Original article - (2025) Volume 1, Issue 2
Association of Vimentin Expression in Tumor Cells with Demographic Parameters and TNM Staging of Oral Squamous Cell Carcinoma
2Dr. Sanjida Rahman, Curator, Department of Pathology, Uttara Adhunik Medical College, Dhaka, Bangladesh
3Dr. Kazi Nishat Ara Begum, Professor, Department of Pathology, Holy Family Medical College and Hos, Bangladesh
4Dr. Babli Sultana Bahar, Lecturer, Department of Pathology, Shaheed Suhrawardy Medical College, Dhak, Bangladesh
5Dr. Mohsina Akter Mou, OSD, DG Health, Mohakhali, Dhaka, Bangladesh
6Dr. Nusrat Jahan Farjana, Pathologist, Department of Pathology, Faridpur Medical College, Bangladesh
7Dr. Munira Anjum, Assistant Professor, Department of Pathology, Marks Medical College, Dhaka, Bangladesh
8Dr. Sayedatus saba, Corresponding author, MBBS, MD (Pathology), Clinical Pathologist, Department of , Bangladesh
Received Date: Jul 10, 2025 / Accepted Date: Aug 11, 2025 / Published Date: Aug 22, 2025
Copyright: ©2025 Sayedatus Saba, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Akther, S., Bahar, B. S., Rahman, S., Mou, M. A., Saba, S., et al. (2025). Association of Vimentin Expression in Tumor Cells with Demographic Parameters and TNM Staging of Oral Squamous Cell Carcinoma. Arch Cienc Investig, 1(2), 01-09.
Abstract
Background: The development of oral cancer has been associated with a number of risk factors. Thus, the search for novel molecular markers that can forecast the course of the malignancy and possible therapeutic targets is urgent. Vimentin has been found to be strongly associated with the development of OSCC (oral squamous cell carcinoma). The purpose of this research is to determine reliable histologic prognostic markers and examine the association between vimentin expression and TNM Staging along with demographic parameters in OSCC.
Objectives: To see the association of vimentin expression in tumor cells with TNM staging and demographic parameters of oral squamous cell carcinoma.
Method: A cross-sectional study was carried out in the Department of Pathology, Shaheed Suhrawardy Medical College and Hospital, Dhaka, Bangladesh from September 2021 to August 2023. A total of 51 histologically diagnosed cases of OSCC were selected for the study. Among these, 25 cases were resected tumor specimens and 26 were oral mucosal biopsy. Paraffin blocks and slides were prepared after grossing. H&E and IHC staining were done and analyzed accordingly. All cases were categorized into groups according to TNM staging. The statistical analysis was carried out using the SPSS version 26.
Result: In this study, there was no statistically significant association between vimentin expression and patients’ demographic characteristics like age and sex. Also, there is no significant association between Vimentin expression and TNM staging of tumor. Association between Lymph node status and Vimentin immunoreactivity was statistically insignificant as well.
Discussion: This study revealed that there is no association of vimentin with patient age, sex and TNM stage. So, these parameters should be used in addition to conventional grading in the case of evaluation of OSCC cases for predicting patient outcomes as well as planning further management.
Keywords
OSCC, Vimentin, H&E, IHC, TNM
Introduction
It is estimated that nearly 95% of all squamous cell carcinomas of the head and neck originate in the oral cavity. The ventral surface of the tongue, the floor of the mouth, the lower lip, the soft palate, and the gingiva are favored sites for the development of squamous cell carcinoma.
Oral cancer is the sixth most prevalent form of cancer in the world [1]. Despite diagnostic and therapeutic advances, oral malignancies remain difficult to diagnose and treat, with a five- year survival rate of approximately 45 percent [2]. Bangladesh experienced 8137 deaths and 13985 (8.9%) new cases in 2020 [3]. Due to local recurrence and lymph node metastasis, the prognosis for oral squamous cell carcinoma is generally poor [4].
Vimentin is a 57kDa protein component of the cytoskeleton. Upregulation of vimentin in epithelial cells leads to alterations in cell morphology, loss of cell-cell contact, and enhanced cell motility, which is the first stage in tissue invasion and metastasis to distant organs [5]. Also, vimentin regulates cell migration in lymph node. Thus, the upregulation of vimentin expression has been closely associated with cancer cell differentiation, invasion, and metastasis.
TNM staging for OSCC
PRIMARY TUMOR (T)
TX Primary tumor cannot be assessed
T0 No evidence of primary tumor
Tis Carcinoma in situ
T1 Tumor 2 cm or less in greatest dimension
T2 Tumor more than 2 cm but not greater than 4 cm in greatest dimension
T3 Tumor more than 4 cm in greatest dimension
T4a Moderately advanced local disease (Tumor invades through cortical bone, inferior alveolar nerve, floor of mouth, or skin of face—that is, chin or nose)
T4b Very advanced local disease Tumor invades masticator space, pterygoid plates, or skull base and/or encases internal carotid artery [6].
REGIONAL LYMPH NODES (N)
NX Regional lymph nodes cannot be assessed
N0 No regional nodes metastasis
N1* Metastasis in a single ipsilateral lymph node, 3 cm or less in greatest dimension
N2* Metastasis in a single ipsilateral lymph node, more than 3 cm but not more than 6 cm in greatest dimension; or in multiple ipsilateral lymph nodes, none more than 6 cm in greatest dimension; or in bilateral or contralateral lymph nodes, none greater than 6 cm in greatest dimension
N2a* Metastasis in a single ipsilateral lymph node, more than 3 cm but not more than 6 cm in greatest dimension
N2b* Metastasis in multiple ipsilateral lymph nodes, none more than 6 cm in greatest dimension
N2c* Metastasis in bilateral or contralateral lymph nodes, none more than 6 cm in greatest dimension
N3* Metastasis in a lymph node more than 6 cm in greatest dimension.
DISTANT METASTASIS (M)
MX Distant metastasis cannot be assessed
M0 No distant metastasis
M1 Distant metastasis
STAGE GROUPING
|
Stage 0 |
Tis |
N0 |
M0 |
|
Stage I |
T1 |
N0 |
M0 |
|
Stage II |
T2 |
N0 |
M0 |
|
Stage III |
T3 |
N0 |
M0 |
|
T1 |
N1 |
M0 |
|
|
T2 |
N1 |
M0 |
|
|
T3 |
N1 |
M0 |
|
|
Stage IVA |
T4a |
N0 |
M0 |
|
T4a |
N1 |
M0 |
|
|
T1 |
N2 |
M0 |
|
|
T2 |
N2 |
M0 |
|
|
|
T3 |
N2 |
M0 |
|
T4a |
N2 |
M0 |
|
|
Stage IVB |
Any T |
N3 |
M |
|
T4b |
Any N |
M0 |
|
|
Stage IVC |
Any T |
Any N |
M1 |
The purpose of this study is to observe vimentin immunostaining of paraffin-embedded oral cancer tissue in biopsy/resected specimens and evaluate its association with histopathological stage and other demographic parameters in order to determine its predictive value for invasion and metastasis of oral squamous cell carcinoma.
Materials & Methods
A cross-sectional study was carried out in the Department of Pathology, Shaheed Suhrawardy Medical College and Hospital, Dhaka, Bangladesh from September 2021 to August 2023. A total of 51 histologically diagnosed cases of OSCC were selected for the study. Among these, 25 cases were resected tumor specimens and 26 were oral mucosal biopsy. Paraffin blocks and slides were prepared after grossing. H&E and IHC staining were done and analyzed accordingly. All cases were categorized into groups according to TNM staging.
Among the 25 resected specimens, only 22 cases included lymph nodes with the excised tumor. So, lymph node status could be evaluated in these 22 cases only. Cases with lymph nodes were divided into metastatic and non-metastatic groups depending on the presence of lymph node metastasis.
Regarding the IHC staining intensity, cases were analyzed under light microscopy (40X), using the following 3-point scoring system:
0 (Negative)
1+ (Weak)
2+ (Moderate)
3+ (Strong)
According to the percentage of positive cells, cases were scored as:
0 = Negative
1 = <10%
2 = 11–50%
3 = 51–80%
4 = >80%
The final score calculated by multiplying staining intensity and positivity in tumor cells (range 0–12) dividing into three groups:
0= Negative
1-4= Low expression
> 4 = High expression
The statistical analysis was carried out using the SPSS version 26 [7].
Results
Ages of the patients ranged from 31 to 95 years. The highest number of cases (30.8%) belonged to 51-60 years age group.
Figure 1: Bar Diagram Represents Distribution of Patients According to Age
Females were found affected slightly more than male. Numbers of female patients were 30 (58.8%) and male patients were 21 (41.2%). Female to male ratio is 1:0.7.
Figure 2: Pie chart showing sex distribution of 51 patients
51-60 age group has the highest 33% vimentin High expression, whereas 31-40 age group has the lowest 8% vimentin low expression. Chi-Square (X2) test p-value (0.66) is not statistically significant.
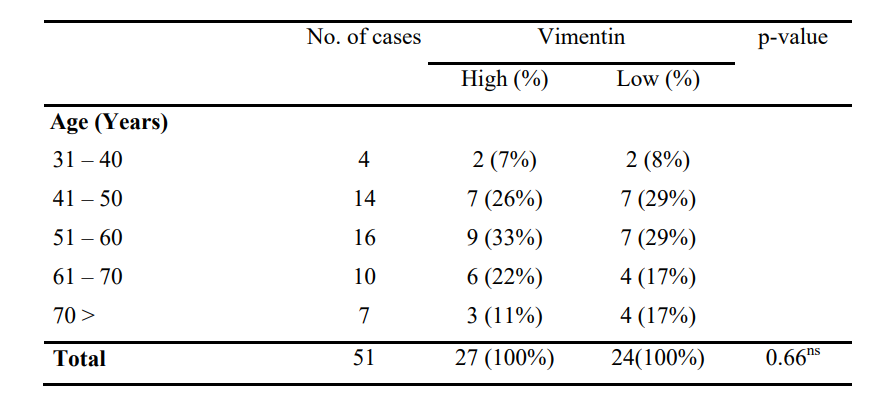
Table 1: Distribution of The Patients with Oscc According to Age by Vimentin Expression (N=51)
59% female showed highest vimentin high score, whereas 42% male show lowest vimentin low expression. Chi-Square (X2) test p-value (0.88) is not statistically significant.
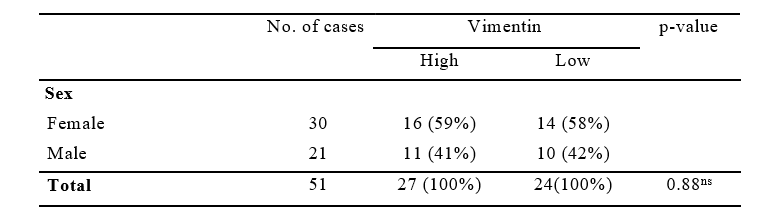
Table 2: Distribution of the Patients with OSCC According to Sex by Vimentin Expression (n=51)
Highest high expression 38.4% was observed in T1 and lowest in T4. Highest low expression 41.7% was observed in T2 and lowest 8.3% in T4. Chi-Square (X2) test p-value (0.9) is not statistically significant.
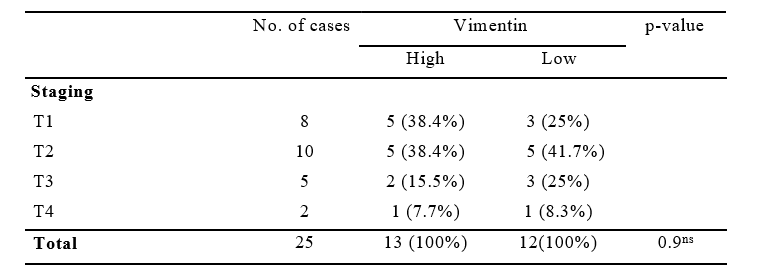
Table 3: Association of Vimentin Expression with Tumor Staging in 25 cases.
50% cases showed high immunoreactivity for Vimentin in both metastatic and non-metastatic group. Chi-Square (X2) test p-value is 1.000, which is not statistically significant.
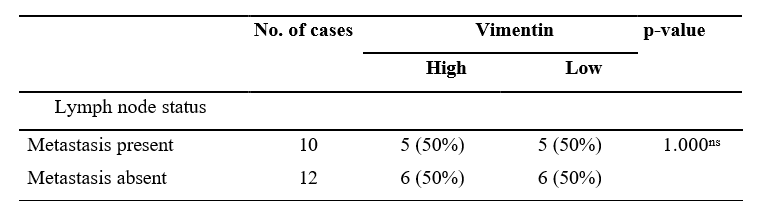
Table 4: Association between Lymph Node Status and Vimentin Immunoreactivity (n=22)
Discussion
The mean age of occurrence of our study is (57.6 ± 12.29). Comparable age distribution is found in the study conducted by Rahman and his team which showed most cases in the fifth decade of life and the mean age was 50.4±3 [8]. This study is also comparable with Smitha and his colleagues, which showed that OSCC was significantly higher in middle aged group (41-60 years) [9].
The gender related findings of the current study (Male: Female ratio, 1:1.4) are also consistent with the local study by Saha and his team which showed the ratio of malignant lesion 1:1.27 [10]. Non-significant association between vimentin immunohisto- chemical expression with different age and sex of OSCC pa- tients is consistent with other studies like Liu and his team.
According to the immunostaining results of the present study, more than half of the OSCC cases (53%) were categorized as having high vimentin expression score and 47% cases showed low vimentin expression scores. No case was detected as negative staining. Similar to these findings, Puneeta and her team showed positive vimentin immunoreactivity in all examined OSCC cases with high expression in 68.3% of the cases [11]. This suggests that vimentin play an important role in oral tumor progression.
In this study we observed that Vimentin over expression was distributed as: T1 38.4%; T2 38.4%; T3 15.5%; T4 7.7% and showed a non-significant p-value (0.9). This result correlates with Liu and his team.
Out of total 51 cases, only 22 specimens included lymph nodes with the excised tumor. Lymph node status was evaluated in these cases only and there were no significant association of expression of vimentin with lymph node metastasis. This supports the result of a previous study done by Indira and her team, who also did not find any association of vimentin with lymph node metastasis [12]. It would be more conclusive if a prospective study could be done with a larger number of excised tumors including lymph nodes and long-term follow-up data.
Conclusion
Vimentin immunohistochemistry is helpful in predicting tumor behavior and prognosis and can serve as a molecular target for therapeutic implementations. If this is used in addition to TNM staging along with conventional grading in the case of evaluation of OSCC cases, could be more beneficial for predicting patient outcomes as well as planning further management.
Human Ethics Declarations
This research work has been approved by Institutional Ethics Committee of Shahid Suhrawardy Medical college and Hospital, Dhaka.
Consent to Participate Declaration
Informed written consents were taken from all the patients or patient's relatives before collecting their samples for histopathological and immunohistochemical studies.
Acknowledgements
I thank my husband Md. Amzad Hossain for his immense support.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.Conflicts of Interest
None.References
- Sah, R., & Akhter, M. (2020). Oral cancer senario in multiple centers of Dhaka, Bangladesh. International Journal of Innovative Research in Medical Science (IJIRMS), 5(11).
- Sridharan, G., & Bhandare, P. (2017). Microinvasive oral squamous cell carcinoma-A clinicopathological study. Medical Research Archives, 5(7)..
- Globocan, W. H. O. (2012). Estimated cancer incidence, mortality and prevalence worldwide in 2012. Int Agency Res Cancer.
- Elango, J. K., Gangadharan, P., Sumithra, S., & Kuriakose,M. A. (2006). Trends of head and neck cancers in urban and rural India. Asian Pacific Journal of Cancer Prevention, 7(1), 108.
- Battaglia, R. A., Delic, S., Herrmann, H., & Snider, N. T. (2018). Vimentin on the move: new developments in cell migration. F1000Research, 7, F1000-Faculty.
- College of American Pathologists. Protocol for the Examination of Specimens from Patients with Carcinoma of the Lip and Oral Cavity, Version 4.1.0.1. November 2021.
- Liu, L. K., Jiang, X. Y., Zhou, X. X., Wang, D. M., Song,X. L., & Jiang, H. B. (2010). Upregulation of vimentinand aberrant expression of E-cadherin/β-catenin complex in oral squamous cell carcinomas: correlation with the clinicopathological features and patient outcome. Modern Pathology, 23(2), 213-224.
- Rahman, T. Y., Mahanta, L. B., Chakraborty, C., Das, A. K., & Sarma, J. D. (2018). Textural pattern classification for oral squamous cell carcinoma. Journal of microscopy, 269(1), 85-93.
- Smitha, T., Mohan, C. V., & Hemavathy, S. (2017). Clinicopathological features of oral squamous cell carcinoma: A hospital-based retrospective study. Journal of Dr. YSR University of Health Sciences, 6(1), 29-34.
- Saha, M.K., Quruni, M.O., Afrin, S.S., Hossain, M.S., Mahmud, F., Mahmud, S.A., Jinnah, M.A. and Dewan, M.R., 2020. Extracellular Matrix Metalloproteinase Inducer (EMMPRIN/CD147) Expression and its Correlation with Progression of Oral Squamous Cell Carcinoma. Journal of Histopathology and Cytopathology, 4(2), pp.122-130.
- Puneeta, N., Santosh, T., Mishra, I., Gaikwad, P., & Sahu, A. (2022). Evaluation of e-cadherin and vimentin expression for different grades of oral epithelial dysplasia and oral squamous cell carcinoma–An immunohistochemical study. Journal of Oral and Maxillofacial Pathology, 26(2), 285.
- Sahu, I., Mathur, K. and Meel, M., 2021.Study of Immunohistochemical Expression Of E-Cadherin and Vimentin in Premalignant and Malignant Squamous Lesions of Oral Cavity and Oropharynx at a Tertiary Care Centre in North India. European Journal of Pharmaceutical and Medical Research, 7, pp. 324-330.
Figure 3: (Case no 46): Photomicrograph of Well Differentiated OSCC (Stage T1)
Figure 4: (Case no 17): Photomicrograph of Well Differentiated OSCC (Stage T1) with Low Cytoplasmic Expression of Vimentin
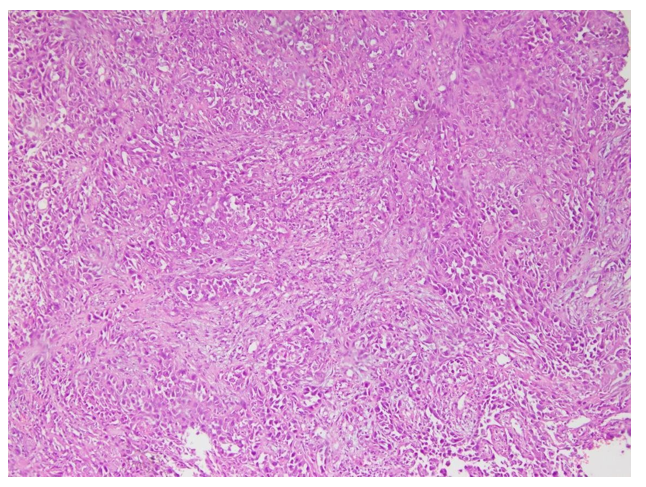
Figure 5: (Case no 49): Photomicrograph of Moderately Differentiated OSCC (Stage T2)
Figure 6: (Case no 49): Photomicrograph of Moderately Differentiated OSCC (Stage T2) with High Cytoplasmic Expression of Vimentin
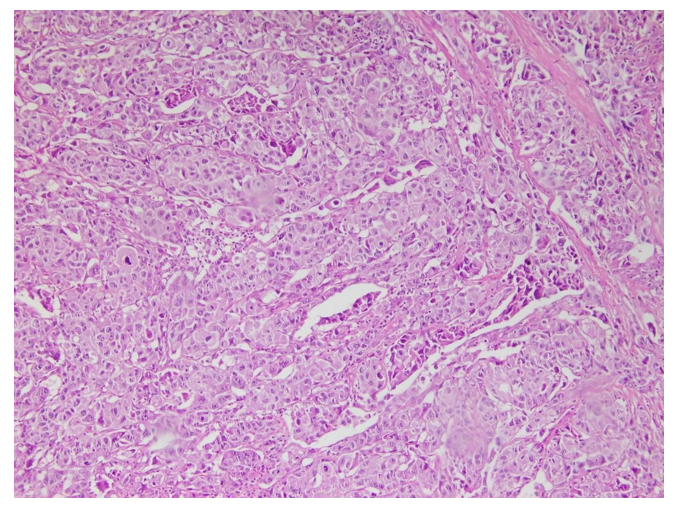
Figure 7: (Case no 51): Photomicrograph of Poorly Differentiated OSCC (Stage T3)
Figure 8: (Case no 51): Photomicrograph of Poorly Differentiated OSCC (Stage T3) with High Cytoplasmic Expression of Vimentin
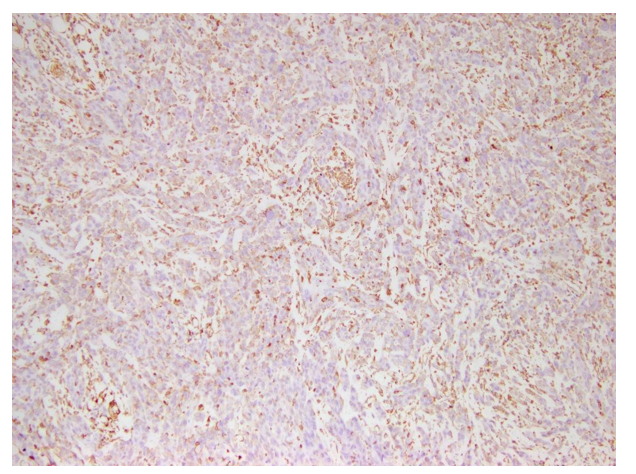
Figure 9: (Case no 3): Photomicrograph of Poorly Differentiated OSCC (Stage 4) with Low Cytoplasmic Expression of Vimentin