Case Report - (2021) Volume 3, Issue 1
Acute Medical (bedside) Peritoneal Dialysis catheter insertion in Intensive Therapy Unit setting, an overlooked Renal Replacement Therapy option, A Case Study
Received Date: Dec 07, 2021 / Accepted Date: Dec 14, 2021 / Published Date: Dec 20, 2021
Copyright: ©Michael Fawzy, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Nikita B Jolapara, Prabhvir Marway, Michael Fawzy. (2021). Acute Medical (bedside) Peritoneal Dialysis catheter insertion in Intensive Therapy Unit setting, an overlooked Renal Replacement Therapy option, A Case Study. Adv J Uro Nephro, 3(1): 9-13.
Abstract
A 56-year-old woman with a background of long-standing type II diabetes and chronic kidney disease presented to hospital with fevers, cough and shortness of breath. She had been self-isolating due to COVID-19 a week prior to admission. Her admission resulted in a long stay in intensive care unit with acute kidney injury. Due to her hypercoagulability state, multiple attempts of haemodialysis and filtration failed resulting in the first attempt of bedside (non-fluoroscopic guided) insertion of peritoneal dialysis catheter in an ITU setting in the region of Essex. We note that usually in an ITU setting, haemodialysis is commonly used. Our case therefore serves as an important reminder that peritoneal dialysis (PD) can serve a significant value for patients with acute kidney injury when haemodialysis cannot help in.
Keywords
Medical/Bedside Peritoneal Dialysis Catheter Insertion, Acute Kidney Injury (AKI), Intensive Therapy/Care Unit (ITU), COVID-19, Peritoneal Dialysis, Haemofiltration
Background
Intensive Therapy units (ITUs) are known to support hemofiltra-tion in an acutely unwell patient. This was extremely useful espe-cially in patients who presented with COVID-19 and acute kidney injury due to severe COVID sepsis. It is important to recognise that there is potential to extend this service and provide Acute peri¬toneal dialysis when haemofilteration can’t be safely maintained due to a variety of factors (access problems, hemodymamic insta¬bility, logistics due to unavailable haemofilters etc).
Peritoneal dialysis (PD) is usually a procedure performed by sur-geons and undertaken in a surgical setting. Post insertion there is a minimum of 2-4 weeks wound healing time before starting PD fluid exchange. Now days, the procedure can be performed in a procedure room by a nephrologist via fluoroscopy. This can be very useful due to:
a. It does not require the involvement of a surgical team so it can bypass surgical list backlog.
b. It can be done for those with high risk to general anaesthesia (eg; poor cardiac function, advanced COPD etc).
c. Another added bonus of bedside insertion is that peritoneal dialysis can be started earlier without a waiting period com-pared to surgical PD catheter due to less trauma elicited.
Our case today was done bedside (blind Seldinger technique with¬out fluoroscopic guidance)
Case Presentation
Our case is a 56-year-old female, a care home nurse, who present¬ed with a week history of fevers, cough and shortness of breath. Her symptoms persisted and as per National Health Service (NHS) guidance she began self-isolation. She presented to the hospital after her symptoms progressed and found it difficult to breath with admission oxygen saturations of 64% on room air. A nasopharyn-geal swab for SARS-CoV-2 PCR was positive on admission. Her background medical history included hypertension, Type 2 diabe¬tes mellitus, chronic kidney disease since 2017 with a baseline of creatinine 142umol/L.
On presentation to the emergency department, the patient was not¬ed to be in clear distress. Her respiratory rate was 28 breaths per minute, oxygen saturations 96% on 15L of oxygen provided, blood pressure of 164/73 mmHg, heart rate of 92 beats per minute and temperature of 38.5C. On clinical examination her chest was clear on auscultation bilaterally.
Her admission arterial blood gas on 15L O2 showed Type 1 respi¬ratory failure with pH 7.38, PO2 8.8, PCO2 4.6, HCO 21.4, BE -4.1, Lac 0.5 Glu 9.0. Her biochemistry showed lymphopenia of 0.68 10*9/L, sodium of 133mmol/L, potassium 4.3mmol/L, urea 31.8mmol/L, Creatinine 652umol/L, CRP 417 mg/L.
Her chest x-ray was consistent with COVID-19 pneumonia
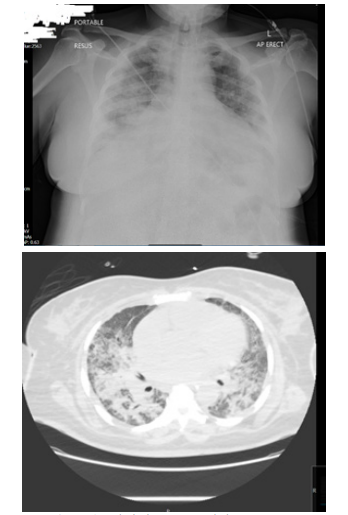
Figure 1: Admission CXR and chest CT scan.
Due to her hypoxic ABG and ongoing respiratory distress, she was intubated in resuscitation suite and admitted into ITU for ventila-tion support.
During her stay in ITU, she started to become anuric with wors-ening acute kidney injury (AKI) so she was established on con-tinuous veno-venous haemofiltration (CVVH). Unfortunately, she developed bilateral stroke on top of her COVID 19 pneumonia and Multi Drug Resistant pseudomonas infection. The story was so far typical for COVID but sadly to complicate the prognosis, she underwent multiple attempts of Central Venous Catheters (These accesses were needed to maintain CVVH support) as they were clotting within few hours of insertions despite being on continuous infusion of maximum dose of heparin due to her hypercoagulable state. During Dr Fawzy’s renal round in ITU, He realized that pa¬tient was not achieving enough clearance and ultrafiltration due to access problem. This has reflected on inability to wean the patien off high oxygen requirement of ventilator (she was on FiO2 of 100 %). It was obviously a failing CVVH and ITU team was running out of options.
Therefore, Dr Fawzy decided to step in and offered to put medical PD catheter and start PD for her. He examined her abdomen and there was only a scar of previous caesarean section with no obvi¬ous hernias.
After calling patient’s next of kin and obtaining the consent based on medical evaluation of patient’s best interest. Dr Fawzy instruct¬ed ITU team to hold heparin infusion for 2 hours.
Procedure
• After scrubbing for 3 minutes, sterile gown, mask and head cover is worn by the operator.
• The abdomen is prepped with chlorohexidine 2% and covered with sterile drapes.
• About 2 cm below the umbilicus a small midline horizontal incision (2-3 cm) is made under local anaesthetic.
• Blunt dissection of subcutaneous tissue is carried out reaching the anterior rectus sheath. Further local anaesthetic is given if necessary.
• 18g needle is then vertically inserted through the rectus sheath until it enters the abdominal cavity. The needle is then pointed toward the left lower quadrant (LLQ) but not advancing more than another 1-2 cm. About 200-300 ml of normal saline is injected through the needle to make sure the needle is in the cavity. Patients should not feel the fluid being injected.
• Guide wire then is passed through the needle aiming at LLQ. Before advancing the guide wire, the patient is informed that he/she may feel a sensation because of the guide wire touching the back of the bladder and some find it slightly unpleasant. The sensation will resolve when the wire is removed. Once the wire is in place the needle is removed.
• Over the guide wire the trocar is passed through the rectus sheath until it enters the abdominal cavity. This is to dilate the insertion point. The trocar is removed and reinserted this time being inside a peel away sheath. Once the instrument is in the abdominal cavity pointing to the LLQ, the peel away sheath is fed forward with trocar kept fixed. When peel away sheath is well in place, trocar and the guide wire are removed leaving the sheath behind.
• The 63 cm PD catheter is then passed through the peel away sheath and as the catheter is advanced, the sheath is pulled out and peeled away until all is out and the catheter is in the abdomen up to the proximal cuff. The cuff is pushed down to sit on the rectus sheath. At this time the catheter is tested to make sure that a normal saline flow in with no resistance and also the efflux is clear. • From the lateral side of the abdominal incision, more local anaesthetic is given and the catheter is advanced subcutane-ously using the tunnelling tool and aiming for the previously marked exit site in the lower part of the abdomen. It is made sure that the distal cuff of the catheter is about 3-5 cm prox¬imal to the exit site and the tunnel is not immediately under the skin.
• The incision is sutured and dressed. The patient will be re-turned to his bed with a Mepore dressing over the insertion site and the PD catheter exit site, and the PD catheter will be looped and secured with Mefix tape. A titanium and PD Extension set will be fitted and the catheter anchored securely. The tube is anchored to prevent trauma and to keep it immo-bilized at all times.
The patient started PD within 2 hours of the insertion. She was kept on assisted automated peritoneal dialysis (AAPD) for three weeks with average daily ultrafiltration of 2.5-3 litres. This en-abled ITU team to wean her off the ventilator. Later on, patient was stepped down to the renal ward after 43 days in ITU. She carried on PD for another 2 months then her urine output started to improve. Her renal function started to show signs of improvement. After 4 months of daily AAPD, PD was stopped and patient was kept on close follow up with regular blood tests. At that stage, pa-tient was still not verbalizing or able to engage in any conversation due to the double stroke she sustained in ITU.
Later on, Dr Fawzy attempted removal of PD catheter bed side under local anaesthetics
• I palpated the SC cuff and infiltrated 5ml of Lidocaine 1% around it.
• Skin incision of 2 cm over the cuff then dissecting the tissues until I managed to free the first cuff.
• Then, I infiltrated another 5ml of Lidocaine below the umbi-licus.
• 2 cm Skin incision sub umbilical was done and dissected the SC tissues down to peritoneum to free the inner cuff.
• Finally, both cuffs were freed and I managed to take the whole tube out successfully.
Outcome and Follow-Up
During the ward admission, her renal function gradually improved, and she was successful weaned off peritoneal dialysis with a new renal baseline creatinine of 212umol/L. Her PD tube was removed at the bedside as detailed above.
The patient underwent a percutaneous endoscopic gastrostomy (PEG) insertion for feeding and nutritional support as a result of her stroke. She further received neuro-rehabilitation therapy in a specialized centre. Miraculously, Patient showed progressive im-provement and then successfully discharge home. The patient was fully independent and AMT (mini mental capacity test) was 10/10. She had a new renal baseline function and no longer requiring a long-term renal replacement therapy (RRT).
Discussion
We note that usually in an ITU setting, haemfilter is commonly used. Our case therefore serves as an important reminder that peri-toneal dialysis can serve a significant value for patients. Patients who presented with severe COVID-19 sepsis and acute kidney injury, with or without a background of chronic kidney disease (CKD), were beneficiaries of haemofiltration in the ITU setting. It is important to recognise that there is potential to extend this ser-vice and provide peritoneal dialysis when haemodialysis can’t be safely maintained due to a variety of factors (e.g.; hypercoagulable states or if capacity to provide haemodialysis is limited).
In this case report we present the first case of a medical perito- neal dialysis insertion, that successfully attempted in ITU setting in Essex for a patient with severe COVID sepsis and AKI, with a beneficial short- and long-term impact for the patient. It obviously managed to keep dialysis ongoing as short term and protected the residual renal function as long term benefit.
Acute kidney injury is very common in critically ill patients and ICU patients. Usually, it is quite common to have renal replace¬ment therapy in such setting. 5-6% of AKI on ICU required RRT in a prospective multinational study carried out by Uchino et al. This was consistently seen across 23 participating countries. Peritone¬al dialysis is often overlooked in an ICU setting with continuous RRT or intermittent haemodialysis favoured more in developed countries. PD dialysis has its advantages due to its ease of admin¬istration, technicality, low bleeding risk and suitability in cardio¬vascular instability.
There has been high incident rate of AKI during COVID pandem-ic and generally COVID patients tend to be in a hypercoagulable state. This is worsened by the fact that patient was in ITU and had background morbidities that tend to substantially augment the hy¬percoagulable state. With these two factors this can put the patient in a much-compromised state as seen in this case report.
There are a few reports which follow established renal replacement therapy patients with COVID-19 infection into the ITU setting. There are also some reports which demonstrate the prevalence of AKI requiring renal replacement therapy and ITU admission, sec¬ondary to COVID-19 infection. A large retrospective cohort study of 4264 critically ill COVID-19 patients, of which 143 were di-alysis patients and 521 were CKD patients, indicated poorer out¬comes in those with pre-existing kidney disease. Dialysis patient shad a shorter time from symptom onset to ICU admission (medi¬an days: 4), compared to other groups (median days: 7 for CKD, 7 for those without pre-existing renal disease). 50% of dialysis and CKD patient died within 28 days of ITU admission, compared to 35% in patient s without pre-existing kidney disease (Hazard Ra¬tio (HR) 1.41 for dialysis patients, HR 1.25 for CKD patients). A small retrospective case series indicated fewer poor outcomes. From 14 patients on HD, or previously advanced CKD and now on HD secondary to COVID-19, four (29%) required mechanical ventilation and continuous RRT. One underwent ECMO for a pe¬riod of three days. From these 14 patients, two died from ARDS (14%), and nine (64%) were discharged from hospital. Another retrospective case series of 57 HD and 2 PD patients showed that eight (14%) required mechanical ventilation, and of these, three (38%) needed continuous RRT.
The remaining eight papers focused on AKI secondary to COVID-19 infection. A significant minority of patients with COVID-19 develop an AKI, ranging from 3-15%. This AKI is characterised by tubulointerstitial injury without glycosuria (8). For patients that required ITU admission, this incidence was much higher, ranging from 50-78%. This subset of patients with AKI (in¬cluding AKI on CKD) and COVID -19 infections appear to have an alarmingly high mortality rate, compared to those patients that do not develop an AKI. This may in part be explained by a strain on resources to provide effective RRT via haemodialysis. Stud¬ies indicate the effectiveness of urgent peritoneal dialysis catheter placement as an alternative, and reflect the patient case described in this report.
Lenfing Liu et al has scrutinized six studies (484 participants). Five studies compared high volume PD with daily haemodialysis, extended daily haemodialysis, or continuous renal replacement therapy. One study focused on the intensity of PD. The overall risk of bias was low to unclear. Compared to extracorporeal therapy, PD probably made little or no difference to all-cause mortality (4 studies, 383 participants: Relative risk (RR) 1.12, 95% Confidence index (CI) 0.81 to 1.55; I2= 69%; moderate certainty evidence), or kidney function recovery (3 studies, 333 participants: RR 0.95, 95% CI 0.68 to 1.35; I2= 0%; moderate certainty evidence). PD probably slightly reduces the amount of fluid removal compared to extracorporeal therapy (3 studies, 313 participants: MD -0.59 L/d, 95% CI -1.19 to 0.01; I2= 89%; low certainty evidence), and probably made little or no difference to infectious complications (2 studies, 263 participants: RR 1.03, 95% CI 0.60 to 1.78; I2= 0%; low certainty evidence). It is uncertain whether PD compared to extracorporeal therapy has any effects on weekly delivered Kt/V (2 studies, 263 participants: MD -2.47, 95% CI -5.17 to 0.22; I2= 99%; very low certainty evidence), correction of acidosis (2 stud-ies, 89 participants: RR 1.32, 95% CI 0.13 to 13.60; I2= 96%; very low certainty evidence), or duration of dialysis (2 studies, 170 par-ticipants: MD -1.01 hours, 95% CI -91.49 to 89.47; I2= 98%; very low certainty evidence). Heterogeneity was high and this may be due to the different extracorporeal therapies used. One study (61 participants) reported little or no difference to all-cause mortality, kidney function recovery, or infection between low and high and intensity PD. Weekly delivered Kt/V and fluid removal was lower with low compared to high intensity PD.
This case report highlights how PD can be used in patients where urgent RRT is required and HD may not be suitable or available. These papers provide strong evidence for the incidence of AKI secondary to COVID, and the additional logistical and service-lev-el challenges posed for new and existing users of RRT.
Furthermore, whilst an extensive discussion on the relative risks and benefits of peritoneal dialysis and haemodialysis is beyond the scope of this case report, it is noted that peritoneal dialysis confers lower relative risk of death compared to haemodialysis in the short term, especially within the first three months of dialy¬sis. Even when the peritoneal dialysis is unplanned, outcomes are non-inferior when compared to haemodialysis. This suits the cat-egory of patient described in this case report, where the dialysis is performed in an acute setting, without foresight of planning. Based on moderate (mortality, recovery of kidney function), low (infec-tious complications), or very low certainty evidence (correction of acidosis) there is probably little or no difference between PD and haemofilteration for treating AKI [1-18].
Learning Objectives
It is possible to have acute peritoneal dialysis safely in an ITU setting. Hypercoagulable patient can achieve more benefits from peritone¬al dialysis than haemodialysis Preserving residual renal function is a key factor on long term ben- eficial impact of peritoneal dialysis
The procedure can be safely done via the nephrologist as solo operator.
Conflict of interest: The authors declare that there is no conflict of interest.
References
- Abraham G, Varughese S, Mathew M, Madhusudan Vijayan (2015) A review of acute and chronic peritoneal dialysis in developing countries. Clin Kidney J 8: 310-317.
- Michael G Argenziano, Samuel L Bruce, Cody L Slater, Jonathan R Tiao, Matthew R Baldwin, et al. (2020) Characterization and clinical course of 1000 Patients with COVID-19 in New York: retrospective case series. MedRxiv 32511507.
- Sébastien Rubin, Arthur Orieux, Renaud Prevel, Antoine Garric, Marie-Lise Bats,et al (2020) Characterization of acute kidney injury in critically ill patients with severe coronavirus disease 2019. Clin Kidney J 13: 354-361.
- Geehan Suleyman, Raef A Fadel, Kelly M Malette, Charles Hammond, Hafsa Abdulla, et al. (2020) Clinical Characteristics and Morbidity Associated with Coronavirus Disease 2019 in a Series of Patients in Metropolitan Detroit 3: PMID: 32543702
- Sreedhar Adapa, Narothama Reddy Aeddula, Venu Madhav Konala, Avantika Chenna, Srikanth Naramala, et al. (2020) COVID-19 and Renal Failure: Challenges in the Delivery of Renal Replacement Therapy. J Clin Med Res 12: 276-285.
- Avital Angel-Korman, Tal Brosh, Karina Glick, Adi Leiba (2020) COVID-19, THE KIDNEY AND HYPERTENSION.Harefuah 159: 231-234.
- Gaiao S, Finkelstein FO, de Cal M, Claudio Ronco, Dinna N. Cruz, et al. (2012) Acute kidney injury: are we biased against peritoneal dialysis?. aPerit Dial Int 32:351-355.
- Abdullah Khalaf Al-Hwiesh (2014) Percutaneous Peritoneal Dialysis Catheter Insertion by a Nephrologist: A New, Simple, and Safe Technique. Perit Dial Int 34: 204-211.
- Ákos PethÅ?, Réka P Szabó, Mihály Tapolyai, László Rosivalle (2019) Bedside placement of peritoneal dialysis catheters - a single-center experience from Hungary. Ren Fail 41: 434-438.
- Brett Cullis, Abdullah Al-Hwiesh, Kajiru Kilonzo, Mignon McCulloch, Abdou Niang, et al. (2021) ISPD guidelines for peritoneal dialysis in acute kidney injury: 2020 update (adults). Perit Dial Int 41: 15-31.
- Koch M, Kohnle M, Trapp R, Haastert B, Rump LC, et al. (2012) Comparable outcome of acute unplanned peritoneal dialysis and haemodialysis. Nephrol Dial Transplant 27: 375-380.
- Linfeng Liu, Ling Zhang, Guan J Liu, Ping Fu (2017) Peritoneal dialysis for acute kidney injury. Cochrane Database Syst Rev PMID: 29199769.
- Prof Claudio Ronco, Thiago Reis, Faeq Husain-Syed, (2020) Management of acute kidney injury in patients with COVID-19 Lancet Respir Med 8: 738-742.
- Mariana Vigiola Cruz, Omar Bellorin, Vesh Srivatana, Cheg-uevara Afaneh (2020) Safety and Efficacy of Bedside Peritoneal Dialysis Catheter Placement in the COVID-19 Era: Initial Experience at a New York City Hospital. World J Surg 44: 2464-2470.
- Sinnakirouchenan R, Holley JL (2011) Peritoneal dialysis versus hemodialysis: risks, benefits, and access issues. Adv Chronic Kidney Dis 18: 428-32
- Hamza Bajwa, Yumna Riaz, Muhammad Ammar, Soban Fa-rooq, Amman Yousaf (2020) The Dilemma of Renal Involvement in COVID-19: A Systematic Review. Cureus 12: e8632.
- Maryanne Y Sourial, Mina H Sourial, Rochelle Dalsan, JayGraham, Michael Ross (2020) Urgent Peritoneal Dialysis in Patients With COVID-19 and Acute Kidney Injury: A Single-Center Experience in a Time of Crisis in the United States. Am J Kidney Dis 76: 401-406.
- Yong K, Dogra G, Boudville N, Mary Pinder, Wai Lim (2011) Acute kidney injury: controversies revisited. Int J Nephrol 2011: 1-17.



