Research Article - (2024) Volume 2, Issue 1
Anaesthesia for Non-Cardiac Surgery in Patients with Hypertrophic Cardiomyopathy
Received Date: Nov 10, 2023 / Accepted Date: Dec 11, 2023 / Published Date: Jan 22, 2024
Copyright: ©Â©2024 Joy JX Chia, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Chia, J. J. X., Valchanov, K. P., Roscoe, A. J. (2024). Anaesthesia for Non-Cardiac Surgery in Patients with Hypertrophic Cardiomyopathy. Trans Med OA, 2(1), 01-08.
Abstract
Hypertrophic cardiomyopathy (HCM) is a common condition. Recent advancements in treatment and access to multidisciplinary care have improved such patients' lifespans and lower morbidity. Anesthetists are thus likely to encounter patients with varying severity of HCM presenting for non-cardiac surgery. The pathophysiology of HCM is complex, and a thorough understanding is vital to provide a safe anesthetic to such patients. We present a review of reported anesthetic techniques that have been safely used in patients with HCM undergoing non-cardiac surgery since 1986, as well as the management of complications that were encountered. In most cases, HCM is a benign condition and patients can expect a normal life expectancy. As long as appropriate hemodynamic goals are achieved intraoperatively, most anesthetic techniques can be safely utilized for patients with HCM with good outcomes.
Keywords
Hypertrophic Cardiomyopathy, Anesthesia, Left Ventricular Obstruction, Hypertrophic Obstructive Cardiomyopathy, Non-Cardiac Surgery.
Introduction
Hypertrophic cardiomyopathy (HCM) is the commonest inherited cardiomyopathy, affecting up to 0.2% of the population [1-4]. The condition was previously called subaortic stenosis, or hypertrophic obstructive cardiomyopathy. However, in about 25% of cases, obstruction may be absent despite provocative maneuvers [4-6]. Thus, the term hypertrophic cardiomyopathy is used in the recent American Heart Association (AHA) 2020 guidelines [4]. The pathophysiology of HCM is complex, involving interplay between multiple abnormalities including diastolic dysfunction, left ventricular outflow obstruction (LVOTO), and systolic anterior motion of mitral valve leaflets.
The anesthetist is likely to encounter patients with varying severity of HCM. It is important to understand the underlying pathophysiology of this condition, and the hemodynamic goals during anesthesia. The aim of this manuscript is to highlight the salient pathophysiology in HCM patients and review the cases in the literature, thereby offering knowledge and solutions for safe perioperative management of HCM patients. In the last two decades, there have been numerous case reports and series which we have identified and included in this literature review, equipping anesthetists with greater numbers of experience in managing patients with HCM. Furthermore, for the last decade, there have been two new medications specifically designed for treatment of HCM, and have undergone randomized controlled trials.
Epidemiology
HCM is a common condition affecting 0.2% of the population [7]. Although there is male preponderance, the female gender is associated with higher mortality and worse outcomes [8,9]. Up to one third of HCM cases are autosomal dominant and involve genes encoding sarcomere-associated proteins. The most common causal genes are MYH7 and MYBPC3, which account for up to 50% of familial cases of HCM [3]. However, among those with the HCM genotype, there is variable penetrance and significant phenotypic heterogeneity. Carriers of the HCM genotype may exhibit less severe HCM, or even the absence of HCM altogether [10]. The AHA/ACC thus recommends genetic testing for individuals with HCM, with appropriate pre-test and post-test counselling [4].
Recent studies suggest that individuals with HCM do not have a higher mortality rate or lower life expectancy than the general adult population [4]. This is in part due to improved risk stratification of at-risk individuals who may benefit from implantable cardioverter-defibrillators (ICD). There are many consensus guidelines for risk stratification of individuals at risk of sudden cardiac death (SCD), and common risk factors include the presence and severity of left ventricular outflow obstruction, severity of left ventricular hypertrophy, episodes of ventricular tachycardia, and genotype [4,11]. However, these risk factors and algorithms fail to represent individuals with undiagnosed HCM, as this population is understandably difficult to identify and study. Nonetheless, this is a population that cannot be ignored; in a postmortem study of sudden deaths in Spain between 2010 and 2015, up to 21% of sudden deaths were attributed to previously undiagnosed HCM [12].
Pathophysiology
HCM can lead to hypertrophy of different parts of the left ventricle. While the basal-septal region is most commonly affected, the apex, free wall, and postero-lateral wall may be affected as well [13]. The right ventricle is rarely involved. LVOTO may occur from asymmetrical hypertrophy of the interventricular septum inferior to the aortic valve (which was why HCM was previously called hypertrophic subaortic stenosis), or from systolic anterior motion (SAM) of the mitral valve. Narrowing of the left ventricular outflow tract (LVOT) leads to increased velocity of blood flow.
This creates drag forces and a Venturi effect on the mitral leaflets. The increased velocity of blood causes reduced pressure in the LVOT, and the lower pressure draws the anterior mitral leaflet (AML) into the LVOT. This displacement of the AML during systole worsens LVOTO and is referred to as SAM (Systolic Anterior Motion of the mitral leaflets). SAM of the anterior leaflet also disrupts the coaptation of the mitral valve leaflets, and often results in mitral regurgitation. The result is a dynamic obstruction that varies with cardiac contractility, preload, and afterload. Microvascular dysfunction couples with the cycle of worsening hypertrophy and obstruction, predisposing HCM patients to myocardial ischemia and heart failure [14].
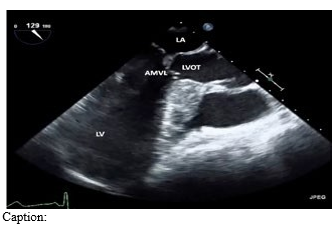
Figure 1: LAX view of septal hypertrophy and SAM, resulting in LVOTO during ventricular systole.
In late stages, 3-5% of HCM patients may progress into the ‘dilated phase of HCM’. This phase is characterized with reduced left ventricular ejection fraction, as well as left ventricular wall thinning and cavity dilatation [15]. It is important to recognize this as a separate entity from dilated cardiomyopathy, as such patients with late stage HCM as they have a higher incidence of ventricular arrhythmias and mortality compared to patients with dilated cardiomyopathy
Treatment of HCM
Medical therapy for HCM involves beta-blockers and calcium channel blockers. In patients at risk of SCD, an automatic implantable cardioverter-defibrillator (AICD) may be considered. Patients whose disease progresses to advanced heart failure despite optimal medical therapy may require invasive septal reduction therapy. Options include trans-aortic septal myectomy, or percutaneous alcohol septal ablation. The latter newer technique involves injection of ethanol through the coronary arteries, to induce a localized infarction of the ventricular septum.
Novel Therapies
Current medical and surgical treatments for HCM do not treat the underlying pathophysiology of abnormal ventricular contractability and hypertrophy. Beta blockers and calcium channel blockers aim to reduce heart rate and address the resultant diastolic dysfunction from ventricular hypertrophy. Septal myectomy and ablation techniques reduce the amount of hypertrophic ventricular septum, but cannot reverse the underlying hypercontractile state of the remaining myocardium. In the last decade, cardiac myosin inhibitors (CMI) mavacamten and aficamten have been developed to treat the molecular basis of HCM. These first-in-class molecules reduce the formation of actin-myosin cross-bridges, targeting the dysregulated hyper-contractility of the hypertrophic ventricle [16,17]. The EXPLORER-HCM and VALOR-HCM trials show promising evidence that CMIs may be beneficial in relieving symptoms of heart failure in advanced HCM, as well as reducing the need for septal myectomy [17,18]. However, mavacamten is recommended only for patients with a left ventricular ejection fraction ≥55% as it may be associated with a reversible reduction in left ventricular ejection fraction by up to 10% [17]. Despite promising early evidence for the use of CMIs, further studies are needed on the long-term efficacy, safety profile, and cost effectiveness of these novel agents [19].
Considerations for Anesthesia
When patients with HCM present for non-cardiac surgery, the hemodynamic goals can be summarized as: 1. Maintain adequate preload 2. Maintain afterload 3. Avoid increases in contractility 4. Avoid tachycardia. Exacerbations of LVOTO reduce cardiac output and coronary perfusion; coupled with impaired diastolic function and a hypertrophic myocardium, this further predisposes HCM patients to myocardial ischemia. Hypovolemia should be avoided, as the hypertrophied myocardium has often adapted to higher left ventricular diastolic pressure. The hypocompliant, hypertrophic left ventricle is frequently associated with impaired ventricular filling during diastole i.e., diastolic dysfunction. Adequate preload is also necessary to promote forward flow due to the pressure gradient in the left ventricular outflow tract. Conversely, hypovolemia exacerbates the Venturi effect and pressure gradient within the LVOT, drawing the AML into the LVOT and worsening mitral regurgitation.
However, judicious fluid management is paramount, as fluid overload can lead to pulmonary oedema. Fluid status needs to account for baseline and can be assessed with clinical maneuvers (such as passive leg raise, end expiratory occlusion test), with point-of-care imaging, or invasive monitoring (such as pulmonary capillary wedge pressure and central venous pressure monitoring), and the use of cardiac output monitors. Reductions in afterload result in a greater pressure gradient across the left ventricular outflow tract, which exacerbates the Venturi effect and worsens LVOTO. Reduction in afterload also reduces perfusion of the aortic sinuses and coronary arteries, which may precipitate ischemia of the hypertrophic left ventricle. Increases in contractility and heart rate increase myocardial oxygen requirements and reduce diastolic time for ventricular filling and coronary perfusion. In addition, positive inotropy is associated with increased pulmonary artery wedge pressure, which can result in pulmonary edema. Thus, using vasopressors to correct hypotension would be most logical. Phenylephrine, a selective alpha-1 adrenergic agonist, causes vasoconstriction and reflex bradycardia, hence is the commonest agent reported in the literature [20-32].
Considerations for Obstetric Anesthesia
Pregnancy and delivery are associated with hemodynamic changes which may be poorly tolerated by the HCM patient. Plasma volume increases from the first trimester and is accompanied by an increase in stroke volume and heart rate [33]. At the time of delivery, there are significant changes to a parturient’s volume status. Obstetric hemorrhage can cause profound hypovolemia over a relatively short period of time and may be subsequently followed by uterine contraction and autotransfusion. There is also a surge in circulating catecholamines due to pain, resulting in tachycardia and reduced time for diastole [34,35]. These physiologic changes worsen LVOTO and may precipitate pulmonary edema in the parturient.
There has been a gradual trend of improving outcomes for parturients with HCM in the last 3 decades. In 1999, Autore et al demonstrated that parturients with HCM and LVOTO experienced a higher mortality than the general population, at 10 deaths per 1000 live births [36]. Parturients who experienced cardiac symptoms even prior to their pregnancy were at an increased risk of deterioration. This finding was echoed in a study by Thaman in 2003 [37]. In more recent years, however, data from the European Pregnancy Society of Cardiology in 2019 suggests that pregnancy and delivery are generally well tolerated by patients with HCM, even if they are known to have LVOTO [38]. This trend could be in part due to improved risk stratification of patients with HCM, and improved access to interventions such as ICD implantation. It may also reflect improved access to multidisciplinary care. The 2020 American Heart Association guidelines suggests that for parturients with HCM, plans for delivery should be made in conjunction with a multi-disciplinary team, involving cardiologists, obstetricians, anesthetists, and fetal medicine specialists. Previously, it was thought that patients with HCM should deliver via elective cesarean section. This was thought to avoid the hemodynamic changes associated with labor, pain, and bearing down during vaginal delivery. Historically, general anesthesia was the choice of anesthetic for the cesarean section; spinal and epidural anesthesia were avoided over concerns of vasodilatation worsening SAM and LVOTO. However, more recent data suggests that vaginal delivery and epidural anesthesia are generally well tolerated by parturients with HCM [4,39]. Further, cesarean section may be associated with greater blood loss compared to normal vaginal delivery [40].
Methodology
We conducted a literature review of case studies and case series on HCM the Pubmed and Medline databases. We hand-searched the keywords hypertrophic cardiomyopathy, hypertrophic obstructive cardiomyopathy, subaortic stenosis, anesthesia, and surgery. Only non-cardiac surgery was included, and pediatric patients were excluded. Only cases involving isolated HCM were included; cases involving genetic syndromes or congenital heart disease were excluded. We excluded articles that were not in English. We reviewed a total of 84 case reports; 28 reports were excluded as the article was either not accessible, or not in English. Our report discusses the remaining 56 reports, involving a total of 64 patients.
Literature Review
Obstetric Anesthesia
Twenty-five of the patients were obstetric, of which 18 underwent cesarean section, 6 had vaginal deliveries, and 1 underwent a laparotomy for post-partum hemorrhage. In addition to standard ASA monitoring, most patients also had invasive blood pressure monitoring, central venous pressure monitoring, and large bore intravenous access [31,32,36,41-51].
The use of trans-thoracic and trans-esophageal echocardiography, pulmonary artery catheterization, and Flotrac were also mentioned [44-46,48,51]. Regional anesthetic was the most common technique of choice, described in 8 cases. Epidural anesthesia was most common, followed by combined spinal-epidural, continuous spinal, and single shot spinal [31,32,43-50,52-54].
Only 7 cases described the use of general anesthetic, of which 1 case involved a pre-induction single-shot spinal, followed by induction of general anesthesia [41,42,46,49,51,55,56]. One case described a judicious crystalloid fluid bolus prior to induction to maintain preload and counteract the vasodilation associated with induction [42]. Rapid sequence induction was commonly induced, and induction agents described include propofol, thiopentone, and etomidate. Relaxants used included suxamethonium and rocuronium, and all cases of general anesthetic used an opioid for induction, such as fentanyl or remifentanil.
Although no mortality was reported, one case reported a perioperative myocardial infarction due to hypotension following the spinal anesthetic [54]. Three cases of pulmonary oedema were reported, and required diuresis with furosemide postoperatively [49,56]. Of the 18 cases of cesarean section, 12 were elective. This underscores the importance of planning early for delivery in parturients with HCM.
Non-Obstetric Anesthesia
We reviewed 30 cases of general anesthesia (4 of which were liver transplant surgeries), 3 cases of central neuraxial anesthesia, and 4 cases of peripheral nerve block. In addition to standard ASA monitoring, most patients also had invasive monitoring; 17 out of 26 patients undergoing general anesthesia for non-transplant surgery had arterial blood pressure monitoring [20–25,27,55,57–63]. IA monitoring was also used in patients undergoing regional techniques [64-66]. Central venous catheterization was less common and was utilized in 7 cases undergoing general anesthesia for non-transplant surgery under general anesthesia, and 2 case under regional anesthesia [21,23,24,55,59,61,64,66,67]. Less commonly used methods include pulmonary artery catheterization (used in 3 cases, all in the 1990s), and echocardiography (4 cases of transesophageal echocardiography under general anesthetic, and 1 case with transthoracic echocardiography under a regional anesthetic) [22,26,60,65,68]. 3 cases in Japan described monitoring with Flotrac [20,57].
For general anesthesia, preload was typically maintained with a gentle fluid bolus prior to induction, in anticipation of relative hypovolemia due to vasodilatation [22,25,26,68]. In two cases, fluid status was guided by intraoperative transesophageal echocardiography [22,26]. A variety of induction agents were described, including propofol, etomidate, and thiopentone. Adjuncts to induction included opioids like fentanyl and remifentanil, benzodiazepines, and ketamine. Relaxants used included suxamethonium, vecuronium, and rocuronium. Attention was paid to avoiding tachycardia associated with anxiety pre-operatively, during laryngoscopy, and postoperatively due to pain. Premedication with an anxiolytic was described in 4 cases [24,27,59,69]. One case even described application of eutectic mixture of local anesthetic (EMLA) prior to intravenous cannulation, as well as topicalization of the airway to reduce the response to laryngoscopy [21]. Beta-adrenergic antagonists were also used to attenuate laryngoscopy-related tachycardia; the use of short-acting agents such as esmolol and landiolol bolus and infusion were described [20,23,59,61,63]. Longer acting beta- adrenergic antagonists such as propranolol and metoprolol were also described [26,27]. Hemodynamic parameters were maintained with phenylephrine and noradrenaline. Maintenance of anesthesia with volatile gas and total intravenous anesthesia were both described.
Postoperatively, in addition to long-acting opioids, regional techniques such as epidural and peripheral nerve block (single shot and catheter) were also used as adjuncts for analgesia [23,59,60,62,68-70]. Adequate postoperative analgesia is paramount, as illustrated in one case from India. Following a bilateral total knee replacement under general anesthetic and bilateral femoral nerve catheter infusions, the patient developed postoperative pain and tachycardia. Despite further boluses through the nerve catheters, as well as metoprolol boluses, the patient developed acute pulmonary oedema requiring furosemide diuresis [23]. This was the only reported case of a patient developing postoperative complications following non- obstetric surgery.
Two patients required postponement of surgery for insertion of an ICD or pacemaker. The first patient presented for elective right lung surgery and developed ventricular tachycardia during insertion of a thoracic epidural. This was aborted with a bolus of lidocaine, however during positioning before surgery, ventricular tachycardia recurred. Surgery was postponed, and the patient underwent insertion of an ICD [70]. The second case presented for a right upper lobectomy, also involving an uneventful thoracic epidural prior to induction [69]. However, post-induction the patient developed profound hypotension and bradycardia. The surgery was also postponed, and the patient underwent insertion of a pacemaker.
Undiagnosed HCM
The cases above describe instances where HCM was diagnosed preoperatively. In those cases, the severity of HCM was assessed preoperatively, and the appropriate anesthetic technique and monitoring was chosen accordingly. We have found 6 cases where the first presentation of HCM occurred perioperatively or intraoperatively. 3 of these were emergency surgeries, and the other half presented for elective surgeries [71-76].
Majority of cases presented with intraoperative hemodynamic instability despite the use of vasoactive agents. In case in 1984, the diagnosis of HCM was made intraoperatively by identifying a click and murmur on esophageal stethoscope [72]. In more recent years, the use of point-of-care ultrasound in perioperative care has led to the diagnosis of HCM and LVOTO with greater accuracy. In two cases, hemodynamic instability not responding to vasopressors prompted the use of intraoperative transesophageal echocardiography, which confirmed the diagnosis of HCM and LVOTO [75,76]. One case presented with LVH incidentally noted on preoperative point-of-care transthoracic cardiac ultrasound, prompting a preoperative cardiology consultation and diagnosis prior to emergent surgery for tracheal rupture [71].
The timely suspicion of HCM in hypotension worsening with positive inotropy and chronotropic allowed the appropriate hemodynamic goals to be instituted, and there are thus far no reported mortalities involving patients with undiagnosed HCM undergoing non-cardiac surgery. Nonetheless, one instance of morbidity was reported in a patient with previously undiagnosed HCM with LVOTO [76]. The patient was known to have symmetric, concentric left ventricular hypertrophy, and presented for elective trans-aortic valve replacement under sedation. LVOTO was not present during pre-operative echocardiographic evaluation of aortic stenosis. However, during rapid ventricular pacing, the patient developed profound hypotension requiring phenylephrine, ephedrine, vasopressin, noradrenaline, and adrenaline infusions. Despite the diagnosis of LVOTO in the presence of ventricular hypertrophy, and the subsequent use of esmolol and phenylephrine to increase afterload and reduce heart rate, the patient developed acute pulmonary edema, requiring intubation and transfer to the intensive care unit.
Patients with HCM and LVOTO may present with sudden profound hypotension during anesthesia due to reduction in afterload and the precipitation of LVOTO. The hypotension may be profound and unresponsive to positive inotropic and chronotropic agents. We suggest that when patients present with hemodynamic instability that worsens with positive chronotropy or inotropy, HCM should be considered as a diagnosis. Resuscitation should be guided either transthoracic or transesophageal intraoperative echocardiography, and treatment should prioritize the goals of rate control, and increase in afterload.
Discussion
We have reviewed all literature on anesthesia for HCM up to 2023. For obstetric patients with HCM, pregnancy is usually well tolerated. Early involvement of a cardiologist and multidisciplinary team to plan for delivery is important where the diagnosis of HCM is known. Both general and regional anesthesia is well tolerated where attention is paid to the hemodynamic changes and shifts in fluid status during pregnancy, labor, and delivery. For non-obstetric surgery, both general and regional techniques are also well tolerated. Attention should be paid to avoiding tachycardia due to pain or laryngoscopy, as this can worsen LVOTO and precipitate myocardial ischemia. Appropriate monitoring of hemodynamic parameters and fluid status can be useful to avoid fluid overload and pulmonary edema.
When the diagnosis of HCM is not known, the management of sudden profound hypotension may be challenging. Due to the mechanism of SAM, vasoactive agents that result in positive chronotropy and inotropy may paradoxically worsen LVOTO and hypotension. Thus hypotension may be refractory to common vasoactive agents such as ephedrine and noradrenaline. Point of care cardiac echocardiography can be invaluable in discerning the diagnosis of HCM and SAM, and guide resuscitation and fluid therapy. The limitations of our literature review include publication bias, as cases with unfavorable outcomes are less likely to be submitted for publication. The diagnosis of HCM may also be missed in certain cases, and as mentioned, previously undiagnosed HCM can be a significant cause of SCD.
Conclusion
Knowledge of the pathophysiology of HCM is important for the anesthetist, who will undoubtedly encounter HCM during their career. It is a benign condition, and most patients with HCM can expect to have a normal life expectancy. With support from a multidisciplinary team, the diagnosis of HCM is often known, and the appropriate care with regards to hemodynamic monitoring can be undertaken for a safe anesthetic. Where the diagnosis of HCM is not known, the hemodynamic status of these patients under anesthesia may be more challenging to manage. Adjuncts such as point of care echocardiography may be helpful in discerning the diagnosis and guiding their therapy.
Key Message
1. The pathophysiology of HCM is dynamic and involves ventricular hypertrophy, systolic anterior movement of the mitral leaflet. This results in left ventricular outflow obstruction.
2. The intraoperative hemodynamic goals for HCM include maintaining adequate preload, maintaining afterload, avoiding hypercontractility, and avoiding tachycardia
3. Pregnancy and parturition are generally well tolerated in patients with HCM. Regional and general anesthesia is well tolerated for delivery.
4. A variety of anesthetic techniques, including general anesthesia, central neuraxial anesthesia, and regional anesthesia, can be safely utilised in patients with HCM insofar as appropriate hemodynamic goals are achieved
5. In patients with undiagnosed HCM, majority presented with intraoperative hemodynamic instability that did not respond to typical vasopressive agents. The used of point-of-care ultrasound may be invaluable in establishing the diagnosis of LVOTO, and aiding the appropriate hemodynamic goals to be instituted.
Conflict of interest
The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Funding
No funding was necessary.
Author Contributions
JCJX: Writing - original draft, Writing - review and editing, methodology, data collection. KPV: Writing - review and editing, methodology, supervision, resources. AR: Writing - review and editing, supervision, resources
References
- Brieler, J. A. Y., Breeden, M. A., & Tucker, J. (2017). Cardiomyopathy: an overview. American family physician, 96(10), 640646.
- Ciarambino, T., Menna, G., Sansone, G., & Giordano,M. (2021). Cardiomyopathies: an overview. International journal of molecular sciences, 22(14), 7722.
- Marian, A. J., & Braunwald, E. (2017). Hypertrophic cardiomyopathy: genetics, pathogenesis, clinical manifestations, diagnosis, and therapy. Circulation research, 121(7), 749-770.
- Ommen, S. R., Mital, S., Burke, M. A., Day, S. M., Deswal, A., Elliott, P., ... & Sorajja, P. (2020). 2020 AHA/ACC guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Journal of the American College of Cardiology, 76(25), 3022-3055.
- Maron, M. S., Rowin, E. J., Olivotto, I., Casey, S. A.,Arretini, A., Tomberli, B., ... & Maron, B. J. (2016). Contemporary natural history and management of nonobstructive hypertrophic cardiomyopathy. Journal of the American College of Cardiology, 67(12), 1399-1409.
- Day, S. M. (2019). Nonobstructive hypertrophic cardiomyopathy—the high-hanging fruit. JAMA cardiology, 4(3), 235-236.
- Semsarian, C., Ingles, J., Maron, M. S., & Maron, B. J. (2015). New perspectives on the prevalence of hypertrophic cardiomyopathy. Journal of the American College of Cardiology, 65(12), 1249-1254.
- Olivotto, I., Maron, M. S., Adabag, A. S., Casey, S. A.,Vargiu, D., Link, M. S., ... & Maron, B. J. (2005). Gender- related differences in the clinical presentation and outcome of hypertrophic cardiomyopathy. Journal of the AmericanCollege of Cardiology, 46(3), 480-487.
- Trongtorsak, A., Polpichai, N., Thangjui, S., Kewcharoen, J., Yodsuwan, R., Devkota, A., ... & Estrada, A. Q. (2021). Gender-related differences in hypertrophic cardiomyopathy: a systematic review and meta-analysis. Pulse, 9(1-2), 38-46.
- Lorenzini, M., Norrish, G., Field, E., Ochoa, J. P., Cicerchia, M., Akhtar, M. M., ... & Elliott, P. M. (2020). Penetrance of hypertrophic cardiomyopathy in sarcomere protein mutation carriers. Journal of the American College of Cardiology, 76(5), 550-559.
- Maron, B. J., Rowin, E. J., & Maron, M. S. (2021). Evolution of risk stratification and sudden death prevention in hypertrophic cardiomyopathy: twenty years with the implantable cardioverter-defibrillator. Heart Rhythm, 18(6), 1012-1023.
- Frontera, A., Anselmino, M., Matta, M., Baccelli, A., Vlachos, K., Bonsignore, A., ... & Haïssaguerre, M. (2022). Ante-mortem characterization of sudden deaths as first- manifestation in Italy. Journal of Interventional Cardiac Electrophysiology, 1-8.
- Marian, A. J., & Braunwald, E. (2017). Hypertrophic cardiomyopathy: genetics, pathogenesis, clinical manifestations, diagnosis, and therapy. Circulation research, 121(7), 749-770.
- Vives, M., & Roscoe, A. (2014). Hypertrophic cardiomyopathy: implications for anesthesia. Minerva Anestesiologica, 80(12), 1310-1319.
- Goto, D., Kinugawa, S., Hamaguchi, S., Sakakibara, M., Tsuchihashi-Makaya, M., Yokota, T., ... & JCARE-CARD Investigators. (2013). Clinical characteristics and outcomes of dilated phase of hypertrophic cardiomyopathy: report from the registry data in Japan. Journal of Cardiology, 61(1), 65-70.
- Masri, A., & Olivotto, I. (2022). Cardiac myosin inhibitors as a novel treatment option for obstructive hypertrophic cardiomyopathy: addressing the core of the matter. Journal of the American Heart Association, 11(9), e024656.
- Olivotto, I., Oreziak, A., Barriales-Villa, R., Abraham, T. P., Masri, A., Garcia-Pavia, P., ... & Yamani, M. (2020). Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): a randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet, 396(10253), 759-769.
- Desai, M. Y., Owens, A., Wolski, K., Geske, J. B., Saberi,S., Wang, A., ... & Nissen, S. E. (2023). Mavacamten in patients with hypertrophic cardiomyopathy referred for septal reduction: week 56 results From the VALOR-HCM randomized clinical trial. JAMA cardiology, 8(10), 968- 977.
- Sykuta, A., Yoon, C. H., Baldwin, S., Rine, N. I., Young, M., & Smith, A. (2023). Cardiac Myosin Inhibitors: Expanding the Horizon for Hypertrophic Cardiomyopathy Management. Annals of Pharmacotherapy, 10600280231180000.
- Koyama, Y., Asami, Y., Nishikawa, H., Ikezaki, H., & Tsuzaki, K. (2022). Cardiovascular monitoring in patients with hypertrophic obstructive cardiomyopathy in a prone position: A report of 2 cases. Saudi Journal of Anaesthesia, 16(1), 108.
- Gosavi, K. S., & Mundada, S. D. (2012). Management of hypertrophic obstructive cardiomyopathy in prone position. Indian Journal of Anaesthesia, 56(3), 310-311.
- Gregory, S. H., & Fierro, M. A. (2016). The role of intraoperative transesophageal echocardiographic monitoring in a patient with hypertrophic cardiomyopathy undergoing laparoscopic surgery. Journal of clinical anesthesia, 34, 124-127.
- Rupal, S., Swami, A. C., Jindal, S., & Lata, S. (2013). Perioperative management of an elderly patient of hypertrophic obstructive cardiomyopathy for knee arthroplasty and the role of peripheral nerve blocks. Indian Journal of Anaesthesia, 57(4), 394.
- Jain, A., Jain, K., Bhagat, H., Mangal, K., & Batra, Y. (2010). Anesthetic management of a patient with hypertrophic obstructive cardiomyopathy with dual-chamber pacemaker undergoing transurethral resection of the prostate. Annals of Cardiac Anaesthesia, 13(3), 246-248.
- Ahmed, A., Zaidi, R. A., Hoda, M. Q., & Ullah, H. (2010). Anesthetic management of a patient with hypertrophic obstructive cardiomyopathy undergoing modified radical mastectomy. Middle East Journal of Anesthesiology, 20(5), 739.
- Popescu, W. M., & Perrino Jr, A. C. (2006). Critical cardiac decompensation during laparoscopic surgery. Journal of the American Society of Echocardiography, 19(8), 1074-e5.
- Edmends, S., & Ghosh, S. (1994). Hypertrophic obsrrucitve cardiomyopathy complicating surgery for cerebral artery aneursysm clipping. Anaesthesia, 49(7), 608-609.
- Kondo, T., Kusunoki, S., Kuroda, M., & Kawamoto, M. (2013). Difficult anesthesia management in a case of living donor liver transplantation with hypertrophic obstructive cardiomyopathy. Hiroshima Journal of Medical Sciences, 62(4), 91-94.
- Harley, I. D., Jones, E. F., Liu, G., McCall, P. R., & McNicol,P. L. (1996). Orthotopic liver transplantation in two patients with hypertrophic obstructive cardiomyopathy. British journal of anaesthesia, 77(5), 675-677.
- Buckenmaier, C. C., Steele, S. M., Nielsen, K. C., & Klein,S. M. (2002). Paravertebral somatic nerve blocks for breast surgery in a patient with hypertrophic obstructive cardiomyopathy. Canadian Journal of Anesthesia, 49(6), 571.
- Ishiyama, T., Oguchi, T., Iijima, T., Matsukawa, T., Kashimoto, S., & Kumazawa, T. (2003). Combined spinal and epidural anesthesia for cesarean section in a patient with hypertrophic obstructive cardiomyopathy. Anesthesia & Analgesia, 96(2), 629-630.
- Ferguson, E. A., Paech, M. J., & Veltman, M. G. (2006). Hypertrophic cardiomyopathy and caesarean section: intraoperative use of transthoracic echocardiography. International journal of obstetric anesthesia, 15(4), 311-316.
- Bedson, R., & Riccoboni, A. (2014). Physiology of pregnancy: clinical anaesthetic implications. Continuing education in anaesthesia, critical care & pain, 14(2), 69-72.
- Schinkel, A. F. (2014). Pregnancy in women with hypertrophic cardiomyopathy. Cardiology in review, 22(5), 217-222.
- Schaufelberger, M. (2019). Cardiomyopathy and pregnancy. Heart.
- Autore, C., Brauneis, S., Apponi, F., Commisso, C., Pinto, G., & Fedele, F. (1999). Epidural anesthesia for cesarean section in patients with hypertrophic cardiomyopathy: a report of three cases. The Journal of the American Society of Anesthesiologists, 90(4), 1205-1207.
- Thaman, R., Varnava, A., Hamid, M. S., Firoozi, S., Sachdev, B., Condon, M., ... & McKenna, W. J. (2003). Pregnancy related complications in women with hypertrophic cardiomyopathy. Heart, 89(7), 752.
- Roos-Hesselink, J., Baris, L., Johnson, M., De Backer, J., Otto, C., Marelli, A., ... & Hall, R. (2019). Pregnancy outcomes in women with cardiovascular disease: evolving trends over 10 years in the ESC Registry Of Pregnancy And Cardiac disease (ROPAC). European Heart Journal, 40(47), 3848-3855.
- American College of Cardiology. (2003). European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. J Am Coll Cardiol, 42, 1687- 1713.
- Ashikhmina, E., Farber, M. K., & Mizuguchi, K. A. (2015). Parturients with hypertrophic cardiomyopathy: case series and review of pregnancy outcomes and anesthetic management of labor and delivery. International Journal of Obstetric Anesthesia, 24(4), 344-355.
- Bouslama, M. A., Ferhi, F., Hacheni, F., Ons, K., Abdeljelil, K., Jazia, K. B., & Khairi, H. (2018). Pregnancy and delivery in woman with implantable cardioverter-defibrillator: what we should know. Pan African Medical Journal, 30(1).
- Mishra, S. K., Bhat, R. R., Kavitha, J., Kundra, P., & Parida,S. (2016). Obstetric hemorrhage in a case of hypertrophic obstructive cardiomyopathy with automatic implantable cardioverter defibrillator: Anaesthesia and intensive care management. Anesthesia, Essays and Researches, 10(1), 111.
- DesRoches, J. M., McKeen, D. M., Warren, A., Allen, V. M., George, R. B., Kells, C., & Shukla, R. (2016). Anesthetic management guided by transthoracic echocardiography during cesarean delivery complicated by hypertrophic cardiomyopathy. A&A Practice, 6(6), 154-159.
- Abe, T., Yamamoto, S., Yasuda, N., Uchino, T., Hidaka, S., Hagiwara, S., ... & Noguchi, T. (2010). Effective control of paroxysmal tachycardia with landiolol hydrochloride during cesarean section in a patient with hypertrophic obstructive cardiomyopathy. Journal of anesthesia, 24, 765-767.
- Okutomi, T., Kikuchi, S., Amano, K., Okamoto, H., & Hoka, S. (2002). Continuous spinal analgesia for labor and delivery in a parturient with hypertrophic obstructive cardiomyopathy. Acta anaesthesiologica scandinavica, 46(3), 329-331.
- Wadsworth, R., Greer, R., MacDonald, J. J. M., & Vohra, A. (2002). The use of remifentanil during general anaesthesia for caesarean delivery in two patients with severe heart dysfunction. International Journal of Obstetric Anesthesia, 11(1), 38-43.
- Della E, S., & JM, B. I. (2000). Elective cesarean section with epidural anesthesia in a pregnant woman withobstructive hypertrophic myocardiopathy. Revista Española de Anestesiología y Reanimación, 47(7), 320-322.
- Fairley, C. J., & Clarke, J. T. (1995). Use of esmolol in a parturient with hypertrophic obstructive cardiomyopathy. British journal of anaesthesia, 75(6), 801-804.
- Tessler, M. J., Hudson, R., Naugler-Colville, M., & Biehl,D. R. (1990). Pulmonary oedema in two parturients with hypertrophic obstructive cardiomyopathy (HOCM). Canadian journal of anaesthesia, 37, 469-473.
- Pryn, A., Bryden, F., Reeve, W., Young, S., Patrick, A., & McGrady, E. M. (2007). Cardiomyopathy in pregnancy and caesarean section: four case reports. International Journal of Obstetric Anesthesia, 16(1), 68-73.
- Nam, E., Toque, Y., Quintard, J. M., Barsam, E., Besserve, P., & Montravers, P. (1999). Use of transesophageal echocardiography to guide the anesthetic management of cesarean section in a patient with hypertrophic cardiomyopathy. Journal of cardiothoracic and vascular anesthesia, 13(1), 72-74.
- Lam, K. K., Ngan, W. K., Chen, P. P., & Gin, T. (2002).Patient-controlled epidural analgesia in a parturient with hypertrophic obstructive cardiomyopathy. International Journal of Obstetric Anesthesia, 11(4), 310-313.
- Paix, B., Cyna, A., Belperio, P., & Simmons, S. (1999). Epidural analgesia for labour and delivery in a parturient with congenital hypertrophic obstructive cardiomyopathy. Anaesthesia and intensive care, 27(1), 59-62.
- Schmitto, J. D., Hein, S., Braeuer, A., Popov, A. F., Quintel, M., & Schoendube, F. A. (2008). Perioperative myocardial infarction after cesarean section in a young woman with hypertrophic obstructive cardiomyopathy.
- Sahoo, R. K., Dash, S. K., Raut, P. S., Badole, U. R., & Upasani, C. B. (2010). Perioperative anesthetic management of patients with hypertrophic cardiomyopathy for noncardiac surgery: A case series. Annals of Cardiac Anaesthesia, 13(3), 253-256.
- Boccio, R. V., Chung, J. H., & Harrison, D. M. (1986). Anesthetic management of cesarean section in a patient with idiopathic hypertrophic subaortic stenosis. The Journal of the American Society of Anesthesiologists, 65(6), 663- 665.
- Sato, A., Morita, M., So, M., Tamura, T., Sano, F., Shibuya, Y., ... & Sobue, K. (2018). General Anesthetic Management of a Patient With Hypertrophic Cardiomyopathy for Oral Surgery: Did Digitalis Contribute to Bradycardia?. Anesthesia Progress, 65(3), 192-196.
- Nama, R. K., Parikh, G. P., & Patel, H. R. (2015). Anesthetic management of a patient with hypertrophic cardiomyopathy with atrial flutter posted for percutaneous nephrolithotomy. Anesthesia, Essays and Researches, 9(2), 284.
- Yemul-Golhar, S. R., Bhalerao, P. M., Gavali, Y., & Kelkar, K. V. (2012). A rare case of hypertrophic obstructive cardiomyopathy posted for adrenalectomy for pheochromocytoma. Indian Journal of Anaesthesia, 56(2), 198.
- Angelotti, T., Fuller, A., Rivera, L., & Schmiesing, C. (2005). Anesthesia for older patients with hypertrophic cardiomyopathy: is there cause for concern?. Journal ofclinical anesthesia, 17(6), 478-481.
- Bekker, A., Sorour, K., & Miller, S. (2002). The use of cardioselective β-blockers in a patient with idiopathic hypertrophic subaortic stenosis and chronic obstructive pulmonary disease. Journal of clinical anesthesia, 14(8), 589-591.
- Stone, J., & Ravalia, A. (2001). A patient with hypertrophic obstructive cardiomyopathy presenting for total knee replacement. Hospital Medicine, 62(5), 315-315.
- Freilich, J. D., & Jacobs, B. R. (1990). Anesthetic Management of Cerebral Aneurysm Resection in a Patient With Idiopathic Hypertrophic Subaortic Stenosis. Anesthesia & Analgesia, 71(5), 558-560.
- Prabhavathi, R., Reddy, P. N., Pathapati, R. M., & Rajashekar, S. T. (2014). Lithotripsy under low dose spinal anaesthesia with dexmedetomidine in a patient with hypertrophic obstructive cardiomyopathy. Indian Journal of Anaesthesia, 58(3), 360-362.
- Yamazaki, S., Omae, T., Koh, K., Sakuraba, S., Katsuda, Y., & Kumemura, M. (2019). Fixation of intracapsular fracture of the femoral neck using combined peripheral nerve blocks and transthoracic echocardiography in a patient with severe obstructive hypertrophic cardiomyopathy: a case report. JA Clinical Reports, 5(1), 1-4.
- Pai, R. V. B., Hegde, H. V., Santhosh, M. C. B., Roopa, S., Deshpande, S. S., & Rao, P. R. (2013). Bilateral brachial plexus blocks in a patient of hypertrophic obstructive cardiomyopathy with hypertensive crisis. Indian Journal of Anaesthesia, 57(1), 72.
- Maki N, Shimizu T, Nishiyama C, Murata A, Kita T. (2010). [Hypertrophic cardiomyopathy]. Masui. 59, 1010–2.
- Ho, A. M., Parlow, J., Allard, R., McMullen, M., & Mizubuti,G. B. (2020). Interscalene block in an anesthetized adult with hypertrophic obstructive cardiomyopathy undergoing clavicle fracture reduction. POCUS journal, 5(1), 6.
- Amagasa, S., Oda, S., Abe, S., Shinozaki, K., Miura, Y., Takaoka, S., & Horikawa, H. (2003). Perioperative management of lobectomy in a patient with hypertrophic obstructive cardiomyopathy treated with dual-chamber pacing. Journal of anesthesia, 17, 49-54.
- Yokoyama, N., Nishikawa, K., Takazawa, T., Saito, S., & Goto, F. (2004). Ventricular tachycardia induced by the change of position for epidural catheter insertion in a patient with hypertrophic obstructive cardiomyopathy. Masui. The Japanese Journal of Anesthesiology, 53(8), 910-913.
- Biladeau, S., & Grell, R. (2023). Severe hypertrophic cardiomyopathy with left ventricular outflow tract obstruction identified with preprocedural point-of-care ultrasound before emergent tracheal repair. BMJ Case Reports, 16(2), e254690.
- Lanier, W., & Prough, D. S. (1984). Intraoperative diagnosis of hypertrophic obstructive cardiomyopathy. Anesthesiology, 60(1), 61-63.
- Loubser, P., Suh, K., & Cohen, S. (1984). Adverse effects of spinal anesthesia in a patient with idiopathic hypertrophic subaortic stenosis. The Journal of the American Society of Anesthesiologists, 60(3), 228-229.
- Özdemir, M., Yanlı, P. Y., & Bakan, N. (2015). Anaesthesia Management of a Patient with Undiagnosed Hypertrophic Cardiomyopathy. Turkish Journal of Anaesthesiology and Reanimation, 43(4), 295.
- Fayad, A. (2007). Left ventricular outflow obstruction in a patient with undiagnosed hypertrophic obstructive cardiomyopathy. Canadian journal of anaesthesia, 54(12), 1019-1020.
- Olsen, K. R., LaGrew, J. E., Awoniyi, C. A., & Goldstein,J. C. (2018). Undiagnosed hypertrophic obstructive cardiomyopathy during transcatheter aortic valve replacement: a case report. Journal of Medical Case Reports, 12, 1-6.



