Research Article - (2022) Volume 1, Issue 1
An Extensive Evaluation and Meta-Analysis of Transcranial Magnetic Stimulations Effectiveness as a Treatment for Alzheimers Disease
Received Date: Sep 07, 2022 / Accepted Date: Oct 04, 2022 / Published Date: Nov 01, 2022
Copyright: ©Copyright: ©2022 Tshetiz Dahal. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Tshetiz Dahal. (2022). An Extensive Evaluation and Meta-Analysis of Transcranial Magnetic Stimulation's Effectiveness as a Treatment for Alzheimer's disease. World J Radiolo Img 1(1), 01-16.
Abstract
Background: For the treatment of Alzheimer's disease, repetitive transcranial magnetic stimulation (rTMS) is frequently employed. The impact of rTMS is still debatable, though. The current study's objective is to assess rTMS's impact on AD patients' cognitive function.
Methods: Before April 28, 2022, I conducted a thorough search of the pertinent literature in four main databases: PubMed, EMBASE, Web of Science, and the Cochrane Central Register of Controlled Trials [Central]. There were both cross-section studies and randomised controlled trials that compared the healing power of rTMS with no stimulation or sham stimulation.
Results: A total of 14 studies involving 513 AD patients were finally included for meta-analysis. It was found that rTMS significantly improved global cognitive function (SMD = 0.24, 95%CI, 0.12 to 0.36, P = 0.0001) and daily living ability (IADL: SMD = 0.64, 95%CI, 0.21to 1.08, P = 0.004) in patients with AD, but did not show improvement in language, memory, executive ability, and mood. In further analyses, rTMS at 10 Hz, on a single target with 20 sessions of treatment was shown to produce a positive effect. In addition, improvement in cognitive functions lasted for at least 6 weeks (SMD = 0.67, 95%CI, 0.05 to 1.30, P = 0.04).
Conclusion: rTMS can help AD patients with their everyday functioning and general cognition. Additionally, rTMS safety in AD patients with seizures needs to be taken into consideration. Our results should not be taken too seriously due to the limited sample size.
Keywords
Repetitive Transcranial Magnetic Stimulation, Alzheimer’s Disease, Cognitive Function, Activities of Daily Living, Therapeutic Efficacy.
Introduction
One of the main causes of reliance, disability, and death, demen- tia is an acquired, gradual cognitive impairment that interferes with daily tasks. There are currently over 44 million dementia patients worldwide, and as the ageing population grows, it is pre- dicted that number will triple by 2050 [1]. Alzheimer's disease (AD), which affects 50–75% of dementia sufferers, is the most common cause of dementia. After the age of 65, its prevalence substantially doubles every five years [2]. Clinically, it primar- ily shows itself as cognitive impairment, aberrant psychomotor activities, and social disengagement, all of which considerably raise the risk of emotional discomfort and detrimental effects on both physical and mental health [3].
There are now five drugs approved by the FDA to treat AD, including glutamate receptor antagonists (memantine) and ace- tylcholinesterase inhibitors [4]. Clinicians and researchers are searching for solutions in the area of non-pharmacological in- terventions due to the limited effectiveness of current pharma- cological therapies for recovering brain functions. Transcranial magnetic stimulation (TMS) is a non-invasive treatment that may enhance cognitive ability and neuroplasticity. It is being given more thought as a possible therapeutic approach to treat AD.
A non-invasive neuro-modulation approach is transcranial magnetic stimulation (TMS). Its magnetic pulses pass through the layer separating the skin's surface from the brain's surface. Variations in the magnetic field's strength produce electric fields that can stimulate particular parts of the brain [5]. According to Gangitano et al., it has the power to control not only the excit- ability of nerves and the operations of the cortices but also the activity of specific neurons [6, 7]. In order to treat depression, pain, fibromyalgia, post-traumatic stress disorder, non-fluent aphasia following stroke, and cognitive impairment, it has been frequently employed [8, 9]. Although numerous clinical trials and meta-analyses have examined the effectiveness and safety of repetitive TMS (rTMS) for patients with AD, no consensus has been reached [10-16]. The effects of single point stimula- tion, multi-point stimulation, the number of treatment sessions, and the combination of treatment with cognitive training on AD were not reported in a recent meta-analysis that summarised the findings from randomised controlled trials that were published in PubMed and Web of Science. In order to get a clear conclu- sion regarding the effectiveness of rTMS in managing AD from many angles, I therefore conducted this systematic review and meta-analysis on all RCT and cross-section studies published by April 28, 2022. Analysis was also done on how rTMS affected several cognitive domains as well as general cognitive function- ing.
Materials and Methods
The present study was conducted by complying with the PRIS- MA guideline for systematic evaluation and meta-analysis [17]. This study was registered at PROSPERO1.
Search Approach for the Literature
Using the following keywords: "Alzheimer's Disease" or "De- mentia of Alzheimer type" or "AD" and "transcranial magnetic stimulation" or "repetitive transcranial magnetic stimulation" or "TMS" or "rTMS" and ("randomized controlled trial" OR "controlled clinical trial" OR "cross-section" OR randomized OR placebo OR ", researcher (Tshetiz) independently searched literatures on PubMed, EMBASE, Web of Science, and the Ran- domized controlled trials and cross-section studies that were published before or on April 28, 2022, were chosen from among these articles. A senior investigator (Dr.Aditya) was invited to review the papers against the inclusion and exclusion criteria and endorse the final list of publications if there was any contra- diction with the researcher.
Eligibility Criteria
According to the National Institute of Neurological and Com- municative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA), all full-text randomised controlled studies and cross-section studies published in English, the Diagnostic and Statistical Manual of Mental Disorders 5th Edition (DSM V) or 4th Edition (DSM IV), and rTMS was administered to an age- and sex-neutral pop- ulation with an AD diagnosis were eligible. rTMS was the only different intervention for particular cortical regions, and sham rTMS in the same cortical regions was designated as the control conditions. Cognitive-related neurobehavioral outcomes includ- ed changes in any cognitive domain between pre- and post-inter- vention, as well as global cognition as assessed by objective (as opposed to subjective) cognitive measures.
When an article appeared in more than one database, the one with the greatest number of patients or the most comprehensive information was used. When relevant data were given at various times, the most recent time point's results were used.
Data Extraction
Authors, year of publication, research type, population charac- teristics (such as age, sex ratio, years of education, the course of the disease, diagnosis criteria), type and characteristics of stim- uli (including intensity, frequency, site, sessions of treatment), neurobehavioral outcomes [mean and standard deviation (SD), and adverse events—were all independently extracted by the researcher from each included study. Studies were mined for quality evaluations of the listed studies. We extracted chang- es in outcomes before and after treatment in a single group for cross-design clinical research. Results from clinical studies with several treatment groups were combined and analyzed. The re- spective authors were contacted to request the Mean and SD when they weren't provided in the text.
Quality Evaluation
The researcher evaluated the calibre of the methodologies used by the included studies in accordance with the evaluation criteria of the Cochrane risk of bias tool [18]. Any differences of opin- ion were settled through conversation or by bringing in a senior researcher. Each study's quality was evaluated in the following seven areas.
(1) creation of random sequences.
(2) concealing the allocation.
(3) Implementers and participants are rendered blind.
(4) Blinding the evaluation of the results.
(5) Reliability of the outcomes.
(6). Strict reporting
(7) Additional biases
Each aspect's bias risk was divided into three risk categories: low, high, and uncertain.
Data Analysis The CochraneRev-Man 5.4 software was used for statistical analysis of the data, calculating and reporting the standardized mean difference (SMD) and 95% confidence interval for each major outcome. SMD describes how much an intervention af- fects outcome, and an effect of ≥ 0.8 is considered significant and potentially clinically relevant. Reports on global cognitive functions (MMSE, MoCA, ADAS-Cog, ACE-III), language (sentence comprehension test), memory (RAVLT), the ability to execute (TMT-A), daily life ability (IADL) and emotion (GDS) were included in the meta-analysis. I2 was used to evaluate the heterogeneity of the included studies: when I2 < 50%, it was considered to have low heterogeneity, and the fixed-effect model was used. When I2 value ≥ 50%, it was considered to have high heterogeneity, and the random-effects model was used to sum- marize the effect size.
If data from at least ten studies were available, a meta-regression analysis was performed to assess the relationship between age, scale choice, frequency of stimulation, site of stimulation, and number of sessions of treatment and TMS treatment outcomes. STATA17.0 software was used to construct funnel plots for qual- itative evaluation of publication bias, Begg and Egger tests were used for quantitative evaluation. The data we used were chang- es in scores assessed using cognition assessment scales relative to baseline scores after completion of treatment. When score changes were not directly provided in the study, use the follow- ing formula to convert the data provided in the article: Mean change = mean final–mean baseline
In all analysis, p < 0.05 was considered statistically significant.
Results
Literature Screening
A total of 1,059 articles were found in 4 major databases us- ing a variety of searching strategies. A total of 263 publications were excluded due to duplicates or incomplete basic informa- tion. Another 754 publications were excluded because they were irrelevant to the condition we were interested in, basic scientific research, or non-randomized controlled studies. When the full text was inspected, 2 were excluded because of the single arm nature, 5 because of inconsistent control measures were taken, 2 because of inconsistent outcome indices, 4 because of meeting summaries or reviews, 8 because of incomplete data, another 7 because they were study protocols. Finally, 14 articles were selected for meta-analysis (Figure 1).
Figure 1: Diagram of literature searching and selection for meta-analysis.
Information of Included Studies
A total of 513 AD patients with varying degrees of severity were included in this meta-analysis. Their age ranged from 60 to 80 years and 53.5% of them were female. Demographic informa- tion of the participants was summarized in Table 1, types and characteristics of the stimuli in Table 2. Sham-rTMS was similar to the real rTMS in sound and feeling upon contacting with the head, but did not produce actual therapeutic effects. Generally, the coil was tilted away from the head to achieve the purpose of false stimulation. For example, in the study of, the same coil was tilted 45° from the scalp so that one side of the coil was in contact with the scalp and the distance between the center of the coil and the target site was greater than 5 cm [19]. Patients would also feel the noise and sensation caused by the same stim- ulation. In the study of, the coil was flipped 180°, where the coil was perpendicular to the scalp in the study of [20, 21]. In other studies, special fake coil was used, or the coil was attached to the scalp, but no therapeutic stimuli were applied.
Table 1: Demographic Characteristics of Included Trials.
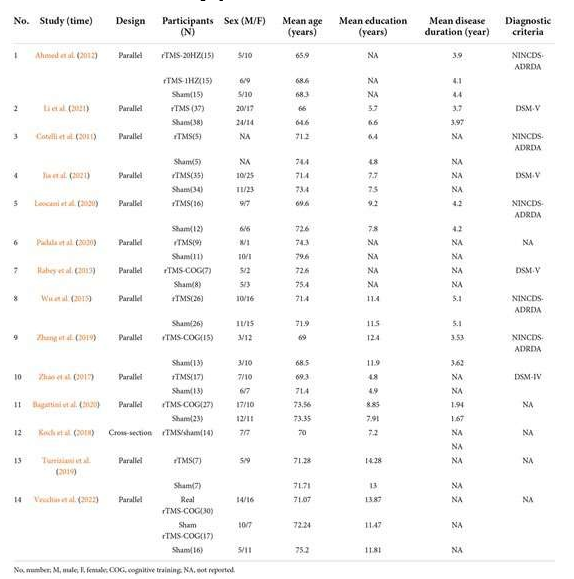
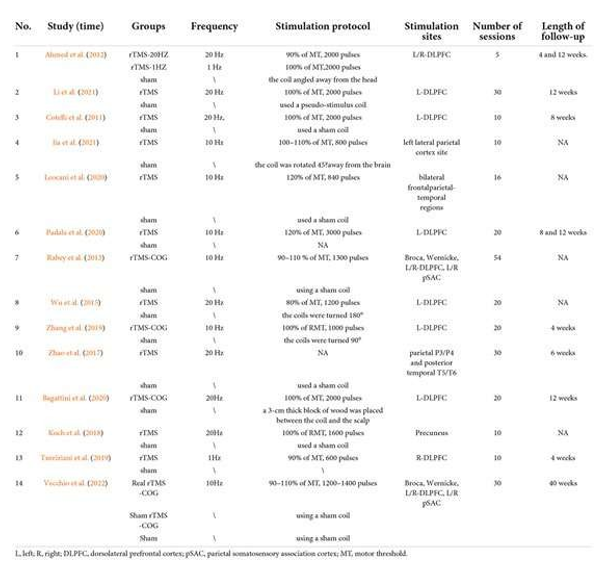
High frequency rTMS (HFrTMS) was dominantly used by in- cluded studies with 7 studies adopting high frequency up to 20 Hz [10-12, 20, 22, 22]. Studies by Ahmed et al compared the effects of high (20Hz) and low frequencies (1Hz) [11]. A study by Turriziani et al used 1 Hz, and the other 6 studies used 10 Hz [19, 24-28]. The dorsolateral prefrontal cortex (DLPFC) was selected as the stimulation site in the majority of studies (10/14) [10, 11, 20-25, 27, 28]. Rabey et al. and Vecchio et al. stimulated Broca, Wernicke, L/R-DLPFC, and L/R pSAC [26, 28]. Zhao et al. stimulated four parietal P3/P4 and posterior temporal T5/T6. Regarding the type of coil used for stimulation, the H-type coil was used in the study of Leocani et al. whereas the rest of the studies used the “8” shaped coil [26]. Among the 14 studies, the majority of them adopted 10 to 30 sessions, with a maximum of 54 and a minimum of 5 sessions.
A number of studies reported cognitive performance at fol- low-up ranging from 1 to 10 months. Four studies reported ad- verse reactions during the treatment [19, 21, 26,].
Table 2: Description of repetitive transcranial magnetic stimulation (rTMS) intervention in the included studies.
Quality of Included Studies
The quality of included studies was independently assessed us- ing the Revman software by two researchers. As shown in Table 2, only the study by Koch et al used the cross design, and the rest were randomized controlled studies [12]. These indicate that the overall quality of the included studies was good (Figure 2).
Figure 2: Analysis of risk of bias.
General Cognitive Functions and Repeated Transcranial Magnetic Stimulation
Thirteen studies assessed the impact of rTMS on global cog- nitive functions, namely, the Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA), Adden- brooke’s Cognitive Examination III (ACE-III) and the Alzhei- mer’s Disease Assessment Scale-Cognitive Sub-scale (ADAS- Cog). Among these studies, 3 used the MMSE and ADAS-cog scales to assess patients’ global cognitive status, 6 studies count- ed MMSE results, 4 studies counted ADAS-cog results [10-12, 19, 20-23, 25-28]. The above findings showed that rTMS sig- nificantly improved global cognitive functions (measured by MMSE or ADAS-Cog) in AD patients (SMD = 0.34,95% CI, 0.16 to 0.52, p = 0.0002, I2 = 0%, Figure 3). When subgroup analysis was conducted to discuss MMSE and ADAS-cog, respectively, rTMS still had a significant effect on the global cognitive functions of AD patients (MMSE: SMD = 0.59, 95%CI, 0.36 to 0.81, I2 = 87%; ADAS-cog: SMD = –0.34, 95%CI, –0.59 to –0.10, I2 = 42%) (Figure 3). It should be noted that the MMSE score is a positive indicator, and the higher the score, the better the cognitive status of patients.
Figure 3: The effect of repetitive transcranial magnetic stimulation (rTMS) on global cognitive functions.
While ADAS-cog is a negative indicator, the lower the score, the better the cognitive status of patients. In our forest map, the ab- scissa is defined according to the MMSE score, that is, the left is the sham stimulation group, and the right is the rTMS treatment group. However, for the subgroup analysis of ADAS-cog, we ac- curately calculated the results of each study, so the SMD values of subgroups that fell on the left indicated that the rTMS treat- ment was effective. In particular, Zhao et al. additionally, used the MoCA scale to show that rTMS had a significant treatment effect [rTMS change (Mean ± SD): 2.3 ± 6.36; sham change (Mean ± SD): 1.2 ± 7.02]. Zhang et al.also observed more sig- nificant results using the ACE-III scale [rTMS change (Mean ± SE): 11.77 ± 1.32; Sham change (Mean ± SE): 2.18 ± 1.43] [21].
Inconsistencies exist in the rTMS intervention parameters among the included studies, and we conducted subgroup analyses on stimulation frequency, stimulation sites, and the number of ses- sions of treatment. As shown in Figure 4, compared with rTMS at 20 Hz, stimulation frequency at 10 Hz had a more significant effect (SMD = 0.29, 95%CI, 0.01 to 0.57, P = 0.04). When ana- lyzing the influence of stimulating loci, it was found that single stimulation of L-DLPFC (SMD = 0.84, 95%CI, 0.08 to 1.59, P = 0.03) was more effective than multi-point stimulation (SMD = 0.30, 95%CI, 0.00 to 0.61, P = 0.05) (Figure 5).
In addition, the therapeutic effect was most significant when the number of treatment sessions was 20 (SMD = 0.61, 95%CI, 0.28 to 0.95, P = 0.0003), followed by treatment sessions over 20 (SMD = 0.39, 95%CI, 0.07 to 0.70, P = 0.02). No statistical difference was observed when the number of treatment sessions was less than 20 (SMD = 0.16, 95%CI, –0.12 to 0.45, P = 0.26) (Figure 6). Some of the studies in the meta-analysis also provid- ed participants with supplemental cognitive training when as- sessing the impact of rTMS on global cognition, but the content of cognitive training varied between studies [21, 22, 25, 27].
Figure 4: Impact of repetitive transcranial magnetic stimulation (rTMS) stimulation frequency on global cognitive functions.
Figure 5: Impact of repetitive transcranial magnetic stimulation (rTMS) stimulation site of rTMS on global cognitive functions.
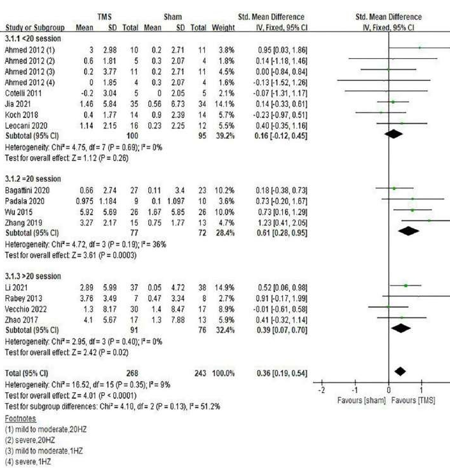
Figure 6: Influence of sessions of repetitive transcranial magnetic stimulation (rTMS) treatment on global cognitive functions.
Figure 7: Influence of repetitive transcranial magnetic stimulation (rTMS) combined cognitive training on global cognitive func- tions
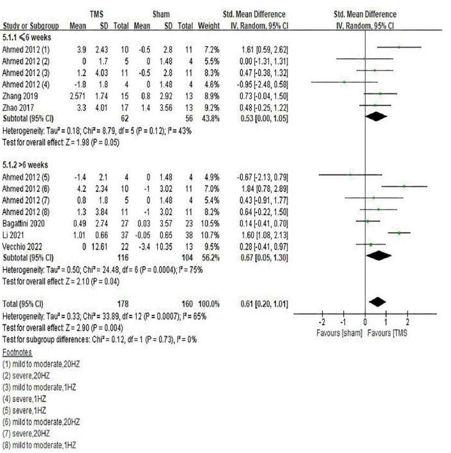
Figure 8: Post-treatment effects of repetitive transcranial magnetic stimulation (rTMS) on global cognitive functions.
My Results showed that rTMS combined with cognitive training had no statistically. significant effect on global cognitive func- tions (SMD = 0.48, 95%CI, –0.08 to 1.05, P = 0.09), but rTMS without combined cognitive training seemed to be superior to the former in improving global cognitive functions. The statis- tical significance of the results remains to be explored (SMD = 0.54, 95%CI, 0.01 to 1.06, P = 0.05) (Figure 7). Among the included studies, 6 reported post-treatment effects [11, 21-23, 27]. Results showed that rTMS had a better effect when the fol- low-up time was > 6 weeks (SMD = 0.67, 95%CI, 0.05 to 1.30, P = 0.04) than ≤6 weeks (SMD = 0.53, 95%CI, 0.00 to 1.05, P = 0.05) (Figure 8).
Influence of Repetitive Transcranial Magnetic Stimulation on Daily Living Among the studies we included, 3 analyzed daily living using the IADL scale [10, 12, 28]. Their results showed that rTMS could significantly improve the daily living ability of AD pa- tients (SMD = 0.64, 95%CI: 0.21–1.08, P = 0.007) (Figure 9).
Figure 9: Forest plot showing the effect of repetitive transcranial magnetic stimulation (rTMS) on IADL of AD patients with 95%CI.
Impact of Repetitive Transcranial Magnetic Stimulation on Language
In the study of Cotelli et al, the sentence comprehension test was used to evaluate the listening comprehension of AD patients, and the results showed that rTMS had a significant therapeutic effect on language [10]. In contrast, in the study of Jia et al. the language results of the MMSE scale did not show significant improvement [19]. In the study by Zhang et al [21], although the therapeutic effect of rTMS on language was observed by the ACE-III scale, the result was not statistically significant (P = 0.08). In addition, Zhao et al. showed significant improvement in speech function in patients treated with rTMS, and the results were statistically significant (P = 0.003). However, it was not clearly explained in this study how the language function score was obtained. According to the research content of Zhao et al. we inferred that the conclusion was obtained by comprehensive calculation of the results of ADAS-cog, MMSE, MoCA and WHO-UCLA AVLT scale.
Impact of Repetitive Transcranial Magnetic Stimulation on Memory
Koch et al and Bagattini et al used RAVLT-IR and RAVLT-DR tests to evaluate the memory function of patients [12, 22]. Since both tests belong to the RAVLT sub-scale and they are strongly correlated, so the results of these two tests were combined for analysis. However, the rTMS group did not show superiority to the sham stimulation group (SMD = 0.22, 95%CI, –0.09 to 0.54, P = 0.17) (Figure 10). In addition, Turriziani et al. found that AD patients did not show any difference in the Rey’s 15 words immediate recall and Rey’s 15 words 15-min delayed recall [24]. However, a study by Zhang et al. showed that rTMS significant- ly improved the memory function in patients who received cog- nitive training [real TMS-CT change (Mean ± SE): 3.87 ± 0.82; Sham TMS-CT change (Mean ± SE): 0.29 ± 1.07]. A note is that this conclusion was drawn from ACE-III results [21].
Figure 10: Forest plot showing the effect of repetitive transcranial magnetic stimulation (rTMS) on RAVLT of AD patients with 95% CI.
Impact of Repetitive Transcranial Magnetic Stimulation on Executive Ability
In studies by Bagattini et al. and Padala et al., rTMS showed no positive effect when the “Visuospatial Ability” and “Writing Movement Speed” were evaluated using the TMT A test (SMD = 0.02, 95%CI, –0.45 to 0.50, P = 0.93) (Figure 11) [22, 28]. However, a positive trend was observed in a study by Zhang et al. on attention and visuospatial function assessed using the ACE-III scale [21].
Figure 11: Forest plot showing the effect of repetitive transcranial magnetic stimulation (rTMS) on TMT-A of AD patients with 95% CI.
Influence of Repetitive Transcranial Magnetic Stimulation on Emotion
Studies by Ahmed et al. and Bagattini et al. assessed psycho- logical and emotional changes of AD patients using the GDS scale [12, 22]. It can be seen from the forest plot, there was no statistical difference in psychological and emotional improve- ment between the TMS treatment group and the sham stimula- tion group (SMD: –0.16, 95%CI: –0.54â?»0.22, P = 0.41) (Figure 12). In another study by Padala et al., Clinical Global Impres- sion-improvement (CGI-I) and Clinical Global Impression-Se- verity (CGI-S) were used to assess the overall mental state of AD patients [28]. It was found that rTMS treatment significantly improved mental state as shown by CGI-S [1.4 (0.5 to 2.3), P = 0.005]. In the rTMS group, CGI-S was significantly better com- pared to that of the baseline (P < 0.001), whereas no significant difference was found between the baseline and after sham stim- ulation (P = 0.238) in the sham group. In contrast, when CGI-I was used to assess psychological and emotional changes [–2.56 (–3.5 to –1.6), P < 0.001], both rTMS and sham stimulation sig- nificantly improved CGI-I compared to the baseline.
Figure 12: Forest plot showing the effect of repetitive transcranial magnetic stimulation (rTMS) on GDS of AD patients with 95%CI.
Publication Bias and Sensitivity Analysis
I firstly used funnel plots to qualitatively assess publication bias, and our results showed that funnel plots were visually symmetric (Figure 13). Then we conducted quantitative evaluation through Begg and Egger tests, and results showed that publication bias was present [Begg (P = 0.0748) and Egger (P = 0.0040)]. Therefore, pruning and filling tests were used for verification. After pruning and filling, our results did not change, suggesting that they were still robust (Figure 13). In addition, it should be pointed out that the methods of random sequence generation and concealment in the study by Cotelli et al. we’re not reported, suggesting a highly potential bias risk (Figure 2) [10]. After excluding this study, the influence of rTMS on AD patients (SMD = 0.57; 95% CI = 0.39, 0.76; P < 0.00001, I2 = 83%) was still significantly higher than those in the sham stimulation group. We then conducted a sensi- tivity analysis using the leave one method, interactively deleting each study and recalculating the summary SMD.
The data showed that the heterogeneity of the results changed significantly when the study of Li et al. was excluded alone [23]. But the results were still statistically significant (SMD = 0.36, 95%CI = 0.17–0.55; P = 0.0002, I2 = 54%). Meta-regression analysis was used to explore the source of heterogeneity, and the results showed that sample size, age and TMS treatment effect were not significant at the level of 5%, that is, sample size and age were not considered as the source of heterogeneity (sam- ple size: P = 0.177; age: P = 0.952). Scale selection and TMS treatment effect were significant at the level of 1%, stimulation frequency was significant at the level of 5%. We have conducted subgroup analysis on rTMS stimulation frequency, number of stimulation sites, and number of sessions using different cogni- tive scales.
Figure 13: Funnel plot for publication bias and result of clipping test.
Adverse Reactions
Adverse reactions were reported in 4 of 14 studies [19, 21, 26, 28,]. The main adverse reactions included local scalp discomfort or headache. In a study by Jia et al., two patients (one in the rTMS group and the other in the sham group) reported transient fatigue. It has to be mentioned that no case of seizure was re- corded [19].
Discussion
The improvement of cognitive function following rTMS treat- ment or sham stimulation was compared in 14 studies, encom- passing a total of 513 AD patients with varied degrees of dis- ease severity. In comparison to sham stimulation, we discovered that rTMS dramatically enhanced global cognitive abilities. We discovered that rTMS was beneficial in enhancing daily living capacity when each cognitive domain was included for further investigation. Our findings are in line with the majority of oth- er studies showing that rTMS improves cognition and everyday functioning in AD patients. rTMS can enhance the plasticity and excitability of the cerebral cortex, which will subsequently im- prove the cognitive performance of AD patients [29]. Previous research has shown that AD patients' cortical plasticity is im- paired, which causes a decline in cognitive functions and self- care ability [30, 31]. Although neural plasticity could not be tested in our investigation, the enhanced performance in some domains and across the board suggests that rTMS may enhance brain plasticity. Cognitive impairment includes alterations in executive, visuospatial, speech, memory and other aspects. Our meta-analysis showed that rTMS can improve global cognitive functions in patients with AD, which is consistent with findings of a previous meta-analysis study [14]. Thirteen of our included studies assessed global cognitive functions in patients with AD using scales, such as MMSE, MoCA, ACE-III and ADAS-cog with consistent and positive results [10, 11, 14, 18, 20-23, 25-28] Even though individual studies in the research we considered had good findings in language, memory, executive function, and affective cognition, the pooled findings were not statistically significant. Language is a tool for symbolically communicat- ing. Grammar, semantics, and pronunciation are the three facets of language. In human social contact, verbal communication is crucial. One of the most prevalent functional deficits in cogni- tive decline is language impairment, which first manifests in the early stages of AD. Performance on language tasks is a crucial diagnostic indicator for AD and moderate cognitive impairment. Fluency in language, naming, semantic awareness, and dis- course processing are the key areas where it shows up [32]. The language assessment scale, which includes the aphasia screening test, Boston Naming test, Word Fluency test, double-listening test, expression vocabulary test, Peabody Graph word test, adult reading test, marking test, and language test of the Wechsler In- telligence Test, is a frequently used technique for evaluating lan- guage disorders. Few studies have examined the effects of rTMS on the language cognitive domain in AD patients, and the study's assessment techniques varied, which made it challenging to per- form additional statistical analysis. Therefore, we described the relevant articles one by one. Cotelli et al. evaluated the patients’ listening comprehension ability through the Sentence Compre- hension test and obtained positive results [10]. However, in the study of Zhang et al. and Jia et al., the results of language func- tion measured by MMSE and ACE-III, respectively, were not statistically significant [19, 21]. In the study of memory cogni- tive domain, Koch et al. and Bagattini et al. used RAVLT as an evaluation tool to conduct the study, and the results showed that there was no significant statistical difference between the TMS treatment group and the sham stimulation control group [12, 22]. In terms of executive ability, Bagattini et al. and Padala et al. did not show positive effects of rTMS when TMT A test was used to evaluate “visuospatial ability” and “writing motor speed” [22, 28] However, Zhang et al. observed positive results in “atten- tion” and “visuospatial ability” using the ACE-III scale [21].
Age-related difficulties, such as diminished activities of daily living (ADL) and instrumental ADL (IADL), account for 35% of the population aged 65 and over and may make it challenging for senior persons to live independently [33]. According to Koyano et al., IADL refers to a person's capacity for using public trans- portation, purchasing daily necessities, preparing meals, paying bills, managing bank accounts, and other activities [34]. It also refers to a person's level of autonomy and independence with re- gard to carrying out activities at home and in the community. In terms of daily living ability, positive results were found in IADL indicators in studies of Cotelli et al., Ahmed et al., and Padala et al. [10, 11, 28].
Previous studies have found that rTMS treatment has definite effects on depression and other psychiatric disorders [35]. In a study on the effect of rTMS on patients with treatment-refrac- tory depression, positive response of patients was related to the increased volume of the left amygdala and the unchanged volume of the hippocampus, whereas the neutral response was associated with the decreased volume of the left hippocampus [36]. Another study reported that an increase in the volume of that the hippocampus on the side of that the brain targeted by HFrTMS was associated with improvement in depression [37]. However, when we analyzed combined results of GDS indica- tors of Ahmed et al. and Bagattini et al., we did not obtain posi- tive results that rTMS treatment could improve the mood of AD patients [11, 22]. Given the small number of included studies and the different scales used in different studies, it is necessary to conduct high-quality larger-sample RCT studies in the future to confirm our findings.
Our meta-analysis also looked at the impact of various rTMS pa- rameters on cognitive performance and discovered that L-DLP- FC, 10 Hz rTMS, and at least 20 sessions were all helpful in enhancing cognitive abilities. Our findings corroborate earli- er research regarding the effects of various rTMS stimulation frequencies, locations, and sessions on cognitive performance [38]. The excitability of particular regions of the cerebral cor- tex can be increased or decreased by rTMS due to variations in the stimulation parameters [38, 39]. The results of our study suggest that 10 Hz rTMS has a more beneficial therapeutic ef- fect on cognitive functioning than HFrTMS, which may raise brain cell excitability with a ceiling effect. Future high-quality larger-sample RCT studies are required to reach an exclusive conclusion since the opposite finding from a meta-analysis re- search by Wang et al., which asserted that 20 Hz rTMS appeared to be more effective than 10 Hz or 1 Hz rTMS, is needed [15]. Prior to using rTMS, stimulating locations must be chosen. The L-DLPFC and the Precuneus were the two most frequently used sites for single-site stimulation [12, 40]. As was already indicat- ed, 10 Hz had better efficacy than 20 Hz, indicating a connection between the frequency of rTMS and the stimulation site. The left and right DLPFC are the most frequently used candidates for multisite stimulation. Whether bilateral DLPFC stimulation is superior to unilateral DLPFC stimulation remains controver- sial. In a meta-analysis published by Liao et al. involving 94 patients with mild to moderate AD, it was found that stimulation of the right or bilateral DLPFC was more superior to stimulation of the L-DLPFC alone. In contrast, meta-analysis by Drumond Marra et al. and Wu et al. found that stimulation of the L-DLP- FC by HFrTMS was more effective in improving cognitive per- formance than multi-point stimulation. Cotelli et al. found that stimulation of the L-DLPFC significantly increased the percent- age of correct response in auditory sentence comprehension, but there was no significant difference in other language abilities or memory [5, 10, 20, 41]. This study only included 10 patients. To draw a clear conclusion on the efficacy of different parameters of rTMS in improving cognitive performance in diverse domains, the long-term results of studies adopting multisite stimulation are needed.
In addition, we also conducted a subgroup analysis on the effect of cognitive training added to rTMS, and we found that cogni- tive training did not show additive effects when applied with rTMS. But the cognitive training varied from study to study. For example, Zhang et al. adopted cognitive training in areas of memory tasks, attention tasks, mathematical calculations, agility drills and logic thinking tasks [21]. Rabey et al. and Vecchio et al. applied cognitive training to test the impact of Broca re- gion, Wernicke region, R-DLPFC, L-DLPFC, R-PSAC, and the L-PSAC on cognitive performance [25, 27]. Cognitive training in the study by Bagattini et al. focused on episodic memory and specifically on face-name associative memory [22]. Although the above studies separately described the positive results of rTMS combined with cognitive training, our results did not show statistical significance in our subgroup analysis (SMD = 0.48, 95%CI: –0.08 to 1.05, P = 0.09). Therefore, future studies should balance the number of AD patients receiving the com- bined cognitive training and those who do not receive this com- bined therapy, in order to better investigate whether cognitive training leads to synergistic effects.
In the study of Zhang et al., MMSE and ACE-III indicators were used to observe the effect of rTMS 4 weeks after rTMS treat- ment, and the results showed that patients treated with rTMS still maintained significant improvement in cognitive functions [21]. Zhao et al. reported the effect of rTMS 6 weeks after rTMS treatment, three cognitive domains of memory, language, and ex- ecutive ability were individually analyzed based on ADAS-cog, MMSE, MoCA, and WHO-UCLA AVLT indicators. The results showed that memory function improved the most, followed by language function. Ahmed et al. reported the effect of rTMS on AD patients 1 and 3 months after rTMS treatment, respectively [11]. MMSE results showed that mild or moderate AD patients who received HFrTMS stimulation benefited the most. Positive results of long-term therapeutic effects of rTMS were reported in studies by Bagattini et al., Li et al., and Vecchio et al. [20, 23, 27]. Their follow-up duration was 2, 3, and 4 months, respec- tively. In our subgroup analysis, patients who received rTMS still maintained good cognitive function improvement within 6 weeks after the end of treatment, but the improvement effect was more apparent after over 6 weeks, evidenced by results of Ahmed et al. [11].
This study was conducted on the basis of the study by Lin et al., apart from global cognitive functions, different cognitive domains such as language, memory, executive ability, daily liv- ing ability and emotion, as well as rTMS parameters were an- alyzed, and compared at different follow-up time points [14]. The forest maps of MMSE and ADAS-cog were drawn for anal- ysis. In terms of publication bias and sensitivity analysis, we conducted more comprehensive validation and discussion, and the results were robust. Meta-regression analysis was used to explore the source of heterogeneity, and the results suggested that there was no correlation between sample size, age, and the therapeutic effect of rTMS. Some limitations should be consid- ered when interpreting our results. The use of different scales to measure global cognitive functions in different studies may lead to high heterogeneity. Meta-regression analysis suggested that scale selection was significantly associated with TMS treatment effect at the level of 1% and stimulus frequency at the level of 5%. Among the included studies, only 6 explored the therapeu- tic effect 1 to 3 months after completing the treatment, longer follow-up period is needed to examine the long-term therapeutic effect of rTMS on AD in the future.
Conclusion
Patients with AD can have their everyday functioning and gener- al cognitive abilities improved with repetitive transcranial mag- netic stimulation (rTMS). rTMS significantly enhanced cogni- tion with 20 sessions or more at a single site at high frequency, according to subgroup analysis. Furthermore, we need to focus more on the rTMS safety in AD patients with seizures. Our re- sults should not be taken too seriously due to the limited sample size. To more accurately determine the effectiveness of rTMS in treating AD patients and the ideal type of stimulation needed to enhance positive outcomes, additional research with bigger sam- ple numbers and longer follow-up periods are required.
Conflict of interest
The authors declare that the research was conducted in the ab- sence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Lane, C. A., Hardy, J., and Schott, J. M. (2018). Alzheimer’s disease. Eur. J. Neurol. 25, 59–70.
- Alzheimer’s Disease International. (2014). World Alzhei- mer Report 2014: Dementia and Risk Reduction: An Analy- sis of Protective and Modifiable Factors.
- Alzheimer’s Association (2021). Alzheimer’s disease facts and figures. Alzheimers Dement 17, 327–406.
- Raina, P., Santaguida, P., Ismaila, A., Patterson, C., Cow- an, D., Levine, M., ... & Oremus, M. (2008). Effectiveness of cholinesterase inhibitors and memantine for treating de- mentia: evidence review for a clinical practice guideline. Annals of internal medicine, 148(5), 379-397.
- Liao, X., Li, G., Wang, A., Liu, T., Feng, S., Guo, Z., ... & Mu, Q. (2015). Repetitive transcranial magnetic stimulation as an alternative therapy for cognitive impairment in Alz- heimer’s disease: a meta-analysis. Journal of Alzheimer's Disease, 48(2), 463-472.
- Gangitano, M., Valero-Cabré, A., Tormos, J. M., Mottaghy,F. M., Romero, J. R., & Pascual-Leone, Á. (2002). Mod-ulation of input–output curves by low and high frequency repetitive transcranial magnetic stimulation of the motor cortex. Clinical Neurophysiology, 113(8), 1249-1257.
- Mueller, J. K., Grigsby, E. M., Prevosto, V., Petraglia, F. W., Rao, H., Deng, Z. D., ... & Grill, W. M. (2014). Simultane- ous transcranial magnetic stimulation and single-neuron re- cording in alert non-human primates. Nature neuroscience, 17(8), 1130-1136.
- Lefaucheur, J. P., Aleman, A., Baeken, C., Benninger, D. H., Brunelin, J., Di Lazzaro, V., ... & Ziemann, U. (2020). Evidence-based guidelines on the therapeutic use of repet- itive transcranial magnetic stimulation (rTMS): an update (2014–2018). Clinical neurophysiology, 131(2), 474-528.
- Trung, J., Hanganu, A., Jobert, S., Degroot, C., Mejia-Con- stain, B., Kibreab, M., ... & Monchi, O. (2019). Transcra- nial magnetic stimulation improves cognition over time in Parkinson's disease. Parkinsonism & Related Disorders, 66, 3-8.
- Cotelli, M., Calabria, M., Manenti, R., Rosini, S., Zanetti, O., Cappa, S. F., & Miniussi, C. (2011). Improved language performance in Alzheimer disease following brain stimu- lation. Journal of Neurology, Neurosurgery & Psychiatry, 82(7), 794-797.
- Ahmed, M. A., Darwish, E. S., Khedr, E. M., & Ali, A. M. (2012). Effects of low versus high frequencies of repetitive transcranial magnetic stimulation on cognitive function and cortical excitability in Alzheimer’s dementia. Journal of neurology, 259(1), 83-92.
- Koch, G., Bonnì, S., Pellicciari, M. C., Casula, E. P., Manci- ni, M., Esposito, R., ... & Bozzali, M. (2018). Transcranial magnetic stimulation of the precuneus enhances memory and neural activity in prodromal Alzheimer's disease. Neu- roimage, 169, 302-311.
- Dong, X., Yan, L., Huang, L., Guan, X., Dong, C., Tao, H.,... & Wan, Q. (2018). Repetitive transcranial magnetic stim- ulation for the treatment of Alzheimer's disease: a systemat- ic review and meta-analysis of randomized controlled trials. PLoS One, 13(10), e0205704.
- Lin, Y., Jiang, W. J., Shan, P. Y., Lu, M., Wang, T., Li, R.H., ... & Ma, L. (2019). The role of repetitive transcranial magnetic stimulation (rTMS) in the treatment of cognitive impairment in patients with Alzheimer's disease: a system- atic review and meta-analysis. Journal of the neurological sciences, 398, 184-191.
- Wang, X., Mao, Z., Ling, Z., & Yu, X. (2020). Repetitive transcranial magnetic stimulation for cognitive impairment in Alzheimer's disease: a meta-analysis of randomized con- trolled trials. Journal of Neurology, 267(3), 791-801.
- Teselink, J., Bawa, K. K., Koo, G. K., Sankhe, K., Liu, C. S., Rapoport, M., ... & Lanctôt, K. L. (2021). Efficacy of non-invasive brain stimulation on global cognition and neu- ropsychiatric symptoms in Alzheimer’s disease and mild cognitive impairment: A meta-analysis and systematic re- view. Ageing Research Reviews, 72, 101499.
- McInnes, M. D., Moher, D., Thombs, B. D., McGrath, T. A., Bossuyt, P. M., Clifford, T., ... & Willis, B. H. (2018). Preferred reporting items for a systematic review and me- ta-analysis of diagnostic test accuracy studies: the PRIS- MA-DTA statement. Jama, 319(4), 388-396.
- Sterne, J. A., Savovic, J., Page, M. J., Elbers, R. G., Blen-cowe, N. S., Boutron, I., ... & Higgins, J. P. (2019). RoB 2: a revised tool for assessing risk of bias in randomised trials. bmj, 366.
- Jia, Y., Xu, L., Yang, K., Zhang, Y., Lv, X., Zhu, Z., ... &Chen, W. (2021). Precision repetitive transcranial magnetic stimulation over the left parietal cortex improves memory in Alzheimer’s disease: a randomized, double-blind, sh- am-controlled study. Frontiers in Aging Neuroscience, 348.
- Yue, W. U., Wenwei, X. U., Xiaowei, L. I. U., Qing, X. U., Li, T. A. N. G., & Shuyan, W. U. (2015). Adjunctive treat- ment with high frequency repetitive transcranial magnetic stimulation for the behavioral and psychological symptoms of patients with Alzheimer's disease: a randomized, dou- ble-blind, sham-controlled study. Shanghai archives of psy- chiatry, 27(5), 280.
- Zhang, F., Qin, Y., Xie, L., Zheng, C., Huang, X., & Zhang,M. (2019). High-frequency repetitive transcranial magnet- ic stimulation combined with cognitive training improves cognitive function and cortical metabolic ratios in Alzhei- mer’s disease. Journal of Neural Transmission, 126(8), 1081-1094.
- Bagattini, C., Zanni, M., Barocco, F., Caffarra, P., Brignani, D., Miniussi, C., & Defanti, C. A. (2020). Enhancing cog- nitive training effects in Alzheimer’s disease: rTMS as an add-on treatment. Brain Stimulation, 13(6), 1655-1664.
- Li, X., Qi, G., Yu, C., Lian, G., Zheng, H., Wu, S., ... &Zhou, D. (2021). Cortical plasticity is correlated with cog- nitive improvement in Alzheimer’s disease patients after rTMS treatment. Brain Stimulation, 14(3), 503-510.
- Turriziani, P., Smirni, D., Mangano, G. R., Zappalà, G., Gi- ustiniani, A., Cipolotti, L., & Oliveri, M. (2019). Low-fre- quency repetitive transcranial magnetic stimulation of the right dorsolateral prefrontal cortex enhances recognition memory in Alzheimer’s disease. Journal of Alzheimer's Disease, 72(2), 613-622.
- Rabey, J. M., Dobronevsky, E., Aichenbaum, S., Gonen, O., Marton, R. G., & Khaigrekht, M. (2013). Repetitive transcranial magnetic stimulation combined with cognitive training is a safe and effective modality for the treatment of Alzheimer’s disease: a randomized, double-blind study. Journal of Neural Transmission, 120(5), 813-819.
- Leocani, L., Dalla Costa, G., Coppi, E., Santangelo, R., Pisa, M., Ferrari, L., ... & Comi, G. (2021). Repetitive Tran- scranial Magnetic Stimulation With H-Coil in Alzheimer's Disease: A Double-Blind, Placebo-Controlled Pilot Study. Frontiers in neurology, 11, 614351.
- Vecchio, F., Quaranta, D., Miraglia, F., Pappalettera, C., Di Iorio, R., L’Abbate, F., ... & Rossini, P. M. (2022). Neu- ronavigated Magnetic Stimulation combined with cogni- tive training for Alzheimer’s patients: an EEG graph study. Geroscience, 44(1), 159-172.
- Padala, P. R., Boozer, E. M., Lensing, S. Y., Parkes, C. M.,Hunter, C. R., Dennis, R. A., ... & Padala, K. P. (2020). Neuromodulation for apathy in Alzheimer’s disease: a dou- ble-blind, randomized, sham-controlled pilot study. Journal of Alzheimer's Disease, 77(4), 1483-1493.
- Pennisi, G., Ferri, R., Lanza, G., Cantone, M., Pennisi, M., Puglisi, V., ... & Bella, R. (2011). Transcranial magnetic stimulation in Alzheimer’s disease: a neurophysiologi-cal marker of cortical hyperexcitability. Journal of NeuralTransmission, 118(4), 587-598.
- Di Lorenzo, F., Motta, C., Casula, E. P., Bonnì, S., Assogna, M., Caltagirone, C., ... & Koch, G. (2020). LTP-like cortical plasticity predicts conversion to dementia in patients with memory impairment. Brain Stimulation, 13(5), 1175-1182.
- Di Lorenzo, F., Motta, C., Bonnì, S., Mercuri, N. B., Calt- agirone, C., Martorana, A., & Koch, G. (2019). LTP-like cortical plasticity is associated with verbal memory impair- ment in Alzheimer's disease patients. Brain stimulation, 12(1), 148-151.
- Taler, V., & Phillips, N. A. (2008). Language performance in Alzheimer's disease and mild cognitive impairment: a comparative review. Journal of clinical and experimental neuropsychology, 30(5), 501-556.
- Kiyoshige, E., Kabayama, M., Gondo, Y., Masui, Y., In- agaki, H., Ogawa, M., ... & Kamide, K. (2019). Age group differences in association between IADL decline and de- pressive symptoms in community-dwelling elderly. BMC geriatrics, 19(1), 1-8.
- Kiyoshige, E., Kabayama, M., Gondo, Y., Masui, Y., In- agaki, H., Ogawa, M., ... & Kamide, K. (2019). Age group differences in association between IADL decline and de- pressive symptoms in community-dwelling elderly. BMC geriatrics, 19(1), 1-8.
- Health Quality Ontario. (2016). Repetitive transcranial magnetic stimulation for treatment-resistant depression: a systematic review and meta-analysis of randomized con- trolled trials. Ontario health technology assessment series, 16(5), 1.
- Furtado, C. P., Hoy, K. E., Maller, J. J., Savage, G., Das- kalakis, Z. J., & Fitzgerald, P. B. (2013). An investigation of medial temporal lobe changes and cognition following antidepressant response: a prospective rTMS study. Brain stimulation, 6(3), 346-354.
- Hayasaka, S., Nakamura, M., Noda, Y., Izuno, T., Saeki, T., Iwanari, H., & Hirayasu, Y. (2017). Lateralized hippocam- pal volume increase following highâ?ÂÃÂ?ÂÂfrequency left prefron- tal repetitive transcranial magnetic stimulation in patients with major depression. Psychiatry and Clinical Neurosci- ences, 71(11), 747-758.
- Nguyen, J. P., Suarez, A., Kemoun, G., Meignier, M., Le Saout, E., Damier, P., ... & Lefaucheur, J. P. (2017). Re- petitive transcranial magnetic stimulation combined with cognitive training for the treatment of Alzheimer's disease. Neurophysiologie Clinique/Clinical Neurophysiology, 47(1), 47-53.
- Jiang, W., Wu, Z., Wen, L., Sun, L., Zhou, M., Jiang, X., & Gui, Y. (2022). The Efficacy of High-or Low-Frequency Transcranial Magnetic Stimulation in Alzheimer’s Disease Patients with Behavioral and Psychological Symptoms of Dementia. Advances in Therapy, 39(1), 286-295.
- Nardone, R., Tezzon, F., Höller, Y., Golaszewski, S., Trin- ka, E., & Brigo, F. (2014). Transcranial magnetic stimula- tion (TMS)/repetitive TMS in mild cognitive impairment and Alzheimer's disease. Acta Neurologica Scandinavica, 129(6), 351-366.
- Marra, H. L. D., Myczkowski, M. L., Memória, C. M., Ar-naut, D., Ribeiro, P. L., Mansur, C. G. S., ... & Marcolin,M. A. (2015). Transcranial magnetic stimulation to address mild cognitive impairment in the elderly: a randomized con- trolled study. Behavioural neurology.
- De Risio, L., Borgi, M., Pettorruso, M., Miuli, A., Ottoma- na,A. M., Sociali, A., ... & Zoratto, F. (2020). Recovering from depression with repetitive transcranial magnetic stim- ulation (rTMS): a systematic review and meta-analysis of preclinical studies. Translational psychiatry, 10(1), 1-19.




