Research Article - (2024) Volume 8, Issue 1
Acute Acquired Comitant Esotropia with Myopia: Longer-Term Follow-Up of Botulinum Toxin vs. Strabismus Surgery in a Western Population
2Suite 22, The Galway Clinic, Doughiska, Galway, H91 HHT0, Ireland
Received Date: Nov 01, 2023 / Accepted Date: Nov 25, 2023 / Published Date: Jan 22, 2024
Copyright: ©Ã?©2024 Clare McCloskey, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: McCloskey, C., Stokes, J., MacCann, A., McParland, D., Shah, S. F. A., et al. (2024). Acute Acquired Comitant Esotropia with Myopia: Longer-Term Follow-Up of Botulinum Toxin vs. Strabismus Surgery in a Western Population. J Ophthalmol Clin Res, 8(1), 01-09.
Abstract
‘Acute’ Acquired Comitant Esotropia with myopia (AACEM) is an unusual form of non-paretic, diplopia-inducing stra- bismus. It is a subset of (acute) acquired comitant esotropia with recent reports of increasing prevalence. No reports ex- ist comparing botulinum toxin (BTX) to surgery beyond the six-month therapeutic effect of BTX in a western population. Clinical characteristics of AACEM are defined and immediate and longer-term results of both interventions are reported in this retrospective review of 16 cases from two centres.
Eight cases received BTX and eight cases had primary surgical intervention. Mean distance and near angle pre-sur- gery was 37.50±10.35 prism diopters (PD) and 35.63±12.66 PD respectively. Mean distance and near angle at final follow-up post-surgery (475.88±301.21 days, median 466.5) was 5.62±3.78 PD and 5.88±5.25 PD respectively. Mean distance and near angle pre-BTX was 36.00±15.33 PD and 38.75±14.58 PD respectively. Mean distance and near angle at final follow-up post-BTX (789±548.98 days, median 700) was 20.13±15.57 PD and 22.25±17.85 PD respectively. In this the longer-term follow-up study surgery was found to be superior as compared to BTX post-intervention. Surgical alignment also appeared more stable.
Keywords
Botulinum toxin, Convergent strabismus, Esotropia, Myopia, Strabismus surgery
Introduction
Esotropia (ET) is most associated with hyperopia and abnormalities of accommodation [1,2]. Acute acquired comitant esotropia (AACE) is classified as a rare subtype of esotropia accounting for 0.3% of all strabismus [2]. AACE with myopia (AACEM) can present acutely with a variable history of preceding diplopia. There is a sense from recent reports that its prevalence is increasing [3]. AACE with myopia subtype was first proposed by Burian and Miller [4]. In this classification Type 1 (Swan type) represents cases that occur after interruption of fusion, Type 2 (Franceschetti type) typically occurs in low hyperopes with a ‘low accommodative’ element and occurring after ‘physical or psychic shock’ and the third Type 3 (Bielschowsky type) though originally described by von Graefe represents the least common form of acute onset comitant esotropia and was adapted by Bielschowsky who described features of myopia (typically under 5 dioptres), mild but equal limitation of abduction (under-abduction) as well as diplopia for distance but ‘fusion for near’, preceded by ‘physical or psychic shock’ [5]. The description differed between von Graefe who reported a combination of slight but symmetric under-abduction as well as slight but symmetric over-adduction whereas Bielschowsky observed only mild symmetric under-abduction [4].
Consider that the Bielschowsky/Type 3 sub classification of acute acquired comitant esotropia with myopia has undergone modification after its first description under von Graefe, subsequently under Bielschowsky and then more recently under Chen, et al and, a further iteration most recently under Lang, et al, to now also include esotropia for near fixation as well, therefore AACEM is a condition with salient features of myopia, comitant esotropia for distance and near (differing by no greater than 10 prism dioptres i.e. no ‘accommodative’ component), and without features of neuropathology or Heavy eye syndrome (i.e. structurally altered extraocular muscle paths) [5-11]. We use these criteria to describe the clinical features and management outcomes of patients treated by 2 consultant strabismus surgeons in two treatment centres and compare the effect of strabismus surgery (surgery) and botulinum A toxin (Allergan) treatment (BTX). This is the first report to our knowledge in a western population and reports the longest follow-up of all heretofore publications.
Patients and Methods
We reviewed the medical records of patients with a diagnosis of new onset diplopia and comitant esotropia at two ophthalmology units in Ireland. Exclusion criteria were subjects with amblyopia (≥ 0.2 logMAR interocular best corrected visual acuity (BCVA) or history of prior amblyopia treatment), previous strabismus surgery, accommodative element and divergence paralysis (greater than 10PD between distant and near measurements), incomitancy (greater than 5PD between right and left gaze measurements) and any other condition known to affect ocular motility such as orbital fracture, myasthenia gravis, thyroid eye disease and cranial nerve palsy. Eight patients fulfilled the study criteria between August 2017 and March 2019 at site 1. Nine patients between 2009 and 2018 recorded at site 2 were included. At each site, patients were managed by one surgeon. At site 1 patients were offered only surgery and at site 2 patients were offered surgery or BTX, the risks and merits of each were discussed and the intervention was chosen eventually by the patient and or guardian. Importantly no published reports of BTX in the treatment of AACEM prior to 2019 existed [8].
Data recorded for each patient included: age, gender, presenting symptoms and duration, refraction (manifest and cycloplegic), logMAR BCVA, strabismus angle for distant (6 metre) and near (1/3 metre) fixation (using alternating prism cover test with full correction) and treatment intervention. When performed prism fusion range and stereopsis were also reported.
The presence of Heavy eye syndrome was outruled clinically and from available imaging (CT or MRI) and/or axial length data [11-15]. The parameters distinguishing Heavy eye syndrome included the axial length being less than 32mm according to Aoki, et al and the angular difference between lateral rectus and superior rectus (closest image to globe equator) on imaging being less than 180 degrees (in both eyes) as described in Fresnia, et al. excluded this diagnosis confidently in all cases by either one or both criteria [11,13]. Axial lengths were recorded using optical biometry measurements (IOL Master model 700, Carl Zeiss Meditec AG, Jena, Germany and Lenstar, LS 900, Haag-Streit). The maximum axial length was 27.71mm in the right and 27.91mm in the left eye (mean ± standard deviation right eye = 25.31±1.10mm, left = 25.30±1.29mm) from ten patients. The maximum angular separation between lateral and superior rectus was 102 degrees in the right and 103 degrees in the left eye (mean ± standard deviation right eye = 97.4±4.57, left eye = 95.3±4.43) from ten patient imaging studies.
Consent was obtained from each patient and written ethical approval was received by the Research Ethics Committee (REC) at each site and the study was conducted in agreement with the tenets of the Declaration of Helsinki. Data are presented as mean, SD and median where appropriate. Statistical analysis was performed using two-sided student’s t-test and statistical significance was defined as a p-value ≤0.05.
Results
Table 1 shows patient demographics and baseline characteristics of our AACEM cohort in a western population. All patients reported diplopia or ghosting of images in the horizontal meridian, but eight patients did not report strabismus despite the strabismus angle exceeding 10 PD. Mean duration of symptoms before first attendance was 9.88±6.54 months (median 12, range 3-24). All patients in our cohort were myopic (one patient (patient 1) had myopic anisometropia with the left eye spherical equivalent of +0.38). In terms of the total prism fusion range (TPFR, Base In + Base Out) the mean for distance pre-intervention was 20.17 (±19.14) PD, and for near was 28.20 (±21.61) PD. All but two patients had radiological imaging of the brain to out rule neuropathology.
|
Patient |
Site |
Intervention |
Age (years) |
Gender |
Symptom duration (months) |
BCVA |
SE |
Pre-intervention angle (PD) |
|||
|
Rt eye |
Lt eye |
Rt eye |
Lt eye |
Distance |
Near |
||||||
|
1 |
1 |
Surgery |
33 |
Female |
3 |
0.08 |
-0.08 |
-1.25 |
0.38 |
25 |
20 |
|
2 |
1 |
Surgery |
43 |
Female |
5 |
0.2 |
0.1 |
-12.88 |
-8.88 |
35 |
30 |
|
3 |
1 |
Surgery |
19 |
Male |
12 |
0 |
0.02 |
-2.38 |
-2.63 |
40 |
40 |
|
4 |
1 |
Surgery |
39 |
Female |
3 |
-0.18 |
-0.18 |
-5.13 |
-6.38 |
30 |
35 |
|
5 |
1 |
Surgery |
21 |
Male |
12 |
0.02 |
0 |
-2.25 |
-1.38 |
30 |
20 |
|
6 |
1 |
Surgery |
16 |
Male |
12 |
0 |
0 |
-2.63 |
-2.75 |
35 |
35 |
|
7 |
1 |
Surgery |
18 |
Female |
24 |
0 |
0 |
-5.13 |
-4.50 |
50 |
50 |
|
8 |
1 |
Surgery |
27 |
Male |
12 |
-0.18 |
-0.18 |
-0.63 |
-0.25 |
55 |
55 |
|
Site 1: Mean ± SD |
27.00 ± 10.27 |
4 F, 4 M |
10.38 ± 6.91 |
0.00 ±0.13 |
-0.04 ±0.10 |
-4.04± 3.93 |
-3.30± 3.15 |
37.50 ±10.35 |
35.63 ±12.66 |
||
|
9 |
2 |
Surgery |
29 |
Female |
6 |
0.1 |
0.1 |
-4.13 |
-4.50 |
37 |
37 |
|
10 |
2 |
Botox |
18 |
Male |
4 |
0.2 |
0.2 |
-6.25 |
-7.25 |
33 |
40 |
|
11 |
2 |
Botox |
17 |
Female |
8 |
0.06 |
0.12 |
-6.13 |
-6.63 |
30 |
40 |
|
12 |
2 |
Botox |
15 |
Male |
12 |
0.2 |
0 |
-2.25 |
-2.75 |
25 |
30 |
|
13* |
2 |
Botox |
18 |
Male |
12 |
-0.1 |
-0.1 |
-4.25 |
-4.50 |
70 |
70 |
|
14* |
2 |
Botox |
10 |
Male |
3 |
0.04 |
0 |
-3.75 |
-3.88 |
35 |
35 |
|
15* |
2 |
Botox |
21 |
Male |
24 |
0.2 |
0.04 |
-3.25 |
-3.00 |
25 |
25 |
|
16* |
2 |
Botox |
16 |
Male |
4 |
0.06 |
0.14 |
-3.25 |
-2.75 |
25 |
25 |
|
17* |
2 |
Botox |
22 |
Female |
12 |
0.1 |
0.1 |
-6.38 |
-6.50 |
45 |
45 |
|
Site 2: Mean ± SD |
18.44 ±5.27 |
3 F, 6 M |
9.44 ±6.58 |
0.10 ±0.10 |
0.07 ±0.10 |
-4.23 ±2.81 |
-4.01 ±2.52 |
36.11 ±14.35 |
38.56 ±13.65 |
||
|
Total (Site 1 +2): Mean ± SD |
22.47 ±8.91 |
7 F, 10 M |
9.88 ±6.54 |
0.05 ±0.12 |
0.02 ±0.11 |
-4.23 ±2.81 |
-4.01 ±2.52 |
36.76 ±12.26 |
37.12 ±12.87 |
||
Table 1. Acute Acquired Comitant Esotropia (AACE) Patient Demographics
Demographics of the entire AACE cohort, all patients at site 1 and 2 and the interventions. Data are expressed as mean (± Standard Deviation). BCVA = Best Corrected Visual Acuity, Rt = Right, Lt = left, SE = Spherical Equivalent, PD = Prism Dioptre
To compare interventions of surgery versus BTX in patients with AACEM, patient 9 at site 2 was the only one to choose surgery as their initial intervention therefore we conducted our statistical analysis (see below) with and without patient 9 data and no difference in statistical conclusions resulted. Patient 9 has been excluded in these presented results to avoid bias for outlier effects (results including patient 9 are available in supplementary data (S1)).
Table 2 shows eight patients at site 1 who had surgery and eight patients at site 2 who had BTX. Pre-operative demographics between the two groups before intervention were similar, gender (p=0.33), symptom duration (p=0.89), BCVA (p=0.08), spherical equivalent (p=0.55), pre-operative distance angle (p=0.82) and pre-operative near angle (p=0.65) except for age, (p=0.02) whereby site 1 had slightly older patients (mean age 27) than site 2 (mean age 17). All patients at site 1 underwent bi-medial rectus recessions with an adjustable suture on one medial rectus based on forced duction test (patient 1-8). Surgery at site 2 when performed was done in the same way but no adjustable stitch was employed. The eight patients at site 2 had BTX to a single medial rectus at a dose of 1.25 units in 0.10ml for each 30 D of strabismus correction (Botulinum toxin A, Allergan) under electromyographic control using a monopolar needle under topical anesthesia.
Importantly, the data subjected for statistical analysis in this work represents only those relating to the first treatment with BTX. The length of follow-up for this study is long (years) and some patients were controlled for over 3 years before presenting again to choose repeat BTX but these data from subsequent injections are not included in either the tables nor are they used for the purpose of statistical analysis (supplementary data).
Five in the BTX group (patient 13-17), initially had BTX but significant strabismus recurred (median 11 months, range 8 months to 9 years) after initial BTX injection and opted for surgery subsequently. The strabismus angles used for data analysis in these patients are those measured at the time of their decision for surgery.
|
Patient |
Site |
Intervention |
Pre-intervention angle (PD) |
Early post-in-tervention angle (PD) |
Follow-up after intervention (days) |
Post-intervention angle (PD) |
Follow-up after intervention (days) |
Further intervention |
Final post-in-tervention angle (PD) |
Final follow-up (days) |
||||
|
Distance |
Near |
Distance |
Near |
Distance |
Near |
Distance |
Near |
|||||||
|
1 |
1 |
Surgery |
25 |
20 |
|
|
|
6 |
6 |
624 |
|
|
|
|
|
2 |
1 |
Surgery |
35 |
30 |
|
|
|
6 |
6 |
687 |
|
|
|
|
|
3 |
1 |
Surgery |
40 |
40 |
|
|
|
4 |
4 |
672 |
|
|
|
|
|
4 |
1 |
Surgery |
30 |
35 |
|
|
|
4 |
4 |
239 |
|
|
|
|
|
5 |
1 |
Surgery |
30 |
20 |
|
|
|
10 |
18 |
155 |
|
|
|
|
|
6 |
1 |
Surgery |
35 |
35 |
|
|
|
1 |
1 |
155 |
|
|
|
|
|
7 |
1 |
Surgery |
50 |
50 |
|
|
|
12 |
6 |
966 |
|
|
|
|
|
8 |
1 |
Surgery |
55 |
55 |
|
|
|
2 |
2 |
309 |
|
|
|
|
|
Surgery: Mean±SD |
37.50± 10.35 |
35.63± 12.66 |
|
|
|
5.62± 3.78 |
5.88± 5.25 |
475.88± 301.21 |
|
|
|
|
||
|
10 |
2 |
Botox |
33 |
40 |
0 |
0 |
12 |
35 |
40 |
1188 |
|
|
|
|
|
11 |
2 |
Botox |
30 |
40 |
-10 |
-10 |
13 |
4 |
4 |
798 |
|
|
|
|
|
12 |
2 |
Botox |
25 |
30 |
|
|
|
0 |
0 |
1026 |
|
|
|
|
|
13* |
2 |
Botox |
70 |
70 |
4 |
6 |
55 |
10 |
12 |
1837 |
BTX then Sx |
0 |
0 |
423 |
|
14* |
2 |
Botox |
35 |
35 |
10 |
14 |
12 |
35 |
40 |
602 |
BTX then Sx |
10 |
10 |
1341 |
|
15* |
2 |
Botox |
25 |
25 |
0 |
0 |
17 |
12 |
9 |
264 |
BTX then Sx |
0 |
0 |
143 |
|
16* |
2 |
Botox |
25 |
25 |
0 |
0 |
25 |
25 |
30 |
344 |
BTX then Sx |
0 |
0 |
228 |
|
17* |
2 |
Botox |
45 |
45 |
4 |
2 |
19 |
40 |
43 |
253 |
BTX then Sx |
0 |
0 |
164 |
|
Botox: Mean±SD |
36.00± 15.33 |
38.75± 14.58 |
1.14± 6.09 |
1.71± 7.25 |
21.86± 15.35 |
20.13± 15.57 |
22.25± 17.85 |
789± 548.98 |
Mean±SD |
2± 4.47 |
2± 4.47 |
459.8± 504.84 |
||
Table 2: Surgery versus botox in Acute Acquired Comitant Esotropia with Myopia (AACEM)
Surgery versus botox for AACEM cohort. Patients at site 1 (all who had surgery (Sx)) are compared with patients at site 2 (following their first botox (BTX) injection). No data from repeat BTX injections are included in this study. (*) patients are those who subsequently had surgery after BTX with final outcomes displayed on right hand side of the table. Blank cells indicate no data available for this time-point. Patient 10 received BTX and successfully re-aligned but decompensated 3.25 years later whereupon elected to have repeat BTX and remained aligned at final follow up. Patient 13 was managed with three BTX injections over eight years before eventually opting for surgery. Patient 14 received BTX with suboptimal realignment and 15 months later proceeded to surgery. Patient 15 received BTX and successfully re-aligned, decompensated 8.6 months after and proceeded to surgery. Patient 16 successfully re-aligned with a single BTX injection, decompensated at 5 months and opted for surgery 11.28 months after BTX treatment. Patient 17 successfully aligned after a single BTX injection decompensated 5.85 months after and opted for surgery 8.28 months after this single BTX treatment. As patient 9 was the only patient at site 2 to choose surgery as an initial intervention, they were eliminated from the presented data to remove outliers (however there was no difference in statistical significance with patient 9 included or excluded – see supplementary data with patient 9 included). Data are expressed as mean (± Standard Deviation). P-value <0.05. PD = Prism Dioptre
Strabismus Surgery Results (Site 1)
Amongst the eight patients in site 1 who had surgery, the mean pre-surgery distance angle was 37.50±10.35 PD and near 35.63±12.66 PD. The mean post-surgery distance angle at final follow-up (475.88±301.21 days, median 466.5) was 5.62±3.78 PD (p<1.0 x 10-6) and near 5.88±5.25 (p<2.0 x 10-5). All patients at end of follow-up remained aligned and symptom free.
Botulinum Toxin Results (Site 2)
Eight patients had BTX. The mean ± SD distance angle pre-BTX was 36.00±15.33 PD and mean near angle pre-BTX was 38.75±14.58 PD. The mean distance angle at final follow-up (789±548.98days, median 700) was 20.13±15.57 PD (p=0.06) and mean near angle at final follow-up was 22.25±17.85 PD (p=0.06). Two patients remained aligned and symptom-free at end of follow-up with BTX treatment alone. One patient required further BTX (39 months after initial BTX treatment) but has remained symptom free at end of their follow-up (46 months). Five patients who received BTX decompensated and opted for surgery (8.3, 8.7, 11, 19 and 60 months after initial BTX). The surgery undertaken in these patients was successful and at their final follow-up (of 459.8±504.84 days, median 228) the mean distance angle was 2±4.47 PD (p=0.008) and near angle of 2±4.47 PD (p=0.009).
Strabismus Surgery Versus Botox
When comparing the eight patients who underwent strabismus surgery in site 1 to the eight patients who had their first BTX treatment at site 2 (Figure 1), the mean pre-surgery distance (37.50±10.35 PD) and near angle (35.63±12.66 PD) was not statistically different to the mean pre-BTX distance (36±15.33 PD, p=0.82) and near angles (38.75±14.58 PD, p=0.65). All other parameters were similar pre-intervention (patients in site 2 were slightly younger, see above). The mean post-surgery distance (5.62±3.78 PD) and near angle (5.88±5.25 PD) was statistically different to the mean post-BTX distance (20.13±15.57 PD, p=0.02) and near (22.25±17.85 PD, p=0.03) indicating surgery was superior in its effect after a single intervention.
Figure 1: Title: AACEM – Effect of Surgery and Botox.
Acute Acquired Comitant Esotropia with Myopia (AACEM) strabismus angles before and at final post intervention follow up for the entire cohort treated with strabismus surgery (Red) and with botulinum toxin (Blue). The upper panel (A) shows the change in distance fixation angles and lower panel (B) shows the change in near fixation angles. There is a reduction in strabismus angle within each group, strabismus surgery is statistically significant (distance; p<0.00001 and near; p<0.0001) and there is a trend towards reduction in the botox group although not statistically significant (distance; p = 0.06 and near; p = 0.06). When comparing the effectiveness of surgery with botox post intervention, the surgery group shows a statistically significant reduction in angle compared with botox (distance; p = 0.02 and near; p = 0.03). Error bars represent 1 standard error of the mean.
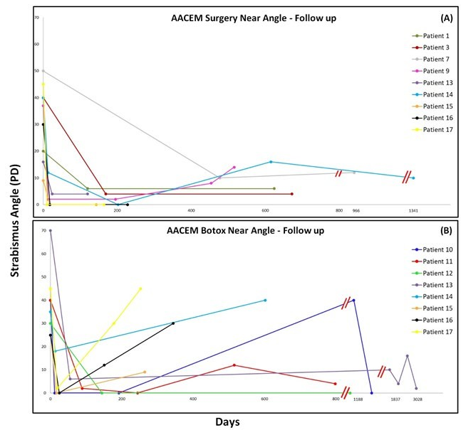
Longer Term Outcome Botulinum Toxin and Strabismus Surgery
Data in Figures 2 and 3 show essentially that drift away from alignment in the early post-operative period occurs for both groups, earlier for BTX than surgery patients and a seemingly greater magnitude drift in the BTX than the surgery group. This trend seems similar for both the distance (Figure 2) and near (Figure 3) strabismus angles.
Figure 2: Title: AACEM – Longer term Follow-up for Surgery and Botox (Distance Angle).
‘Acute’ Acquired Comitant Esotropia with Myopia (AACEM) strabismus angles for distance fixation before and after re-alignment intervention with Surgery in Panel A (upper) and BTX in panel B (lower) and the recorded change with extended follow up (days, x-axis). Strabismus angles are plotted on the y-axis in prism diopters (PD). Patient 1, 3 and 7 who had surgery are from site 1. Patient 9 to 17 are from site 2. Patient 9 had surgery only, patients 10 to 17 had BTX but patient 13 to 17 had surgery following failed BTX. We included patient 13 to 17’s surgical outcomes (six months after BTX treatment) in Panel A for display purposes. There is apparent drift away from initial alignment for both treatment groups but the recurrence of esotropia appears greater for the BTX treated group than the surgery treated group and occurs earlier. The double red lines indicate a scale break.
Stereoacuity
Those in whom binocular status was recorded pre-intervention (number of patients, n=12) and, post-intervention (n=16) demonstrated improved binocularity. Best stereoacuity thresholds were measured by a variety of tests (TNO-distance and near, Frisby-distance and near, Wirt Fly stereotest, Lang) whilst wearing a prismatic correction of their manifest strabismus. Pre-intervention only 4 cases achieved measurable stereothreshold with a mean of 134.25 arcsec (min 55, max 200). Final follow-up stereothreshold was measurable in 14 cases with a mean of 80.8 arcsec (min 40, max 300). This means that the realignment resulted in greater proportion of patients achieving a stereothreshold and in many cases improved stereoperformance, even amongst individuals in whom pre-operative testing predicted no improvement.
Discussion
AACE differs from neuropathological, accommodative and myopic (Heavy Eye) strabismus and has itself been classified into three distinct subtypes. The data reported here are specific to AACE with myopia (AACEM) and we have taken care to exclude confounding or competing aetiologies that may bias our results. The relevance of myopia in AACE strabismus is likely underscored not simply by the increasing prevalence of myopia worldwide but also by the increased frequency of ACCE reports since 2019, most concentrated in non-western populations where myopia is more prevalent. Our experience differs significantly from previous reports with shorter follow up in that we report surgery to be more predictably successful than BTX in this cohort of AACEM [16].
There have been a number of recent publications on the subject of AACEM but only a handful that compare BTX to surgery. Huang, et al recently described 78 AACE patients over a two-year period [7-9,12,17,18]. Forty-six (46) patients had strabismus surgery and 32 patients had BTX. Sixty-five (83.33%) of their cohort had myopia but the majority of these patients also had divergence insufficiency (n=52) and in this way their findings may not be comparable to ours. Shi, et al performed a prospective study over three years treating 60 patients (40 BTX, 20 surgery), mean follow up 17.4 months [18]. They had similar relapse rates in both treatment groups with 22.5% of BTX group at 15.6 months and 20% of surgery group at 6 months requiring additional interventions. Initial cumulative success rates after first intervention was 85% for surgery and 95% for BTX. At final follow-up they reported success rates of 72.5% for BTX and 65% for surgery (p = 0.55) following additional treatments. Tong, et al report treatment of mostly myopic (n=22, 91%) with BTX alone with 3 months follow up [12]. The BTX dose administered is uncertain. They state both medial recti were injected using 3.5 units (4.0 units if pre-injection angle greater than 25 dioptres) but the total dose is not specified. This dose is higher than that used in this report, and no overcorrections were reported. It is possible this dose could result in better longer-term control than that reported here. The authors accept the limitation of their short follow-up and that the longer-term recurrence of this strabismus is not currently known. Lang, et al (25 of 29 myopic) compare BTX to surgery but analyse their success categorically (orthotropia, overcorrection, undercorrection) so actual outcome data for each patient is unavailable [8]. They conclude at six months post treatment, the success rate for BTX was 84.6% and for surgery was 81.3% with no statistical difference between the interventions (p=1.00).
Reasonable criticisms following our lesser success rate with BTX could be that of injector skill or insufficient BTX dose. Injector skill seems unlikely to explain the fact that all our cases of BTX injection resulted in a significant reduction in strabismus angle while it was rather the early loss of alignment after BTX that lead to the ultimate failure of this treatment modality in some but not others. The dose of BTX used here was similar to Huang, et al [17]. A recent inquiry of practising consultant paediatric and strabismus surgeons in the United Kingdom and Ireland including high volume BTX injectors finds broad consensus with the dosage of botox in this report (personal communication SC on BIPOSA forum, 2023).
Data reported here represents to our knowledge, the longest post-intervention follow-up (26 months BTX, 16 months surgery) for a comparison study in AACEM [8,16,17]. This is important for two reasons. Firstly, BTX has a pharmacologic action of 6 months [13]. Secondly, in our experience BTX is not uniformly successful compared with surgery in this defined subgroup of AACEM.
The nomenclature around comitant esotropia without neuropathology often makes reference to its presentation as acute but in the vast majority of reports (including this one) strabismus and diplopia / visual disturbance is present for some time before the patient presents (Table 1) [3,4,7-10,12,17-18]. Considering all relevant reports of acute acquired comitant esotropia whether myopic or hyperopic and where symptom duration was provided or calculable the mean duration of symptoms (diplopia and/or strabismus) ranges from a minimum of 2.40 years to a maximum of 5.42 years [7,9]. The term ‘acute’ may therefore not be appropriate. More importantly it means that accurate timing of the causative event(s) is often impossible in many cases and the potential for clinical and vergence-related features to be altered after the inciting event (if there is one) is a real possibility.
A criticism of our data is that it is retrospective and therefore open to bias. In that AACEM is uncommon, randomized data would be difficult to collect.
Taken together AACEM could be considered not as a specific entity but as an unusual form of esotropic srabismus associated with myopic refractive error. The trigger for decompensation may be excessive convergence increasing medial rectus tonus [19]. Convergence interneurons outnumber divergence interneurons by a factor of 3:2 in primate midbrain [14]. In hyeropes acute comitant esotropia may be triggered by undercorrection with increased cilliary muscle tonus to which convergence is also linked [14]. Additionally there may be asynchrony or anisoaccommodation that triggers or contributes [15]. Following from this it makes sense to examine the accommodation and vergence systems in these and all forms of strabismus.
Conclusion
Our cohort of patients who fulfilled the inclusion and exclusion criteria for AACE, all were myopic (AACEM). Strabismus surgery and BTX for AACEM are both successful but surgery appears more predictable and stable.
Conflict of Interest: None.
Acknowledgements
Dr. Derek Lohan, Consultant Radiologist, The Galway Clinic for calculation of angulation in extraocular muscles. Dr. Eszter Feher, for translation of manuscripts in German. Dr. Peter Roggenkamper, Bonn, Germany, for providing a copy of the von Graefe manuscript.
Funding
none
Author Contributions
C McC – data collection, analysis and manuscript preparation.
A MC – data collection, manuscript preparation.
D McP – data collection, manuscript preparation.
S F – data collection, manuscript preparation.
J S – data collection, manuscript preparation.
P ON – research and manuscript preparation.
SIC – data collection, analysis and manuscript preparation.
Ethics Approval
Consent was obtained from each patient and written ethical approval was received by the Research Ethics Committee (REC) at each site and the study was conducted in agreement with the tenets of the Declaration of Helsinki.
Data Availability
The datasets generated during and/or analysed during the current study are not publicly available due to identifiable patient information but are available from the corresponding author on reasonable request.
References
- Repka, M. X., Lum, F., & Burugapalli, B. (2018). Strabismus, Strabismus Surgery, and Reoperation Rate in the United States: Analysis from the IRIS Registry. Ophthalmology, 125(10), 1646-1653.
- Mohney, B. G. (2007). Common forms of childhood strabismus in an incidence cohort. American journal of ophthalmology, 144(3), 465-467.
- Okita, Y., Kimura, A., Masuda, A., Mochizuki, Y., & Kondo, M., et al. (2023). Yearly changes in cases of acute acquired comitant esotropia during a 12-year period. Graefes Arch Clin Exp Ophthalmol, 261(9), 2661-2668.
- Burian, H. M., & Miller, J. E. (1958). Comitant convergent strabismus with acute onset. Am J Ophthalmol, 45(4 Pt 2), 55-64.
- von Graefe, A. (1864). Ueber die von Myopie abhangige Form convergiernden Schielens und deren Heilung. Archiv für Ophthalmologie, 10(1), 156-175.
- Bielschowsky, A. (1922). Das Einwartsschielen der Myopen. Ber Deutsche Ophth Gesell, 43, 245-248.
- Chen, J., Deng, D., Sun, Y., Shen, T., & Cao, G., et al. (2015). Acute Acquired Concomitant Esotropia: Clinical features, Classification, and Etiology. Medicine, 94(51), e2273.
- Lang, L.J., Zhu, Y., Li, Z.G., Guang-Ying, Z., & Hai-Ying, P., et al. (2019). Comparison of botulinum toxin with surgery for the treatment of acute acquired comitant esotropia and its clinical characteristics. Scientific Reports, 9(1), 13869.
- Ohtsuki, H., Hasebe, S., Kobashi, R., Okano, M., & Furuse, T., (1994). Critical period for restoration of normal stereoacuity in acute-onset comitant esotropia. Am J Ophthalmol, 118(4), 502-508.
- Spierer, A. (2003). Acute concomitant esotropia of adulthood. Ophthalmology, 110(5), 1053-1056.
- Aoki, Y., Nishida, Y., Hayashi,. O, Nakamura, J., & Oda, S., et al. (2003). Magnetic resonance imaging measurements of extraocular muscle path shift and posterior eyeball prolapse from the muscle cone in acquired esotropia with high myopia. Am J Ophthalmol, 136(3), 482-489.
- Tong, L., Yu, X., Tang, X., Zhang, Y., & Zheng, S., et al. (2020). Functional acute acquired comitant esotropia: clinical characteristics and efficacy of single Botulinum toxin type A injection. BMC Ophthalmol, 20(1), 464.
- Fresina, M., Finzi, A., Versura, P., & Campos, E. C. (2014). Muscle belly union associated with simultaneous medial rectus recession for treatment of myopic myopathy: results in 33 eyes. Eye (Lond), 28(5), 557-561.
- Tychsen, L. (2005). Strabismus: The Scientific Basis. In: Taylor D, Hoyt CS, eds. Pediatric Ophthalmology and Strabismus. 3rd ed. Elsevier, 76, 836-848.
- Toor, S., Horwood, A. M., & Riddell, P. (2018). Asymmetrical accommodation in hyperopic anisometropic amblyopia. Br J Ophthalmol, 102(6), 772-778.
- Wolffsohn, J. S., Jong, M., Smith, E., Resnikoff, S. R., & Jonas, J. B. et al. (2021). ‘IMI 2021 Reports and Digest – Reflections on the implications for clinical practice’. Investigative Ophthalmology & Visual Science, 62(5), 1.
- Huang, X., Meng, Y., Hu, X., Zhao, Y., & Ye, M., et al. (2022). The Effect of Different Treatment Methods on Acute Acquired Concomitant Esotropia. Comput Math Methods Med, 2022, 5001594.
- Shi, M., Zhou, Y., Qin, A., Cheng, J., & Ren, H. (2021).Treatment of acute acquired concomitant esotropia. BMC Ophthalmol, 21(1), 9.
- Vagge, A., Giannaccare, G., Scarinci, F., Cacciamani, A., & Pellegrini, M., et al. (2020). Acute Acquired Concomitant Esotropia From Excessive Application of Near Vision During the COVID-19 Lockdown. J Pediatr Ophthalmol Strabismus, 57, e88-e91.



