Research Article - (2020) Volume 5, Issue 1
Antioxidants Effect Changes in Systemic Parasympathetic and Sympathetic Nervous System Responses and Improve Outcomes
2Professor of Clinical Medicine, Hahnemann Hospital/Drexel University College of Medicine;, Associate Chief of Cardiology at the Drexel University College of Medical/Hahnemann Hospital; Medical Director, Franklin Cardiovascular Associates, PA, USA
3Franklin Cardiovascular Associates, PA, USA
4Parasympathetic & Sympathetic Nervous System Consultant, Franklin Cardiovascular Associates, PA and Autonomic Dysfunction and POTS Center; Neurocardiology Research Corporation, LLC, Wilmington, DE, USA
Received Date: Mar 25, 2020 / Accepted Date: Mar 30, 2020 / Published Date: Apr 16, 2020
Copyright: ©J Colombo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Parasympathetic and Sympathetic (P&S) imbalance is associated with increased morbidity and mortality risk, including heart failure, coronary artery disease, atrial and ventricular arrhythmias, hypertension and orthostatic disorders, and syncope. Most cardiac medications effect only one or the other of the P&S nervous systems, including: β-blockers, α-blockers, α-agonists, and anti-cholinergics. Non-pharmaceutical treatments also affect the P&S nervous systems; however, nonpharmaceutical treatment is rarely addressed or studied, despite significant evidence-based data demonstrating normalization of Sympathovagal Balance. Fifty consecutive patients from a busy suburban cardiology practice were enrolled in a supplement study. Patients were provided supplements that included 200mg of Alpha-Lipoic Acid (ALA) and 100mg of Co-Enzyme Q10 (CoQ10) which they were instructed to take twice a day, one each with breakfast and dinner. P&S Monitoring (Physio PS, Inc., Atlanta, GA, USA) and Quality of Life (QoL) questionnaires were administered at three month intervals. The combination of supplements, ALA and CoQ10, had a positive effect on the P&S nervous systems as measured directly and also indicated by improvements in BP, HR and reported QoL. The study indicates these supplements help to improve both the resting and challenge P&S responses and resultant physiology. The results suggest antioxidant therapy is a potential complimentary therapy to pharmacological management of patients with poorly controlled BP (i.e., hypertension and possible hypotension) as well as in patients with tachycardia or palpitations. This is a hypothesis generating study of significant importance in an often neglected area of cardiovascular disease in which additional research and studies are needed.
Keywords
Parasympathetic Response, Sympathetic Responses, Sympathovagal Balance, Supplements, Alpha-Lipoic Acid, Co- Enzyme Q10, Quality of Life, Cardiovascular Disease
Introduction
The Autonomic Nervous System, including the Parasympathetic and Sympathetic (P&S) Nervous Systems, control and coordinate all bodily functions including respirations, heart rate (HR), blood pressure (BP), the gastrointestinal system, the bladder, the immune system, sex function, and hormone function [1]. In fact, the P&S controls or coordinates all systems of the body. As with many things in life, there should be a proper balance between the P&S Systems. Certain supplements are able to effect changes in the P&S systems and therefore, affect the balance between the two autonomic branches [2].
P&S imbalance is often associated with heart failure, coronary artery disease, atrial and ventricular arrhythmias, hypertension and orthostatic disorders, and syncope. Cardiovascular autonomic control is an intricately balanced dynamic process designed for optimal functioning of the human body. Autonomic dysfunction or neuropathies promote and sustain the disease processes. Most cardiac medications effect only one or the other of the P&S nervous systems, including: β-blockers, α-blockers, α-agonists, and anti- cholinergics. Non-pharmaceutical treatments also affect the P&S nervous systems; however, non-pharmaceutical treatment is rarely addressed or studied, despite significant evidence-based data demonstrating normalization of Sympathovagal Balance (SB) [1- 11]. Antioxidants, both from the Mediterranean Diets and similar diets (e.g., the “Japomediterranean” Diet and the Okinawa Diet) rich in fruits and vegetables with fish and seafood as the primary sources of protein and supplements have demonstrated effectiveness in cardiovascular disorders [12-30]. Two supplemental antioxidants are studied: Alpha-Lipoic Acid (ALA) and Co-enzyme Q10 (CoQ10).
ALA and CoQ10 are two of the most powerful antioxidants produced in the body, not only in and of themselves, but also because they help to recycle other antioxidants, including Vitamins A, C, and E [2]. Alpha-Lipoic Acid tends to be selective for nerves [31-56]. CoQ10 tends to be selective for the heart muscle [57-66]. The body’s production of these antioxidants declines with age and duration of illness. This statistically significant volume of evidence validates that these antioxidants taken as supplements help to slow or normalize this decline. Slowing this decline prevents stress at the cellular level, thereby reducing stress at the system level. They do so by reducing both inflammatory and oxidative stress. Cellular stress reduction significantly helps to improve the health of mitochondria. Improved mitochondrial health improves the efficiency of energy production in the body and increases the availability of the energy molecule Adenosine Triphosphate (ATP). Nerve and cardiac tissue are some of the largest consumers of ATP. Antioxidants support the health and wellness of all systems throughout the body, both directly and indirectly, including via healthy nervous and cardiovascular systems. This helps to keep all cells within the body healthy and energized.
Methods
In 2016, in a busy cardiovascular center outside a large metropolitan area in the northeast United States, patients with established cardiovascular disease agreed to participate in a prospective, pilot, observational study. Fifty (50) consecutive patients were enrolled and 46 completed the study (ages 55.6 ± 15.3, max/min: 78/21 and 24/46 or 52.2% female). Each patient served as their own control. The cohort’s primary cardiac diagnoses include: Syncope (18/46, 39.1%), well controlled type 2 Diabetes Mellitus (DM2, 12/46, 26.1%), stable hypertension and related disorders (HTN, 27/46, 58.7%), and established and stable coronary artery disease (CAD, 10/46, 21.7%), congestive heart failure (CHF, 2/46, 4.3%), or arrhythmia (14/46, 30.4%). Patients were provided supplements that included 200mg of ALA and 100mg of CoQ10 which they were instructed to take twice a day, one each with breakfast and dinner. Also included in the supplements were: Vitamins D (1000 IU bid), B6 (25mg bid) & B12 (550mcg bid). No prescription medications were introduced or changed throughout the study. All supplements were over-the-counter.
Exclusion criteria included: prior history of myocardial infarction, cerebral vascular accident (stroke), unstable cardiac arrhythmias or heart failure, uncontrolled diabetes or hypertension, pregnancy, expectation of pregnancy, nursing or planning to nurse, any prior sensitivity to the supplement compounds or any component of the supplements, or unable to understand or sign a consent form. If prescription medicine regimen changed during the duration of the study, whether dosing or classification, those patients were also excluded. Otherwise, all other patients were welcomed to be included and those that agreed read and signed consent forms. All data were handled in accordance with HIPAA regulations.
At the start of the study, and after three and six months on the supplements, all subjects completed two Quality of Life (QoL) questionnaires: the SF-36 Questionnaire and the Nottingham Health Profile. The SF-36 Questionnaire is a general wellness questionnaire. The results may range from 0.0, indicating the poorest results, to 100.0 indicating the best results. The Nottingham Health Profile is also a general wellness questionnaire, but with scored sub-categories. The results may range from 0.0, indicating the best results, to 1.0 indicating the worst results.
To document P&S responses, patients were tested with the P&S Monitor (Physio PS Inc., Atlanta, GA, USA). The P&S Monitor is a diagnostic monitor that analyzes a patient’s instantaneous HR (cardiogram), BP and respiratory activity (RA) in response to the following maneuvers [1]: 5-minute of rest while sitting back and relaxed on a chair with firm back support, then 1-minute of 10-second deep breaths (6 breathes per minute or 0.1 Hz), followed by a minute of rest, then five Valsalva maneuvers over 1:35 minutes of less than 15 seconds each (including a 15 sec Valsalva maneuver), followed by two minutes of rest, and ending with a quick postural change to standing with 5-minutes of quiet stand [67]. The P&S Monitor quantifies Parasympathetic nervous system activity (P-activity) mathematically independent of and simultaneously with, Sympathetic nervous system activity (S-activity) [1]. Parameters recorded, both at rest and during challenge, include: average HR, BP, S-activity (LFa), P-activity (RFa), and Sympathovagal balance (SB = S-activity / P-activity).
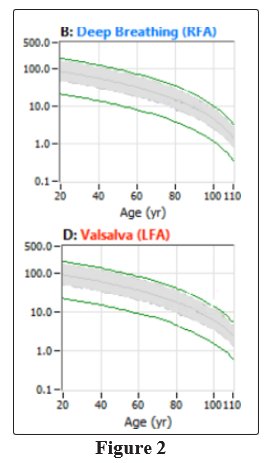
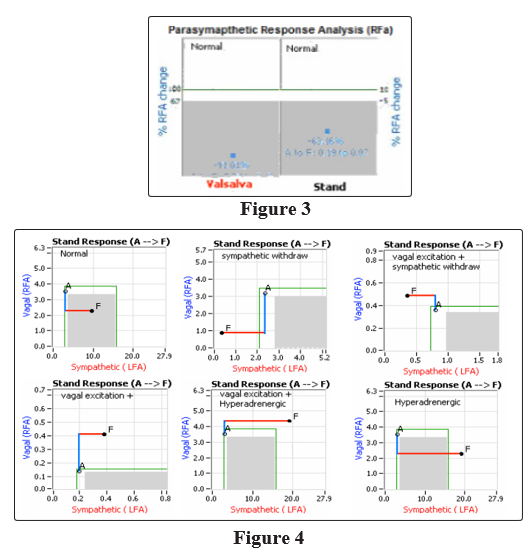
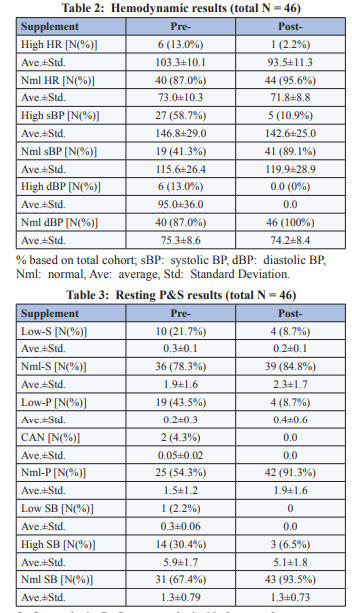
Normal resting ranges are: 1.0 bpm2 to 10.0 bpm2 for both P- and S-activity, and 0.4 to 3.0 for SB (unitless, see Figure 1) [1]. Normal ranges for P-activity during deep breathing and S-activity during Valsalva are age-, and baseline-, adjusted (see Figure 2) [1, 68]. Normal range for S-activity during deep breathing is a decrease from rest. Normal P-activity during Valsalva is a response less than a 6-fold increase with respect to rest (including any decrease, see Figure 3, left panel) [1]. Normal P-activity during stand is a response less than a 10% increase with respect to rest (including any decrease, see Figure 3, right panel) [1]. Normal S-activity during stand is a 10% to 500% increase with respect to rest (see Figure 4, upper left panel) [1]. As shown in Figures 1 through 4, the normal areas are shaded gray. Normal resting HR & BP are 60 to 90 bpm and 90/60 to 130/90 mmHg, respectively. Normal HR & BP responses to challenge are with respect to rest and include: a 10% to 20% decrease in response to deep breathing, and a 10% to 20% increase in response to Valsalva or Stand. Abnormally low BP responses to deep breathing or Valsalva indicate possible Baroreceptor Reflex dysfunction.
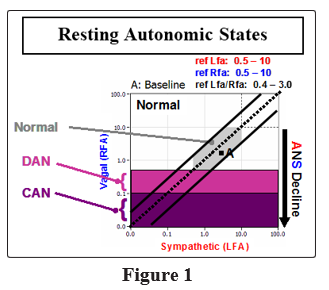
High SB (> 3.0, the area below and to the right of the lower diagonal line of Figure 1) indicates a resting Sympathetic Excess (SE) relative to resting P-activity and may be associated with stress, anxiety, pain, hypertension, or cardiovascular stress. Low SB (< 0.4, the area above and to the left of the upper diagonal line of Figure 1) indicates a resting PE relative to resting S-activity and may be associated with lightheadedness, fatigue, depression, or poor circulation. Low resting S- or P-activity (S or P < 0.5 bpm2, and P > 0.1 bpm2, see Figure 1, light purple shaded region) indicates Advanced Autonomic Dysfunction, which is similar to Diabetic Autonomic Neuropathy without the insulin affects; and is associated with increased morbidity risk [1, 69]. Very low resting P-activity (P < 0.1 bpm2, see Figure 1, dark purple shaded region) indicates Cardiovascular Autonomic Neuropathy (CAN), which is associated with increased mortality risk [70]. CAN is a normal function of aging, but may indicated advanced aging due to illness or injury. CAN with high SB (resting SE) is associated with high mortality risk (e.g., Major Adverse Cardiovascular Events or MACE) [71]. CAN with low SB (resting PE) is associated with elevated mortality risk (e.g., “Broken Heart Syndrome,” or Depression).
Valsalva or stand challenge PE is associated with patients’ unstable responses to disease or therapy. PE is associated with difficult to control BP, blood sugar, or hormone levels (e.g., thyroid hormones or estrogen), as well as unexplained arrhythmia and seizure. PE is also associated with symptoms of (preclinical) depression or anxiety, fatigue, exercise intolerance, persistent weight gain, sleep disturbances, hypertension secondary to P&S imbalance, GI upset, frequent headache or migraine, and dizziness or lightheadedness [1].
Valsalva Challenge SE (the area above the upper curve in Figure 2, lower panel) is associated with stress, hypertension, risk of cerebrovascular accident, myocardial infarction, or other MACE [1]. It tends to be an earlier indicator than high SB or CAN. Valsalva SE may present with symptoms of high stress (a SE response to lifestyle), excessive pain responses, hyper-activity, or symptoms of extreme fatigue (a consequence of SE). Valsalva Challenge SE is often a consequence of PE during Valsalva. PE may cause the Sympathetics to over-reacting to otherwise normal stimuli, thereby inflating the S-response to stress (SE) and thereby BP or HR responses. As a result, these normal stimuli are perceived as stressful or excessive. For example: 1) light touch may be perceived as painful as in Fibromyalgia, or 2) small concerns may lead to anxiety. Persistent challenge SE leads to excessive BP or HR responses.
Stand Sympathetic insufficiency or Sympathetic Withdrawal (SW, see Figure 4, upper and middle, right panels) is a postural change abnormality that indicates a lack proper coordination between the two autonomic branches and indicates Orthostatic Dysfunction or Orthostatic Intolerance (OI) [1]. Abnormalities in BP or HR responses with postural change help to specify the Orthostatic Dysfunction, including differentiate OI from Orthostatic Hypotension from Postural Orthostatic Tachycardia Syndrome (POTS). Stand SE (“Hyperadrenergic”, see Figure 4 bottom two panels) is associated with (pre-clinical) Syncope. The Syncope is specified as Vasovagal Syncope by Stand SE with PE (e.g., “Vagal Excitation,”, see Figure 4, lower middle). Syncope is specified as Neurogenic Syncope by Stand SE (e.g., see Figure 4, lower right) with a weak HR response to stand. Cardiogenic Syncope is a diagnosis by omission, and additional testing is needed to positively identify [72].
All patients read, understood, and signed patient consent forms and were provided supplements, at no cost, while participating in the study. Patients served as their own controls. Data analysis was performed in SPSS v22.0. Significance was defined as p < 0.10.
Results
The cohort’s secondary diagnoses include: chronic obstructive pulmonary disease (COPD; 10/46, 10.9%), Anxiety/Depression and associated syndromes (29/46, 63.0%), and Chronic Fatigue Syndrome (CFS; 9/46, 19.6%) and Autonomic (P&S) Dysfunction (46, 100%).
Hemodynamic Analysis
There were a total of 46 patients who completed the study, including remaining on the supplements and taking more than one P&S test and completing more than one questionnaire. These results are based only on these 46 who completed the study. Upon enrollment (prior to supplementation, or pre-supplement), 6/46 (13.0%) patients demonstrated resting HRs over 90 bmp. At the end of the six month study (post-supplement), only 1/46 (2.2%) patient remained with high HR (p<0.001). None of the patients presented with low HR. The remaining patients (40/46, 87%) presented with normal HRs upon enrollment and 39/46 (84.8%) patients ended the study with normal HRs, the one patient whose HR became abnormal ended with a high HR. In total 6/46 (13%) started with high HR and 2/46 (4%) ended with high HRs.
Pre-supplement, there were 27/46 (58.7%) patients who demonstrated systolic BPs greater than 130 mmHg. Post-supplement, there were 4/46 (8.7%) patients who remained with high systolic BP (p<0.001). Of the remaining 19/46 (41.3%) patients, all with normal systolic BPs pre-supplement, only 1 ended the study with high BP (p < 0.001). Pre-supplement, there were 6/46 (13.0%) patients who demonstrated diastolic BPs greater than 90 mmHg. Post-supplement, no (0/46) patient remained with high diastolic BP (p<0.001). No patient demonstrated low BP Resting P&S Analysis.
From an analysis of absolute resting P&S activity, 10/46 (21.7%) of the patients demonstrated abnormally low resting S-activity (measured as LFa < 0.5 bpm2) prior to administration of the supplement [1]. Low resting S-activity contributes to the condition known as Advanced Autonomic Dysfunction or Diabetic Autonomic Neuropathy, if diagnosed with diabetes. Post-supplement, the number of patients who remained with low S-activity was 4/46 (8.7%, p=0.057). Three (3/46 or 6.5%) of the patients who started the study with resting S-levels within normal limits, declined to abnormally low levels by the end of the study; however, all three of these patients started in the borderline low range of resting S-activity.
From a similar analysis of resting P-activity, 19/46 (43.5%) of the patients demonstrated abnormally low resting P-activity (measured as 0.1 ≤ RFa < 0.5 bpm2) prior to administration of the supplement [1]. Low resting P-activity also contributes to Advanced Autonomic Dysfunction or Diabetic Autonomic Neuropathy. In addition, 2/46 (4.3%) of the patients demonstrated very low resting P-activity (measured as RFa < 0.1 bpm2) prior to administration of the supplement. Very low resting P-activity defines the condition known as Cardiovascular Autonomic Neuropathy (CAN) [1]. Post-supplement, the number of patients who remained with low P-activity was 4/46 (8.7%, p=0.045). None of the patients who started the study with resting P-levels within normal limits, declined to abnormally low levels by the end of the study. One patient who started the study with low, resting P-levels declined further, but not to very low levels. Both patients who demonstrated very low resting P-activity pre-supplement improved to low P-activity by the end of the study. While this is seems to be a reversal of the aging process (CAN, which is accelerated by chronic disease), it merely means that the initial indication of CAN was only “Functional CAN,” not “Structural CAN.” Functional CAN is a condition that mimics the symptoms of CAN, but is not permanent and it is due to prolonged P&S imbalance. Functional CAN is relieved by normalizing SB.
These resting changes P&S had a significant impact on (resting) Sympathovagal Balance (SB). Low SB (SB < 0.4; indicating a resting PE relative to resting S-activity; e.g., as traditionally demonstrated by patients diagnosed with Depression) was demonstrated by 1/46 (2.2%) of the patients pre-supplement and that patient improved with supplement therapy. The rest of the cohort (45/46, 97.8%) pre- supplement demonstrated SB > 0.4 and all patients’ (46/46, 100%) ended the study with a SB > 0.4. However, pre-supplement, there were 14/46 (30.4%) patients that demonstrated high SB (> 3.0; indicating a resting SE relative to resting P-activity; e.g., (p<0.001). Two of these three patients that ended the study with high SB, the last SB recorded was significantly lower (p=0.009). The one patient who increased their SB, the increase (9.4%) was insignificant. The remainder demonstrated normal SB (a proper balance between P&S) and demonstrated no significant change.
Challenge P&S Analysis
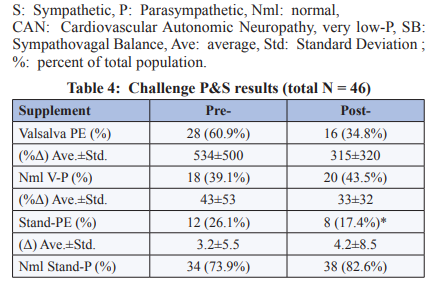
Challenge PE is PE in response to stress, a Sympathetic stimulus. Typically, in response to a Sympathetic stress, P-activity first decreases and then S-activity increases (as an example of these responses and a proper coordination between the P&S nervous systems, see the Normal plot in Figure 4). During the P&S test, challenge PE may occur in response to either Valsalva or stand. Pre-supplement, Valsalva PE (an increase in P-activity in response to Valsalva > 600%) was demonstrated in 28/46 (60.9%) patients. Post-Supplement, 16/46 (34.8%) patients remained with Valsalva PE (p<0.001). Of the 18/46 (39.1%) patients that demonstrated P-responses to Valsalva challenge within normal limits pre- supplement, 4/46 (17.4%) completed the study with Valsalva PE.
From an analysis of stand challenge PE (see the plots with Vagal Excitation in Figure 4), pre-supplement stand PE (an increase in P-activity in response to stand > 10%) was demonstrated in 12/46 (26.1%) of the patients. Seven of these patients (7/12, 58.3%) also demonstrated Valsalva PE pre-supplement. Post-Supplement, no patient remained with stand PE (p<0.001). Of the 34/46 (73.9%) patients that demonstrated P-responses to stand challenge within normal limits, pre-supplement, 8/46 (17.4%) completed the study with stand PE (p<0.001). Only 1/8 of these patients started the study with PE. None of these patients (0/8) demonstrated Valsalva PE, post-supplement.
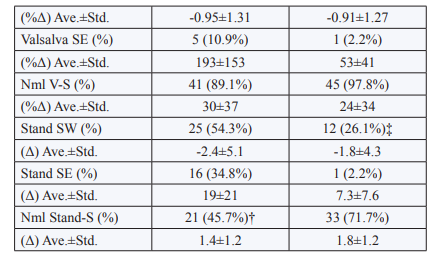
Overall analysis of PE (whether from Valsalva or stand), indicates no significant correlation between amplitude of PE and severity of symptoms, including: difficult to control BP, blood glucose, or hormone level, difficult to describe pain syndromes (including CRPS and Fibromyalgia), difficult to manage weight-loss, unexplained arrhythmia (palpitations) or seizure, and symptoms of depression or anxiety, fatigue, exercise intolerance, sex dysfunction, sleep or GI disturbance, lightheadedness, cognitive dysfunction or “brain fog”, or frequent headache or migraine. Analyzing Valsalva challenge SE, 5/46 (10.9%) patients presented with Valsalva SE pre-supplement, and all but one were relieved post-supplement (p<0.001). Of the 41/46 (89%) that began the study (pre-supplement) with S-responses to Valsalva within normal limits, only one completed the study (post-supplement) with Valsalva SE.
From the analysis of the stand S-response, SW (a S-response to stand that is less than a 10% increase, including any decrease; see the plots with Sympathetic Withdrawal in Figure 4) was demonstrated by 25/46 (54.3%) patients, pre-supplement. Post-supplement, 16/25 (64%) demonstrated a reversal (normalization) of SW (p=0.022). Of the 21/46 (45.7%) patients that demonstrated an S-response to stand within normal limits, pre-supplement, 4/21 (19.0%) demonstrated SW, post-supplement (p=0.003). Stand PE is known to mask SW. Of the 12/46 (26.1%) of the patients that demonstrated stand PE, 9/12 (75.0%) demonstrate normal S-responses to stand, but then demonstrated SW with relief of stand PE, post-supplement; indicating that SW was masked by PE in these patients. Another 2/12 (16.7%) who did not demonstrate SW pre-supplement, but demonstrated stand PE, also demonstrated SW with relief of stand PE, post-supplement. Similar to PE, analyses of SW also indicate no significant correlation between amplitude of SW and severity of symptoms (e.g., lightheadedness, headache or migraine, sleep dysfunction, another other symptoms associated with poor coronary or brain perfusion).
It has been documented that SW precedes an abnormal BP response to stand, and relieve of SW precedes relief of abnormal BP responses to stand [1]. Considering the systolic BP (sBP) response to stand, with respect to the S-response to stand, of the 25/46 (54.3%) patients that demonstrated SW, pre-supplement, 15/25 (60.0%) also demonstrated an abnormal sBP response to stand (less than a 10% increase over resting baseline, p=0.035). Of the 21/46 (45.7%) patients that demonstrated normal S-responses to stand, pre-supplement, 4/21 (19.0%) also demonstrated an abnormal sBP response to stand (p=0.019). Considering the diastolic BP (dBP) response to stand, with respect to the S-response to stand, of the 25/46 (54.3%) patients that demonstrated SW, pre-supplement, 11/25 (44.0%) also demonstrated an abnormal dBP response to stand (less than a 10% increase over resting baseline, p=0.031). Of the 21/46 (45.7%) patients that demonstrated normal S-responses to stand, pre-supplement, 2/21 (9.5%) also demonstrated an abnormal sBP response to stand (p=0.008).
Autonomically, Orthostatic Intolerance (OI, an abnormal BP response to standing) is defined by SW. OI may also be due to vascular damage (i.e., damage to the valves or the smooth muscles in the walls, blood clots, or calcification of the walls). All the above may lead to OI. OI may lead to Neurogenic Orthostatic Hypotension (NOH) or Postural Orthostatic Tachycardia Syndrome (POTS). From BP analysis, 25/46 (54.3%) patients demonstrate OI. Of the pre-supplement subpopulation that demonstrated SW, 18/25 (72%) also demonstrated OI, all of the stand PE patients demonstrated OI, and 8/21 (38.1%) of the non-SW patients demonstrated OI. Post- supplement, 13/46 (28.3%) continued to demonstrate OI, including all of the patients whose SW (8/25) was not relieved.
While no OI patient demonstrated NOH (> 20/10 mmHg drop in BP upon standing), 12/25 demonstrate pre-clinical NOH (> 5 mmHg drop in BP upon standing). They presented with an average 8.3 mmHg drop in standing BP at baseline and ended the study with an average 5.7 mmHg increase in standing BP, an average overall, significant change (increase) of 11.9 mmHg. This improvement was accompanied by an average 7.4 mmHg drop in resting sBP throughout the study, from an average of 135.6 mmHg at baseline to an average of 128.2 mmHg at the conclusion of the study (p = 0.04). The non-OI sub-population (14 or 31.1%) in comparison, presented with an average 5.57 mmHg increase in standing BP at baseline and ended the study with an average 9.64 mmHg increase in standing BP, an average overall change of 4.07 mmHg increase (p > 0.10, not significant). These changes were accompanied by virtually no change to the average resting sBP throughout the study. Average resting sBP increased from an average of 125.0 mmHg at baseline to an average of 125.6 mmHg at the conclusion of the study.
Of this cohort, 9 patients (20% of the total or 29% of the OI patients) presented with diagnoses of POTS, based on history of excessive HR-responses to stand or tilt. These include the 3 patients from above, whose SW was masked by stand PE. Of this POTS sub- population: 5 (55.5%) patients also demonstrated OI symptoms and 4 did not; 6 (66.7%) patients demonstrated SW and 3 (33.3%) did not. Only 1 (11.1%) patient did not demonstrate SW. Of the POTS patients, 2 (22.2%) patients also demonstrated stand SE and were diagnosed with Vasovagal Syncope (as confirmed by P&S Monitoring). Another 2 (22.2%) of the POTS patients also demonstrated stand SE but were not diagnosed with Syncope. One of the POTS patients was diagnosed with Neurocardiogenic Syncope but that was not confirmed with stand SE by P&S Monitoring. Rather this patient was diagnosed with only SW. As per P&S Monitoring, it seemed to be a misdiagnosis. Another POTS patient presented with both Syncope (instantaneous stand SE) and POTS (SW). Three (33.3%) of the POTS patients were diagnosed solely on the basis of excessive HR response to stand (HR > 120bpm, with no diagnosis of Syncope or stand SE).
Of the entire cohort, 27 patients presented with diagnosis or P&S indications of Syncope, pre-supplement. Note, stand SE (a β-adrenergic response) and stand SW (a α-adrenergic response) may occur simultaneously in some patients. Of these 27: 7/46 (15.2%) patients were diagnosed with Syncope and confirmed with SE by P&S Monitoring, 11/46 (39.3%) patients were diagnosed with Syncope but not confirmed with stand SE by P&S Monitoring, and 9/46 (19.6%) patients were not diagnosed with Syncope but were found with stand SE (considered pre-Syncope) by P&S Monitoring. Of the 11 patients who were diagnosed with Syncope, but not confirmed by SE, 3 patients demonstrated SW and 8 patients demonstrated PE.
Table 1: Cohort demographic table (total N = 46)
|
Primary Diagnosis |
DM2 |
HTN |
CAD |
CHF |
Arr |
Sync |
TOTAL |
|
Female [N(%)] |
7 (15.2%) |
16 (34.8%) |
6 (13.0%) |
1 (2.2%) |
8 (17.4%) |
10 (21.7%) |
24 (52.2%) |
|
Male [N(%)] |
5 (10.9%) |
11 (23.9%) |
4 (8.7%) |
1 (2.2%) |
6 (13.0%) |
8 (17.4%) |
22 (47.8%) |
|
Total [N(%)] |
12 (26.1%) |
27 (58.7%) |
10 (21.7%) |
2 (4.3%) |
14 (30.4%) |
18 (39.1%) |
46 (100%) |
%: Percent of total cohort; Arr: Arrhythmia, atrial and ventricular combined, Sync: Syncope.
See text for the other abbreviations.
Quality of Life (QoL) Questionnaire Analysis
The average results of the three SF-36 Questionnaires are: Pre- Supplement) 46.18, and Post-Supplement) 50.59. The indicated QoL results are mediocre. The average overall results of the three Nottingham Health Profiles are: Pre-Supplement) 0.29, and Post- Supplement) 0.27. Again, the indicated overall results are mediocre. These mediocre results are due to the diversity associated with autonomic dysfunction. However, the Nottingham offers a deeper look into more specific wellness issues.
Additional analysis from the Nottingham Profile shows that reported energy levels improved from 0.52 (pre-Supplement), to 0.36 (post- Supplement); a net significant improvement of 30.8%. Reported emotional reactions improved from 0.25 (pre-Supplement) to 0.17 (post-Supplement); a net significant improvement of 32.0%. Reported pain levels improved from 0.38 (pre-Supplement) to 0.21(post-Supplement); a net significant improvement of 44.7%. Reported physical mobility improved from 0.31 (pre-Supplement) to 0.24 (post-Supplement); a net significant improvement of 22.6%. Reported restorative sleep improved from 0.29 (pre-Supplement) to 0.15 (post-Supplement); a net significant improvement of 48.3%. Reported social isolation improved from 0.11 (pre-Supplement) to 0.07 (post-Supplement); a net significant improvement of 36.4%. While the overall improvement is only 6.9%, the improvements of the more specific indices were over three time’s higher (p < 0.10).
Discussion
The antioxidants (Alpha-Lipoic Acid and CoQ10) used in this study supplement natural, common factors in the human body. The supplements had a positive effect on the P&S nervous systems as measured directly and indicated by improvements in BP, HR, and QoL questionnaires. Both are well known to help restore health and wellness, by normalizing fundamental cellular functions, including mitochondrial health. In normalizing these functions, these supplements work with the body to naturally help to raise or lower autonomic and cardiovascular functions as needed to restore the individual patient’s health.
Hemodynamic dynamic activity (e.g., HR & BP) is driven by P&S activity, both: 1) directly, and 2) indirectly through hormones and secondary messengers. Atherosclerosis and other disorders may also affect HR & BP. These antioxidants help to reduce the risk and presence of atherosclerosis by preventing LDLs from oxidizing, thereby restoring more normal hemodynamic activity. Normalizing P&S balance also normalizes hemodynamic activity. Normalizing resting, elevated P-activity helps to raise HR & BP. Normalizing resting, elevated S-activity helps to lower HR & BP. These are the reasons why the effects of the supplements used in this study are not homogeneous. Whether the supplements increase or decrease the measured factors depends on the individual patient and that patient’s history. These supplements have either effect on any given patient, and therefore help to promote, not only, health but wellness in patients with varied histories.
Since the P&S nervous systems control or coordinate virtually every cell in the body, the signs of autonomic dysfunction are many and varied. They may include: lightheadedness; orthostatic dysfunction; syncope or pre-syncope; fatigue; brain fog; difficulty finding words; short term memory loss; hypersensitivity to light, sound, motion, or touch; neuropathy or paresthesia in extremities; cold extremities; coat hanger pain; tension headaches; migraine; nausea/vomiting; chest pain; palpitations; dyspnea; hypermobility; depression; anxiety; hyperhidrosis or anhydrosis; dry mouth; dimmed vision; tinnitus or dimmed hearing; and intolerance to hot or cold weather.
P&S Monitoring, while based on traditional non-invasive, beat- to-beat, cardiac measures (HR or BP, in this case HR variability), the cardiac measures are not the whole story. Traditional beat- to-beat cardiac measures are measures of only total autonomic function. They fail to satisfy fundamental mathematical requirements for fully characterizing a system with two independent parts: the Parasympathetic and Sympathetic nervous systems. Do to this failing, they force assumption and approximation to theorize P&S activity. As an example, the parameter typically assumed to be a measure of Sympathetic activity, LF, is actually a mixture of both Sympathetic and Parasympathetic activity; according to the HR Variability Standards articles [73, 74]. The typical assumption, which is typically buried in the patient population studies, is that the Parasympathetics are extremely weak as compared to the Sympathetics. This may be true in advanced disease states such as Diabetes, Hypertension, CAD, CHF, post-MI, post-stroke, chronic pain, and other long-term chronic patients. However, once that patient is medicated, typically with more than one sympatholytic, the assumption is no longer valid; therefore, what is being measured? This is a resting example.
The same Standards articles, state that the traditional HRV ratio of the Sympathetic measure, LF, to Parasympathetic measure, HF, (LF/ HF) is a better measure of a patient’s response to head-up postural change (stand, a net Sympathetic stimulus) than LF alone. This in part confirms that LF includes Parasympathetic activity (because it is being divided out, or cancelled by taking the ratio) [73, 74]. However, this is still based on approximation. The HF measure is not only of Parasympathetic activity, it by its definition [73, 74] includes noise. All of this to state that the addition of the respiratory activity analysis to the HRV analysis in P&S Monitoring, resolves this failing by completely satisfying the mathematical requirements of two independent measures (measures of the heart and lungs in this case) to fully characterize an system with two independent components. As a result, the P&S parameters (RFa and LFa, respectively), as well as Sympathovagal Balance (LFa/RFa), are more precise and specific measures of autonomic function. The result, for example, is a monitor that is able to fully characterize and differentiate the head-up, postural change (stand) challenge, including: α-SW (indicating possible orthostatic dysfunction), β-SE (indicating possible Syncope), and PE (contributing to lightheadedness and the more difficult to manage type patient , and with stand, indicating possible Vasovagal Syncope).
It should be noted that changes to the P&S nervous systems must be small and consistent over a relatively long period of time so as to prevent the creation of added co-morbidities or “side-effects.” P&S changes are foundational. They change how the organ is controlled, and are required for long lasting changes. Any symptoms that remain after P&S changes tend to be structural organ changes and may be permanent, therefore requiring long term therapy. Non-structural organ changes are relieved over time once the P&S changes are effected. The P&S changes tend to require two to three months depending on age or length of disease. Then the symptomatic changes from end-organ changes tend to require another two to three months.
These supplements also affect challenge responses as well. Challenge responses typically become abnormal before resting responses. Since most physicians assess patients only at rest (sitting or supine), many causes of symptoms, especially early symptoms, are not assessed. Abnormal challenge responses are associated with increased morbidity risk. Abnormal resting responses are associated with increased mortality risk. Abnormal challenge responses without abnormal resting responses are often associated with more acute conditions, whereas the presence of abnormal resting responses is typically associated with chronic conditions.
The abnormal challenge PE response with Valsalva is often associated with an abnormal decreased BP response to Valsalva as compared with Rest. This is an indication of Baroreceptor reflex dysfunction. Note, decreases in HR have not been observed with abnormal Valsalva responses, perhaps due to the inflated S-response to Valsalva secondary to Valsalva PE.
Valsalva Challenge SE is associated with stress, hypertension, risk of cerebrovascular accident, myocardial infarction, or other MACE. Valsalva SE tends to be an earlier indicator than high SB or CAN. Valsalva SE may present with symptoms of high stress (a SE response to lifestyle) or symptoms of extreme fatigue (a consequence of prolonged SE). This is a consequence of over- reacting to otherwise normal stimuli due to inflated S-responses (and therefore high BP or HR responses) to PE. As a result, these normal stimuli are perceived as stressful.
Sympathetic responses to stand help to differentiate causes of lightheadedness. Orthostatic Dysfunction is based on S-Withdrawal (SW) and includes Orthostatic intolerance (OI: SW with a normal BP response), Orthostatic Hypotension (OH: SW with a drop in BP of > 20 mmHg systolic or > 10 mmHg diastolic; pre-clinical OH is SW with a drop in BP of > 5 mmHg systolic or > 0 mmHg diastolic), and Postural Orthostatic Tachycardia Syndrome (POTS: SW with a standing HR >120 bpm or an increase in HR upon standing of >20% in the absence of OH drop in BP). Again, Orthostatic Dysfunction is an alpha-S, peripheral vascular response, and therefore may present with syncope (a beta-S response, typically demonstrated as an instantaneous SE). Also note, while SW indicates risk of Orthostatic Dysfunction, Orthostatic Dysfunction doesn’t always include SW, and SW may precede symptoms of Orthostatic Dysfunction (including falls). Orthostatic Dysfunction may also be due to peripheral vascular (wall and valve) dysfunction.
Another abnormal S-response to stand is SE. SE may result in pre-clinical or clinical Syncope. This includes Vasovagal Syncope (stand SE with PE at some time during the clinical exam) and Neurogenic Syncope (stand SE with an abnormal HR-response to stand, typically little or no increase in HR). Cardiogenic Syncope is a diagnosis by omission. This formula helps to further differentiate Neurocardiogenic Syncope into its components. Again, syncope is a beta-S, cardiac response, and therefore may present with OI, an alpha-S response.
As is typical of autonomic management and seen in the results of the questionnaires, the initial P&S responses to therapy are often an initial decrease or an apparent decrease in QoL followed by an increase in the results indicating an apparent improvement in QoL. Again, this is not unusual. As we tell our patients, oftentimes patients feel worse before feeling better because we have to work to repair the “foundation” before the rest of the “structure” may be improved. Further, the decline is due to the fact that latent symptoms are often unmasked and must also be addressed before improvement in QoL may be enjoyed. The overall results of both questionnaires are mediocre. This is due to the diversity of symptoms and conditions associated with autonomic dysfunction; a highly individualistic disorder. From the deeper look into more specific wellness issues through the Nottingham questionnaire significant (net) improvements are recognized.
This QoL questionnaire analysis highlights the power of P&S monitoring, and the individuality of the P&S nervous systems. The power of P&S Monitoring is that it measures the individual patients own physiology, based on their individual history and condition. The individuality of the P&S Nervous System from patient to patient is that it remembers the individual patient’s own unique history and, as a result, tends to present with symptoms that are unique to the individual patient.
Study Limitations
This study is an observational, single center analysis of quantitative autonomic (P&S) testing function data after supplement administration over a period of six months. The sample size is small but the statistical significance was quite dramatic. Observational studies, however, include a potential selection bias. Furthermore, it is important to stress that the absence of a control group should be considered before concluding that the combination of antioxidant supplements in this study is superior to other treatment options. This could be done in a perspective randomized trial comparing supplement treated patients with controls who are in a double- blinded fashion, and a third group with an alternative therapy could be employed. The results of this study could be considered hypothesis-generated, but in view of the vast literature supporting their use, it is reasonable to assume beneficial parameters will be reproduced in other populations.
As the patients did not provide pill counts, it is possible that there was some degree of noncompliance in certain patients and not in others. It is possible that there was a dosage effect with results. For example, patients who were 100% compliant may have had better results than patients who were less than 100% compliant with daily intake of the supplements. In addition, the supplement dosages (200mg, bid ALA & 100mg bid CoQ10) were extremely low compared to other clinical studies in the literature, which for example, have used up to 1200 and 1800 mg of ALA. Future studies with higher dosages may demonstrate more dramatic and statistically significant results. Furthermore, both antioxidants (ALA & CoQ10) are naturally produced by the human body, and that production declines with age and duration of disease. The levels of the experimental supplements were not controlled during the test and may have biased the results to the younger patients.
In this study, each patient served as their own control. The possible selection bias, for example, may have included the fact that the 46/51 patients who were enrolled in the study and completed the study may have also adhered to better lifestyle changes, such as diet and exercise, which could not be controlled in this study. This would have additionally benefited autonomic function results. In addition, quality of life questionnaires are subjective and individuals undergoing active treatment without a double-blinded control group have an inherent bias toward answering questions in a positive fashion, and there was no comparison to be made with the control group in this study. Future studies with controlled groups should be done.
Due to the non-controlled, non-randomized nature of this study, including analysis and results, the observational nature of the study and the fact that there were no pre-established primary and secondary endpoints, future studies should target these goals.
As mentioned, as in any prevention study we cannot rule out healthy user bias [75]. Other healthy activities, such as exercise and use of a healthy diet, including other antioxidants, supplemental vitamins (including Biotin), may have contributed to beneficial results in autonomic parameters measured. Also, it should be noted that the study population was from a single medical center which involves subjects with comprehensive access to medical care, which may limit the generalizing to other localities.
Conclusion
This trial demonstrates the combined effect of the antioxidants ALA and CoQ10 on the P&S Nervous Systems. The supplements had a positive effect on the P&S nervous systems as measured directly and also indicated by improvements in BP, HR and reported QoL. The study indicates these supplements help to improve both the resting and challenge P&S responses and resultant physiology. In fact, it suggests a possible complimentary therapy in patients with poorly controlled BP (i.e., hypertension and possible hypotension) as well as in patients with tachycardia or palpitations. This is a hypothesis generating study of significant importance in an often neglected area of cardiovascular disease in which additional research and studies are needed.
References
- Colombo J, Arora RR, DePace NL, Vinik AI (2014) Clinical Autonomic Dysfunction: Measurement, Indications, Therapies, and Outcomes. Springer Science + Business Media.
- DePace NL, Colombo J (2019) Clinical Autonomic and Mitochondrial Disorders – Diagnosis, Prevention, and Treatment for Mind-Body Wellness. Springer Science + Business Media.
- Ziegler D, Ametov A, Barinov A, Dyck PJ, Gurieva I, et al. (2006) Oral treatment with Alpha-lipoic Acid improves symptomatic diabetic polyneuropathy: The SYDNEY 2 trial. Diabetes Care 29: 2365-2370.
- Ametov AS, Barinov A, Dyck PJ, Hermann R, Kozlova N, et al. (2003) The sensory symptoms of diabetic polyneuropathy are improved with alpha-lipoic acid: The SYDNEY trial. Diabetes Care 26: 770-776.
- Ziegler D, Low PA, Litchy WJ, Boulton AJM, Vinik AI, et al. (2011) Efficacy and safety of antioxidant treatment with α-lipoic acid over 4 years in diabetic polyneuropathy: the NATHAN 1 Trial Group . Diabetes Care 34: 2054-2060.
- Gao L, Mao Q, Cao J, Wang Y, Zhou X, et al. (2012) Effects of coenzyme Q10 on vascular endothelial function in humans: a meta-analysis of randomized controlled trials. Atherosclerosis 221: 311-316.
- Rosenfeldt F, Marasco S, Lyon W, Wowk M, Sheeran F, et al. (2005) Coenzyme Q10 therapy before cardiac surgery improves mitochondrial function and in vitro contractility of myocardial tissue. J Thorac Cardiovasc Surg 129: 25-32.
- Folkers K, Littarru GP, Ho L, Runge TM, Havanonda S, et al. (1970) Evidence for a deficiency of coenzyme Q10 in human heart disease. Int Z Vitaminforsch 40: 380-390.
- Folkers K, Vadhanavikit S, Mortensen SA (1985) Biochemical rationale and myocardial tissue data on the effective therapy of cardiomyopathy with coenzyme Q10. Proc Natl Acad Sci 82: 901-904.
- Mortensen SA (2003) Overview on coenzyme Q10 as adjunctive therapy in chronic heart failure. Rationale, design and end- points of “Q-symbio”--a multinational trial. Biofactors 18: 79-89.
- Rosenfeldt F, Hilton D, Pepe S, Krum H (2003) Systematic review of effect of coenzyme Q10 in physical exercise, hypertension and heart failure. Biofactors 18: 91-100.
- Widmer RJ, Flammer AJ, Lerman LO, Lerman A (2015) The Mediterranean diet, its components, and cardiovascular disease. Am J Med 128: 229-238.
- Liyanage T, Ninomiya T, Wang A, Neal B, Jun M, et al. (2016) Effects of the Mediterranean Diet on Cardiovascular Outcomes-A Systematic Review and Meta-Analysis. PLoS One 11: e0159252.
- Grosso G, Marventano S, Yang J, Micek A, Pajak A, et al. (2017) A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: Are individual components equal? Crit Rev Food Sci Nutr 57: 3218-3232.
- Nordmann AJ, Suter-Zimmermann K, Bucher HC, Shai I, Tuttle KR, et al. (2011) Meta-analysis comparing Mediterranean to low-fat diets for modification of cardiovascular risk factors. Am J Med 124: 841-851.
- Satija A, Bhupathiraju SN, Spiegelman D, Chiuve SE, Manson JE, et al. (2017) Healthful and Unhealthful Plant-Based Diets and the Risk of Coronary Heart Disease in U.S. Adults. J Am Coll Cardiol 70: 411-422.
- Ros E, Martínez-González MA, Estruch R, Salas-Salvadó J, Fitó M, et al. (2014) Mediterranean diet and cardiovascular health: Teachings of the PREDIMED study. Adv Nutr 5: 330-336.
- Estruch R, Ros E, Salas-Salvadó J, Covas MI, Corella D, et al. (2013) Primary prevention of cardiovascular disease with a Mediterranean diet. N Engl J Med 368: 1279-1290.
- Khera AV, Emdin CA, Drake I, Natarajan P, Bick AG, et al. (2016) Genetic Risk, Adherence to a Healthy Lifestyle, and Coronary Disease. N Engl J Med 375: 2349-2358.
- Fuentes F, López-Miranda J, Sánchez E, Sánchez F, Paez J, et al. (2001) Mediterranean and low-fat diets improve endothelial function in hypercholesterolemic men. Ann Intern Med 134: 1115-1119.
- Ludwig DS (2016) Lowering the Bar on the Low-Fat Diet. JAMA 316: 2087-2088
- Nissensohn M, Román-Viñas B, Sánchez-Villegas A, Piscopo S, Serra-Majem L, et al. (2016) The Effect of the Mediterranean Diet on Hypertension: A Systematic Review and Meta-Analysis. J Nutr Educ Behav 48: 42-53.
- Wang Q, Afshin A, Yakoob MY, Singh GM, Rehm CD, et al. (2016) Impact of Nonoptimal Intakes of Saturated, Polyunsaturated, and Trans Fat on Global Burdens of Coronary Heart Disease. J Am Heart Assoc 5: 1.
- Kris-Etherton P, Eckel RH, Howard BV, St Jeor S, Bazzarre TL, et al. (2001) Nutrition Committee Population Science Committee and Clinical Science Committee of the American Heart Association. AHA Science Advisory: Lyon Diet Heart Study. Benefits of a Mediterranean-style, National Cholesterol Education Program/American Heart Association Step I Dietary Pattern on Cardiovascular Disease. Circulation. 103: 1823-1825.
- Estruch R, Ros E, Salas-Salvadó J, Covas MI, Corella D, et al. (2013) PREDIMED Study Investigators Primary prevention of cardiovascular disease with a Mediterranean diet. N Engl J Med 368: 1279-1290.
- Gay HC, Rao SG, Vaccarino V, Ali MK (2016) Effects of Different Dietary Interventions on Blood Pressure: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Hypertension 67: 733-739.
- Bloomfield HE, Koeller E, Greer N, MacDonald R, Kane R, et al. (2016) Effects on Health Outcomes of a Mediterranean Diet with No Restriction on Fat Intake: A Systematic Review and Meta-analysis. Ann Intern Med 165: 491-500.
- Sofi F, Cesari F, Abbate R, Gensini GF, Casini A, et al. (2008) Adherence to Mediterranean diet and health status: meta- analysis. BMJ 337: 1344.
- Esposito K, Maiorino MI, Bellastella G, Chiodini P, Panagiotakos D, et al. (2015) A journey into a Mediterranean diet and type 2 diabetes: a systematic review with meta-analyses. BMJ Open 5: e008222.
- Ros E, Hu FB (2013) Consumption of plant seeds and cardiovascular health: Epidemiologic and clinical trial evidence. Circulation 128: 553-565.
- Berkson B (1998) Alpha Lipoic Acid Breakthrough: The Superb Antioxidant That May Slow Aging, Repair Liver Damage, and Reduce the Risk of Cancer, Heart Disease, and Diabetes. Kindle Edition by Burt Berkson.
- Jacob S, Streeper RS, Fogt DL, Hokama JY, Tritschler HJ, et al. (1996) The antioxidant alpha-lipoic acid enhances insulin- stimulated glucose metabolism in insulin-resistant rat skeletal muscle. Diabetes 45: 1024-1029.
- Ziegler D, Gries F (1997) Alpha-lipoic Acid and the treatment of diabetic peripheral autonomic cardiac neuropathy. Diabetes 46: 62-66.
- Prendergast JJ (2001) Diabetic autonomic neuropathy: Part 1.Early detection. Practical Diabetology 2001: 7-14.
- Prendergast JJ (2001) Diabetic autonomic neuropathy: Part 2.Treatment. Practical Diabetology 2001: 30-36.
- Manning PJ, Sutherland WHF, Walker RJ, Williams SM, De Jong SA, et al. (2004) Effect of high-dose Vitamin E on insulin resistance and associated parameters in overweight subjects. Diabetes Care 27: 2166-2171.
- Rudich A, Tirosh A, Potashnik R, Khamaisi M, Bashan N, et al. (1999) Lipoic acid protects against oxidative stress induced impairment in insulin stimulation of protein kinase B and glucose transport in 3T3-L1 adipocytes. Diabetologia 42: 949-957.
- Maddux BA, See W, Lawrence JC, Goldfine AL, Goldfine ID, et al. (2001) Protection against oxidative stress-induced insulin resistance in rat L6 muscle cells by micromolar concentrations of alpha-lipoic acid. Diabetes 50: 404-410.
- Jungas T, Motta I, Duffieux F, Fanen P, Stoven V, et al. (2002) Glutathione levels and BAX activation during apoptosis due to oxidative stress in cells expressing wild-type and mutant cystic fibrosis transmembrane conductance regulator. J Biol Chem 277: 27912-27918.
- Dominy JE, Hwang J, Stipanuk MH (2007) Overexpression of cysteine dioxygenase reduces intracellular cysteine and glutathione pools in HepG2/C3A cells. Am J Physiol Endocrinol Metab 293: 62-69.
- Ziegler D, Gries F (1997) Alpha-lipoic Acid and the treatment of diabetic peripheral autonomic cardiac neuropathy. Diabetes 46: 62-66.
- Ziegler D, Ametov A, Barinov A, Dyck PJ, Gurieva I, et al. (2006) Oral treatment with Alpha-lipoic Acid improves symptomatic diabetic polyneuropathy: The SYDNEY 2 trial. Diabetes Care 29: 2365-2370.
- Ametov AS, Barinov A, Dyck PJ, Hermann R, Kozlova N, et al. (2003) The sensory symptoms of diabetic polyneuropathy are improved with alpha-lipoic acid. The SYDNEY trial. Diabetes Care 26: 770-776.
- Ziegler D, Low PA, Litchy WJ, Boulton AJM, Vinik AI, et al. (2011) Efficacy and safety of antioxidant treatment with α-lipoic acid over 4 years in diabetic polyneuropathy. Diabetes Care 34: 2054-2060.
- Ali AM, Awad TG, Al-Adl NM (2010) Efficacy of combined topiramate/thioctic acid therapy in migraine prophylaxis. Saudi Pharm J 18: 239-243.
- Mahboob A, Farhat SM, Iqbal G, Babar MM, Zaidi NU, etal. (2016) Alpha-lipoic acid-mediated activation of muscarinic receptors improves hippocampus- and amygdala-dependent memory. Brain Res Bull 122: 19-28
- Kuhla A, Derbenev M, Shih HY, Vollmar B (2016) Prophylactic and abundant intake of α-lipoic acid causes hepatic steatosis and should be reconsidered in usage as an anti-aging drug. Biofactors 42: 179-189.
- Logan AC, Wong C (2001) Chronic fatigue syndrome: oxidative stress and dietary modifications. Altern Med Rev 6: 450-459.
- Hiller S, DeKroon R, Hamlett ED, Xu L, Osorio C, et al. (2016) Alpha-lipoic acid supplementation protects enzymes from damage by nitrosative and oxidative stress. Biochim Biophys Acta 1860: 36-45.
- Wollen KA (2010) Alzheimer’s disease: the pros and cons of pharmaceutical, nutritional, botanical, and stimulatory therapies, with a discussion of treatment strategies from the perspective of patients and practitioners. Altern Med Rev 15: 223-244.
- Ajith TA, Padmajanair G (2015) Mitochondrial Pharmaceutics: A New Therapeutic Strategy to Ameliorate Oxidative Stress in Alzheimer’s Disease. Curr aging Sci 8: 235-240.
- Li YH, He Q, Yu JZ, Liu CY, Feng L, et al. (2015) Lipoic acid protects dopaminergic neurons in LPS-induced Parkinson’s disease model. Metab Brain Dis 30: 1217-1226.
- Zhao H, Zhao X, Liu L, Zhang H, Xuan M, et al. (2017) Neurochemical effects of the R form of α-lipoic acid and its neuroprotective mechanism in cellular models of Parkinson’s disease. Int J Biochem Cell Biol 87: 86-94.
- Zhao H, Zhao X, Liu L, Zhang H, Xuan M, et al. (2017) Neurochemical effects of the R form of α-lipoic acid and itsneuroprotective mechanism in cellular models of Parkinson’sdisease. Int J Biochem Cell Biol 87: 86-94.
- Namazi N, Larijani B, Azadbakht L (2018) Alpha-lipoic acid supplement in obesity treatment: A systematic review and meta- analysis of clinical trials. Clin Nutr 37: 419-428.
- Murray GL, Colombo J (2019) (R) alpha lipoic acid is a safe, effective pharmacologic therapy of chronic orthostatic hypotension associated with low sympathetic tone. Int J Angiol 28: 188-193.
- Mordente A, Martorana GE, Santini SA, Miggiano GA, Petitti T, et al. (1993) Antioxidant effect of coenzyme Q on hydrogen peroxide-activated myoglobin. Clin Investig 71: 92-96.
- Hargreaves IP (2015) Coenzyme Q10: From Fact to Fiction. Nova Science Publishing Inc 364 pp.
- Zhou M, Zhi Q, Tang Y, Yu D, Han J, et al. (1999) Effects of coenzyme Q10 on myocardial protection during cardiac valve replacement and scavenging free radical activity in vitro. J Cardiovasc Surg (Torino) 40: 355-361.
- Sander S, Coleman CI, Patel AA, Kluger J (2006) White CM. The impact of coenzyme Q10 on systolic function in patients with chronic heart failure. J Card Fail 12: 464-472.
- Kumar A, Singh RB, Saxena M, Niaz MA, Josh SR, et al. (2007) Effect of carni Q-gel (ubiquinol and carnitine) on cytokines in patients with heart failure in the Tishcon study. Acta Cardiol. 62: 349-354.
- Morisco C, Trimarco B, Condorelli M (1993) Effect of coenzyme Q10 therapy in patients with congestive heart failure: a long- term multicenter randomized study. Clin Investig 71: 134-136.
- Matthews RT, Yang L, Browne S, Baik M, Beal MF, et al. (1998) Coenzyme Q10 administration increases brain mitochondrial concentrations and exerts neuroprotective effects. Proc Natl Acad Sci USA 95: 8892-8897.
- Rustin P, von Kleist-Retzow JC, Chantrel-Groussard K, Sidi D, Munnich A, et al. (1999) Efffect of idebenone on cardiomyopathy in Friedreich’s ataxia: a preliminary study. Lancet 354: 477-479.
- Lodi R, Schapira AH, Manners D, Styles P, Wood NW, et al. (2000) Abnormal in vivo skeletal muscle energy metabolism in Huntington’s disease and dentatorubropallidoluysian atrophy. Ann Neurol 48: 72-76.
- Huntington Study Group (2001) A randomized, placebo- controlled trial of coenzyme Q10 and remacemide in Huntington’s disease. Neurology 57: 397-404.
- Bloomfield DM, Kaufman ES, Bigger JT, Fleiss J, Rolnitzky L, et al. (1997) Passive head-up tilt and actively standing up produce similar overall changes in autonomic balance. Am Heart J 134: 316-320.
- Akinola A, Bleasdale Barr K, Everall L, Mathias CJ (1999) Investigation of Autonomic Disorders: Appendix I In: Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System. Oxford Medical Publications, London, England. Mathias CJ and Bannister R (eds).
- Vinik A, Ziegler D (2007) Diabetic cardiovascular autonomicneuropathy. Circulation 115: 387-397.
- Vinik AI, Maser RE, Nakave AA (2007) Diabetic cardiovascularautonomic nerve dysfunction. US Endocrine Disease 2007: 2-9.
- Murray LG and Colombo J. Routine measurements of cardiac parasympathetic and sympathetic nervous systems assists in primary and secondary risk stratification and management of cardiovascular clinic patients (2019) Clinical Cardiol Cardiovascular Med 3: 27-33.
- DePace NL, Bateman JA, Yayac M, Oh J, Siddique M, et al. (2018) Acosta C, Pinales JM, Vinik AI, and Bloom HL. Improved Patient Outcomes by Normalizing Sympathovagal Balance: Differentiating Syncope-Precise Subtype Differentiation Leads to Improved Outcomes. Cardiology Research and Practice 2018: 8.
- Malik M (1996) The Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability, standards of measurement, physiological interpretation, and clinical use. Circulation 93: 1043-1065.
- Malik M (1996) The task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability, standards of measurement, physiological interpretation, and clinical use. European Heart Journal 17: 354-381.
- Shrank WH, Patrick AR, Brookhart MA (2011) Healthy user and related biases in observational studies of preventive interventions: a primer for physicians. J Gen Intern Med 26: 546-550.




