., ... & Ha, E. (2019). Exposure to prenatal secondhand smoke and early neurodevelopment: Mothers and Children’s Environmental Health (MOCEH) study. Environmental Health, 18(1), 22.
- Weinstein, J. R., Asteria-Peñaloza, R., Diaz-Artiga, A., Davila, G., Hammond, S. K., Ryde, I. T., ... & Thompson, L.M. (2017). Exposure to polycyclic aromatic hydrocarbons and volatile organic compounds among recently pregnant rural Guatemalan women cooking and heating with solid fuels. International journal of hygiene and environmental health, 220(4), 726-735.
- White, A. J., Bradshaw, P. T., Herring, A. H., Teitelbaum,S. L., Beyea, J., Stellman, S. D., ... & Gammon, M. D. (2016). Exposure to multiple sources of polycyclic aromatic hydrocarbons and breast cancer incidence. Environment international, 89, 185-192.
- Annavarapu, R. N., & Kathi, S. (2016). Cognitive disorders in children associated with urban vehicular emissions. Environmental Pollution, 208, 74-78.
- Parajuli, R. P., Fujiwara, T., Umezaki, M., & Watanabe, C. (2013). Association of cord blood levels of lead, arsenic, and zinc with neurodevelopmental indicators in newborns: a birth cohort study in Chitwan Valley, Nepal. Environmental research, 121, 45-51.
- Miyashita, C., Sasaki, S., Saijo, Y., Okada, E., Kobayashi, S., Baba, T., ... & Kishi, R. (2015). Demographic, behavioral, dietary, and socioeconomic characteristics related to persistent organic pollutants and mercury levels in pregnant women in Japan. Chemosphere, 133, 13-21.
- Jurewicz, J., Polanska, K., & Hanke, W. (2013). Chemical exposure early in life and the neurodevelopment of children– an overview of current epidemiological evidence. Annals of agricultural and environmental medicine, 20(3).
- Toscano, C. D., & Guilarte, T. R. (2005). Lead neurotoxicity: from exposure to molecular effects. Brain Research Reviews, 49(3), 529-554.
- Wright, R. O., & Baccarelli, A. (2007). Metals and neurotoxicology. The Journal of nutrition, 137(12), 2809- 2813.
- Guxens, M., Garcia-Esteban, R., Giorgis-Allemand, L., Forns, J., Badaloni, C., Ballester, F., ... & Sunyer, J. (2014). Air pollution during pregnancy and childhood cognitive and psychomotor development: six European birth cohorts. Epidemiology, 25(5), 636-647.
- Boucher, O., Muckle, G., & Bastien, C. H. (2009). Prenatal exposure to polychlorinated biphenyls: a neuropsychologic analysis. Environmental health perspectives, 117(1), 7-16.
- Nakajima, S., Saijo, Y., Kato, S., Sasaki, S., Uno, A., Kanagami, N., ... & Kishi, R. (2006). Effects of prenatal exposure to polychlorinated biphenyls and dioxins on mental and motor development in Japanese children at 6 months ofage. Environmental health perspectives, 114(5), 773-778.
- Grandjean, P., & Landrigan, P. J. (2006). Developmental neurotoxicity of industrial chemicals. The Lancet, 368(9553), 2167-2178.
- Park, H. Y., Hertz-Picciotto, I., Sovcikova, E., Kocan, A., Drobna, B., & Trnovec, T. (2010). Neurodevelopmental toxicity of prenatal polychlorinated biphenyls (PCBs) by chemical structure and activity: a birth cohort study. Environmental Health, 9(1), 51.
- Forns, J., Torrent, M., Garcia-Esteban, R., Grellier, J., Gascon, M., Julvez, J., ... & Sunyer, J. (2012). Prenatal exposure to polychlorinated biphenyls and child neuropsychological development in 4-year-olds: an analysis per congener and specific cognitive domain. Science of the total environment, 432, 338-343.
- Sagiv, S. K., Thurston, S. W., Bellinger, D. C., Tolbert,P. E., Altshul, L. M., & Korrick, S. A. (2010). Prenatal organochlorine exposure and behaviors associated with attention deficit hyperactivity disorder in school-aged children. American journal of epidemiology, 171(5), 593-601.
- Epa, E. (2009). Reregistration eligibility decision (RED) for malathion. Washington, DC: United States Environmental Protection Agency.
- Eskenazi, B., Marks, A. R., Bradman, A., Harley, K., Barr, D. B., Johnson, C., ... & Jewell, N. P. (2007). Organophosphate pesticide exposure and neurodevelopment in young Mexican- American children. Environmental health perspectives, 115(5), 792-798.
- Bouchard, M. F., Chevrier, J., Harley, K. G., Kogut, K., Vedar, M., Calderon, N., ... & Eskenazi, B. (2011). Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environmental health perspectives, 119(8), 1189-1195.
- Engel SM, Wetmur J, Chen J, Zhu C, Barr DB, Canfield RL, Wolff MS: Prenatal exposure to organophosphates, paraoxonase 1, and cognitive development in childhood. Environmental health perspectives 2011, 119(8):1182-1188.
- Gump, B. B., Wu, Q., Dumas, A. K., & Kannan, K. (2011).Perfluorochemical (PFC) exposure in children: associations with impaired response inhibition. Environmental science & technology, 45(19), 8151-8159.
- Hoffmann, S., de Vries, R. B., Stephens, M. L., Beck, N. B.,Dirven, H. A., Fowle III, J. R., ... & Tsaioun, K. (2017). A primer on systematic reviews in toxicology. Archives of toxicology, 91(7), 2551-2575.
- Becerra, T. A., Wilhelm, M., Olsen, J., Cockburn, M., & Ritz,B. (2013). Ambient air pollution and autism in Los Angeles county, California. Environmental health perspectives, 121(3), 380-386.
- Froehlich, T. E., Anixt, J. S., Loe, I. M., Chirdkiatgumchai, V., Kuan, L., & Gilman, R. C. (2011). Update on environmental risk factors for attention-deficit/hyperactivity disorder. Current psychiatry reports, 13(5), 333-344.
- Hoffman, K., Webster, T. F., Weisskopf, M. G., Weinberg, J., & Vieira, V. M. (2010). Exposure to polyfluoroalkyl chemicals and attention deficit/hyperactivity disorder in US children 12–15 years of age. Environmental health perspectives, 118(12), 1762-1767.
- Gascon, M., Vrijheid, M., Martínez, D., Forns, J., Grimalt,J. O., Torrent, M., & Sunyer, J. (2011). Effects of pre and postnatal exposure to low levels of polybromodiphenyl ethers on neurodevelopment and thyroid hormone levels at 4 years of age. Environment international, 37(3), 605-611.
- Chiu, Y. H. M., Hsu, H. H. L., Coull, B. A., Bellinger, D. C., Kloog, I., Schwartz, J., ... & Wright, R. J. (2016). Prenatal particulate air pollution and neurodevelopment in urban children: examining sensitive windows and sex-specific associations. Environment international, 87, 56-65.
- Chiu, Y. H. M., Bellinger, D. C., Coull, B. A., Anderson, S., Barber, R., Wright, R. O., & Wright, R. J. (2013). Associations between traffic-related black carbon exposure and attention in a prospective birth cohort of urban children. Environmental health perspectives, 121(7), 859-864.
- Nie, J., Li, J., Cheng, L., Deng, Y., Li, Y., Yan, Z., ... &Tang, D. (2019). Prenatal polycyclic aromatic hydrocarbons metabolites, cord blood telomere length, and neonatal neurobehavioral development. Environmental research, 174, 105-113.
- Perera, F. P., Rauh, V., Tsai, W. Y., Kinney, P., Camann, D., Barr, D., ... & Whyatt, R. M. (2003). Effects of transplacental exposure to environmental pollutants on birth outcomes in a multiethnic population. Environmental health perspectives, 111(2), 201-205.
- Abraham, E., Rousseaux, S., Agier, L., Giorgis-Allemand, L., Tost, J., Galineau, J., ... & EDEN Mother-Child Cohort Study Group. (2018). Pregnancy exposure to atmospheric pollution and meteorological conditions and placental DNA methylation. Environment international, 118, 334-347.
- Al-Gubory, K. H. (2014). Environmental pollutants and lifestyle factors induce oxidative stress and poor prenatal development. Reproductive biomedicine online, 29(1), 17-31.
- Lee, K. H., Shu, X. O., Gao, Y. T., Ji, B. T., Yang, G., Blair, A.,... & Kang, D. (2010). Breast cancer and urinary biomarkers of polycyclic aromatic hydrocarbon and oxidative stress in the Shanghai Women's Health Study. Cancer epidemiology, biomarkers & prevention, 19(3), 877-883.
- Bose, S., Chiu, Y. H. M., Hsu, H. H. L., Di, Q., Rosa, M. J.,Lee, A., ... & Wright, R. J. (2017). Prenatal nitrate exposure and childhood asthma. Influence of maternal prenatal stress and fetal sex. American journal of respiratory and critical care medicine, 196(11), 1396-1403.
- Yin, W., Hou, J., Xu, T., Cheng, J., Li, P., Wang, L., ... & Yuan,J. (2018). Obesity mediated the association of exposure to polycyclic aromatic hydrocarbon with risk of cardiovascular events. Science of the Total Environment, 616, 841-854.
- Jedrychowski, W. A., Perera, F. P., Maugeri, U., Mroz, E., Klimaszewskaâ?ÂÂRembiasz, M., Flak, E., ... & Spengler, J. D. (2010). Effect of prenatal exposure to fine particulate matter on ventilatory lung function of preschool children of nonâ? smoking mothers. Paediatric and perinatal epidemiology, 24(5), 492-501.
- Genc, S., Zadeoglulari, Z., Fuss, S. H., & Genc, K. (2012). The adverse effects of air pollution on the nervous system. Journal of toxicology, 2012(1), 782462.
- Jedrychowski, W., Perera, F., Mrozek-Budzyn, D., Mroz, E., Flak, E., Spengler, J. D., ... & Skolicki, Z. (2009). Gender differences in fetal growth of newborns exposed prenatally to airborne fine particulate matter. Environmental research, 109(4), 447-456.
- Clifford, A., Lang, L., Chen, R., Anstey, K. J., & Seaton, A. (2016). Exposure to air pollution and cognitive functioning across the life course–a systematic literature review. Environmental research, 147, 383-398.
- Bolton, J. L., Huff, N. C., Smith, S. H., Mason, S. N., Foster,W. M., Auten, R. L., & Bilbo, S. D. (2013). Maternal stress and effects of prenatal air pollution on offspring mental health outcomes in mice. Environmental health perspectives, 121(9), 1075-1082.
- Bellinger, D. C. (2013). Prenatal exposures to environmental chemicals and children’s neurodevelopment: an update. Safety and health at work, 4(1), 1-11.
- Costa, L. G., Chang, Y. C., & Cole, T. B. (2017). Developmental neurotoxicity of traffic-related air pollution: focus on autism. Current environmental health reports, 4(2), 156-165.



