Research Article - (2024) Volume 1, Issue 2
Antioxidant, Antimicrobial and Anti-Inflammatory Activities Development of Crude Extract of Lamium Garganicum Subsp. Longiflorum (Ten) Kerguélen = Lamium Longiflorum Ten
2Laboratory of Phytotherapy Applied to Chronic Diseases, Faculty of Natur and Life Sciences, Ferhat Abbas University, Sétif-1, Algeria
Received Date: Jan 23, 2024 / Accepted Date: Aug 20, 2024 / Published Date: Sep 24, 2024
Copyright: ©�2024 Gaamoune Sofiane, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Sofiane, G., Wafa, N. (2024). Antioxidant, Antimicrobial and Anti-Inflammatory Activities Development of Crude Extract of Lamium Garganicum Subsp. Longiflorum (Ten) Kerguélen = Lamium Longiflorum Ten, Archives Biol Life Sci, 1(2), 1-5.
Abstract
Lamium genus revealed the presence of various compounds such as phenylpropanoids, flavonoids, polysaccharides, tannins, phenolics, triterpene saponins, phytoecdysteroids, iridoids, anthocyanins. Lamium garganicum subsp. longiflorum (Ten.) Kerguélen = Lamium longiflorum Ten., was studies for their biological activities (antioxidant activity using DPPH test, antimicrobial activity and anti-inflammatory activity by human red blood cell (HRBC) membrane stabilization method). The results demonstrate a strong antibacterial power against Staphylococcus aureus methicillin resistant and an interesting anti- inflammatory activity.
Keywords
Lamium, DPPH, MRSA, HRBCIntroduction
The Lamium L. is a genus of the Lamiaceae family. This genus comprises about 40 annual and perennial species, which spread in several areas throughout Asia, Europe, and Africa. Some of these species are well-known, such as L. album, L. purpureum, L. maculatum, and L. garganicum, which are found in the mountains of Southern Europe [1]. According to the World Checklist of Lamiaceae and Verbenaceae [2,3] there are 24 accepted species in the genus as well as several infraspecific taxa. Lamium includes both annual and perennial herbaceous plants that are widely distributed throughout temperate Eurasia [4].
Lamium genus is used in different traditional preparations to treat some disorders: urinary, gastric (diarrhea and constipation), and respiratory (cough, cold). Plus, it is very effective as a topical healing against varicose veins, wounds, and other skin damage [5]. Previous phytochemical investigations of the genus Lamium resulted in the isolation of iridoid glucosides, flavonoids, phenolics, phenylpropanoids, polysaccarides, triterpene saponins, tannins and phytoecdysteroids [6,7].
The results of molecular studies were congruent with the observed morphological variability of pericarp also with respect to the status of L. garganicum subsp. laevigatum. The difference noticed in sculpturing of the fruits of this taxon compared to a second subspecies of L. garganicum is an argument to consider L. garganicum subsp. laevigatum a separate taxon. This result is consistent with the analysis of variation barcoding sequences in the genus Lamium, which also indicated the distinctiveness of this subspecies [8,9].

Botanically, Lamium longiflorum Ten (Lamium garganicum subsp. longiflorum (Ten.) Kerguélen)., is a hairless or nearly hairless plant, with whorls close together. Corolla purpurin very rarely white, hairy on the helmet, 25-30mm long. Calyx with acute lanceolate lobes, mutic. Hairy anthers. This Mediterranean plant prefers Forests, wet ravines [10].
Materials and Methods
Plant Material
The random sampling was used during the harvesting of simples. The aerial parts of Lamium garganicum subsp. longiflorum (Ten.) Kerguélen = Lamium longiflorum Ten., were collected from the mountain of BABOR (36° 30′ 06″ Nord, 5° 28′ 27″ East); Determined by Dr. Nouioua Wafa in Laboratory of National Institute of Agricultural Research – Setif – Algeria.
Preparation of Extract
Plant material was powdered and macerated three times in 80% methanol for 24 hours each, at the laboratory conditions (ratio: 1:10 w/v). The resulted solutions were filtered and evaporated to dryness under vacuum. The dry extract was stored at a temperature of – 18 °C for ulterior use.
Determination of Total Phenolic Content
Total polyphenol dosage was determined by the Foline Ciocalteu method [11]. The samples (0.2 mL) were mixed with 1 mL of the Folin-Ciocalteu reagent previously diluted to 1:10 in deionized water. The solutions were allowed for 4 minutes at 25 °C. Then, 0.2 mL of saturated sodium carbonate solution (75 mg/mL) was added. The mixed solutions were allowed to stand for another 120 minutes before the absorbance was measured at 765 nm. Gallic acid was used as a standard. The total phenolic compounds content was expressed as mg equivalent of Gallic acid per gram of extract (mg EAG/GE).
Determination of Total Flavonoids Content
The flavonoids content in the extract was estimated by the Aluminium chloride solution [12]. To 1 mL of the extract was added to 1 mL of 2 % AlCl3 in methanol. After 10 minutes, the absorbance was determined at 430 nm. Quercetin was used as a standard. Results were expressed as mg equivalent Quercetin per gram of extract (mg EQ/GE).
DPPH Assay
One millilitre of the extract at different concentrations was added to 0.5 mL of DPPH-methanol solution. The mixtures were shaken vigorously and left standing at the laboratory conditions for 30 minutes in the dark. The absorbance was measured at 517 nm. The antiradical activity was expressed as EC50 (micrograms per millilitre). The ability to scavenge the DPPH radical was calculated using the following equation:
DPPH scavenging effect (%) = [(A0 – A1)/ A0] ×100
Where:
A0: the absorbance of the control at 30 min
A1: is the absorbance of the sample at 30 min. Butylated hydroxytoluene (BHT) was used as standard [13].
Antimicrobial Activity
Bacteria Strains were obtained from the American Type Culture Collection: Gram-positive bacteria (Staphylococcus aureus methicillin resistant ATCC 43300 and Bacillus subtilis ATCC6633), Gram-negative bacteria (Escherichia coli Proteus mirabilis ATCC700603) and one yeast: Candida albicans ATCC1024. The Muller Hinton agar was used to bacteria culture and Sabouraud for yeast.
Anti-Bacterial Activity
Agar disc diffusion method was employed to determine the antibacterial activity of the extract [14,15]. Briefly, a suspension of the tested microorganism (108 CFU / mL) was spread on the solid media plates. Filter paper discs (6 mm in diameter) were impregnated with 10 μL (100 mg/mL) of the extract and placed on the inoculated plates. These plates were incubated at 37 °C for 24 hours. Vancomycin (30 μg/disc) and Gentamicin were used as a standards and Dimethylsulfoxide DMSO as a control. The antibacterial activity was determined by measuring inhibition zone diameters (mm) and was evaluated according the parameters suggested by Alves et al. (2000) [33]:
<9 mm, inactive ; 9–
12 mm, less active ;
13–18 mm, active;
>18 mm, very active.
Antifungal Activity
The antifungal activity was tested by disc diffusion method with modifications [14]. The Candida albicans suspension was obtained in physiological saline 0.9 % from a culture in Sabouraud (incubated before 24 hours at 37 °C), adjusted to 105 CFU / mL.
Briefly, 0.1 mL of suspension were placed over the agar in Petri dishes. Then, sterile paper discs (6 mm diameter) were placed on agar to load 10 μL (100 mg/mL) of each sample. Amphotericin 100 µg was used as standard and dimethylsulfoxide DMSO as control. Inhibition zones were determined after incubation at 27 °C for 48 hours.
The Human Red Blood Cell (HRBC) Membrane Stabilization Method
The principle involved here was stabilization of human red blood cell membrane by hypo tonicity induced membrane lysis. The mixture contains 1 mL phosphate buffer (pH 7.4, 0.15 M), 2 mL hypo saline (0.36 %), 0.5 mL HRBC suspension 10 % v/v (prepared by washing erythrocytes three times with normal saline) and 0.5 mL of plant extract or standard drug (Diclofenac sodium) at various concentrations (10, 50, 100, 250, 500 μg/ mL). The control was distilled water instead of hypo saline to produce 100 % haemolysis. The mixtures were incubated at 37 °C for 30 minutes and centrifuged at 2500 rpm for 5 minutes. The absorbance of haemoglobin content in the suspensions was estimated at 560 nm. The percentage of haemolysis of HRBC membrane can be calculated as follows:
Haemolysis (%) = (Optical density of Test sample / Optical density of Control) ×100 However, the percentage of HRBC membrane stabilization can be calculated as follows:
Protection (%) = 100 – [(Optical density of Test sample / Optical density of Control) × 100] [16].
Statistical Analysis
Results were expressed as the mean ± standard deviation. Data was statistically analysed using t test of Student as primary test followed by Fisher test with the criterion of P < 0.001 to determine whether there were any significant differences between methanol extract of Lamium garganicum subsp. longiflorum (Ten.) Kerguélen and standards, using Graphpad prism 8 Demo Software.
Results and Discussion
Crude extract yield, polyphenols dosage and flavonoids quantity were 20.37 %, 10,59±0,13 mg EAG/GE and 9,00±0,20 mg EQ/ GE respectively.
DPPH bioassay has been widely used to assess the free radical- scavenging activity of plant extracts as well as isolated pure compounds [17,18]. Result of DPPH test of Lamium longiflorum Ten Crude extract was displayed in figure 1:
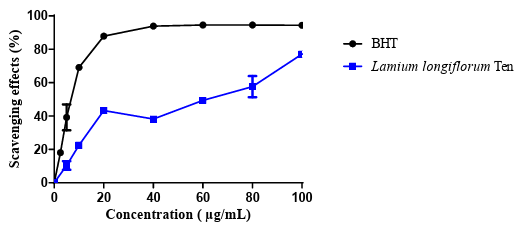
Figure 2: DPPH test of crude extract of Lamium longiflorum Ten.
The antioxidant concentration giving 50% inhibition of the initial DPPH concentration (50% efficiency concentration, EC50) is most frequently applied to compare antioxidant properties of compounds [19]. In this case Lamium longiflorum make 62,13±2,34 µg/mL*** against 8,76±0,69 μg/mL for the standard (BHT).
A direct relationship has been found between the content of total phenolics and antioxidant capacity of plants [20]. This result corroborates with the low quantity of polyphenols founds in crude extract, but still important compared with the results of other species of Lamium as demonstrated by Trouillas et al. (2003) when a value of EC 50 = 280 µg/mL were found in aqueous extract of Lamium album. However, Budzianowski and Budzianowska (2006) found an EC 50 = 1 µg/mL in crude extract of the flowers in the same species [21]. Concerning the antimicrobial activity, a very important results have been obtained (figure 3):
Figure 3: Anti-bacterial power of crude extract of Lamium longiflorum against Methicillin-resistant Staphylococcus aureus (MRSA) ATCC 43300 and Bacillus subtilis ATCC6633.
Lamium longiflorum crude extract make 16,67±0,47 mm against MRSA and 11,33±0,47 mm**** against Bacillus. However, Vancomycin gave 30,00±0,82 mm and Gentamicin give 27,33±0,24 mm.
Methicillin-resistant Staphylococcus aureus (MRSA) continues to be a predominant cause of both nosocomial and community- acquired infections owing to the development of intrinsic and acquired multidrug resistance to b-lactam antibiotics [22]. The majority of instances of nosocomial illness with MRSA are not curable with standard antibiotics, which is a serious concern [23].
Phenolic acids synthesized from phenylpropanoid intermediates such as caffeic acid, ferulic acid, p-coumaric acid, sinapic acid, protocatechuic acid, flavonoids aglycones and glycosides have shown a robust bactericidal and fungicidal activity [24] (Barber et al., 2000). Flavonoids are a group of polyphenols having the capacity to complex with the extracellular proteins and enzymes associated with the stability of bacterial cell wall structure and thus destroy it [25].
The main action of anti-inflammatory agents is the inhibition of cyclooxygenase enzymes, which are responsible for the conversion of arachidonic acid to prostaglandins. Since human red blood cell (HRBC) membranes are similar to these lysosomal membrane components, the prevention of Hypotonicity induced HRBC membrane lysis was taken as a measure in estimating the anti-inflammatory property of various extracts [26]. The power of Lamium longiflorum to prevent the erythrocytes of haemolysis is showed below (figure 4):
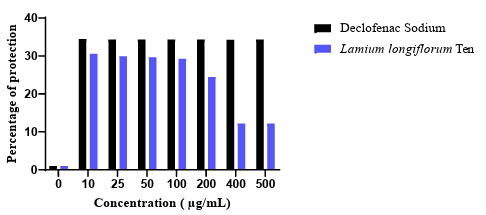
Figure 4: The percentage inhibition of hypotonicity induced haemolysis of HRBCs (%) of standard and crude extract of Lamium longiflorum Ten
It’s clearly that Lamium longiflorum crude extract can prevent HRBC from haemolysis at very low concentration (86,70±2,11 % of protection at a concentration of 10 µg/mL) until 100 µg/mL at which the percentage gradually decreases to reach a value of 29,25±4,39 %**** at 500 µg/mL.
The oxidative damage of erythrocyte membrane is the primary cause of reduced capacity of the red blood cells to withstand mechanical and osmotic stress [27]. However, reports have shown that certain saponins and flavonoids exert profound stabilizing effects on lysosomal membrane both In-vivo and In- vitro, while tannins and saponins possess ability to bind cations, thereby stabilizing erythrocyte membranes and other biological macromolecules [28,29-34]. But, at certain concentration the accumulation of these phytochemicals can plays the opposite role.
Conclusion
For a long time plants were used for the treatment of many human health problems; therefore, human beings have extensively studied these powerful sources of bioactive molecules. Our study focused on In-vitro and Ex-vivo tests to reveals the biological activities of Lamium longiflorum Ten. Results show an interesting antibacterial power against Methicillin-resistant Staphylococcus aureus and antinflammatory effect of 86,70 ±2,11 % of protection of HRBC from haemolysis. Furthers, molecular studies are needed.
Conclusion
For a long time plants were used for the treatment of many human health problems; therefore, human beings have extensively studied these powerful sources of bioactive molecules. Our study focused on In-vitro and Ex-vivo tests to reveals the biological activities of Lamium longiflorum Ten. Results show an interesting antibacterial power against Methicillin-resistant Staphylococcus aureus and antinflammatory effect of 86,70 ±2,11 % of protection of HRBC from haemolysis. Furthers, molecular studies are needed.
References
- Bianco, A., Melchioni, C., Ramunno, A., & Serafini, M. (2003). Iridoid glucosides from Lamium garganicum flowers. Natural Product Research, 17(4), 225-227.
- Harley, R. M., Atkins, S., Budantsev, A. L., Cantino, P. D.,Conn, B. J., Grayer, R., & Upson, T. (2004). Labiatae.In Flowering Plants· Dicotyledons: Lamiales (exceptAcanthaceae including Avicenniaceae) (pp. 167-275). Berlin, Heidelberg: Springer Berlin Heidelberg.
- Bendiksby, M., Thorbek, L., Scheen, A. C., Lindqvist, C., & Ryding, O. (2011). An updated phylogeny and classification of Lamiaceae subfamily Lamioideae. Taxon, 60(2), 471- 484.
- Mennema, J. (1989). A taxonomic revision of Lamium (Lamiaceae) (Vol. 11). Brill Archive.
- Akkol, E. K., Yalçin, F. N., Kaya, D., Çalis, I., Yesilada, E., & Ersöz, T. (2008). In vivo anti-inflammatory and antinociceptive actions of some Lamium species. Journal of Ethnopharmacology, 118(1), 166-172.
- Berezina, V. S., Budantsev, A. L., & Teslov, L. S. (2000). Chemical composition of genus Lamium LSL species. Rastitel'nye Resursy, 36(3), 122-132.
- Savchenko, T., Blackford, M., Sarker, S. D., & Dinan, L. (2001). Phytoecdysteroids from Lamium spp: identification and distribution within plants. Biochemical systematics and ecology, 29(9), 891-900.
- Krawczyk, K., Szczecinska, M., & Sawicki, J. (2014). Evaluation of 11 single-locus and seven multilocus DNA barcodes in L amium L. (L amiaceae). Molecular ecology resources, 14(2), 272-285.
- Krawczyk, K., & Sawicki, J. (2013). The uneven rate of the molecular evolution of gene sequences of DNA-Dependent RNA polymerase I of the Genus Lamium L. International Journal of Molecular Sciences, 14(6), 11376-11391.
- Quezel, P., & Santa, S. (1963). New flora of Algeria and southern desert regions. New flora of Algeria and southern desert regions.
- Li, W., Wei, C., White, P. J., & Beta, T. (2007). High-amylose corn exhibits better antioxidant activity than typical and waxy genotypes. Journal of agricultural and food chemistry, 55(2), 291-298.
- Bahorun, T., Gressier, B., Trotin, F., Brunet, C., Dine, T., Luyckx, M., ... & Pinkas, M. (1996). Oxygen species scavenging activity of phenolic extracts from hawthorn fresh plant organs and pharmaceutical preparations. Arzneimittel- forschung, 46(11), 1086-1089.
- Bettaieb, R. I., Bourgou, S., Ben, I., Slimen, D., Jabri, K.I., Hamrouni, S. I., Msaada, K., Limam, F., Marzouk, B. (2011). Food Bioprocess Techno, 1007.
- National Committee. Clinical Laboratory Standards (1999) Performance standards for antimicrobial disc susceptibility test. Approved Standard. NCCLS Publication M2-A5, Villanova, PA, USA.
- NCCLS (National Committee for Clinical Laboratory Standards)., 1997. Performance standards for antimicrobial disk susceptibility test. Wayne Pa. 6th ed. Approved Standard, M2-A6.
- Chippada, S. C., Volluri, S. S., Bammidi, S. R., & Vangalapati, M. (2011). In vitro anti-inflammatory activity of methanolic extract of Centella asiatica by HRBC membrane stabilisation. Rasayan J Chem, 4(2), 457-60.
- Al-Hadhrami, R. M. S., & Hossain, M. A. (2016). Evaluation of antioxidant, antimicrobial and cytotoxic activities of seed crude extracts of Ammi majus grown in Oman. Egyptian journal of basic and applied sciences, 3(4), 329-334.
- Al Rashdi, R. S. Y., Hossain, M. A., & Al Touby, S. S. J. (2021). Antioxidant and antibacterial activities of leaves crude extracts of Adenium obesum grown in Oman National Botanical Garden. Advances in Biomarker Sciences and Technology, 3, 8-14.
- Sridhar, K., & Charles, A. L. (2019). In vitro antioxidant activity of Kyoho grape extracts in DPPH and ABTS assays: Estimation methods for EC50 using advanced statistical programs. Food Chemistry, 275, 41-49.
- biological, and anatomical methods, Authentication Food Wine. (2007). 290–304. Chapter 19.
- Tsai, C., Lin, L. H., Wu, C. S., & Kwan, C. C. (2010).Surface properties of lithospermum-containing multiple phase emulsion systems. Journal of Applied Polymer Science, 117(2), 1041-1046.
- Lin, L. H., Lai, Y. C., Chen, K. M., & Chang, H. M. (2015).Oxyethylene chain length affects the physicochemical properties of sugar-based anionic surfactants with phosphates groups. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 485, 118-124.
- Lin, L. H., Lai, Y. C., Chen, K. M., & Li, C. S. (2016).Preparation and surface activities of modified soy protein– dextrin surfactants. Journal of Surfactants and Detergents, 19, 19-28.
- Lin, L. H., & Chou, Y. S. (2010). Surface activity and emulsification properties of hydrophobically modified dextrins. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 364(1-3), 55-60.
- Barreira, J. C., Ferreira, I. C., Oliveira, M. B. P., & Pereira,J. A. (2008). Antioxidant activities of the extracts from chestnut flower, leaf, skins and fruit. Food chemistry, 107(3), 1106-1113.
- Yordanova, Z. P., Zhiponova, M. K., Iakimova, E. T., Dimitrova, M. A., & Kapchina-Toteva, V. M. (2014). Revealing the reviving secret of the white dead nettle (Lamium album L.). Phytochemistry Reviews, 13, 375-389.
- Murray, P. R., Baron, E.J., Jorgensen, J. H., Pfaller, M. A., Tenover, F. C., Yolken, R. H. (2003). 8th ed. Manual of Clinical Microbiology, mBio, Washington, pp. 304–404.
- Muhsinah, A. B., Maqbul, M. S., Mahnashi, M. H., Jalal,M. M., Altayar, M. A., Saeedi, N. H., ... & Mohammed, T. (2022). Antibacterial activity of Illicium verum essential oil against MRSA clinical isolates and determination of its phyto-chemical components. Journal of King Saud University-Science, 34(2), 101800.
- Barber, P. H., Palumbi, S. R., Erdmann, M. V., & Moosa,M. K. (2000). A marine Wallace's line?. Nature, 406(6797), 692-693.
- TP, C. (2005). Antimicrobial activity of flavonoids. Int J Antimicrobial Agents, 26, 343-356.
- Nagaharika, Y., & Rasheed, S. (2013). Anti-inflammatory activity of leaves of Jatropha gossypifolia L. by HRBC membrane stabilization method. Journal of Acute Disease, 2(2), 156-158.
- Chikezie, P. C., Uwakwe, A. A., & Monago, C. C. (2010). Comparative osmotic fragility of three erythrocyte genotypes (HbAA, HbAS and HbSS) of male participants administered with five antimalarial drugs. African Journal of Biochemistry Research, 4(3), 57-64.
- Middleton, Jr., MD, E. (1996). Biological properties of plant flavonoids: an overview. International Journal of Pharmacognosy, 34(5), 344-348.
- El-Shabrawy, O. A., El-Gindi, O. D., Melek, F. R., Abdel- Khalik, S. M., & Haggag, M. Y. (1997). Biological properties of saponin mixtures of Fagonia cretica and Fagonia mollis.



