Research Article - (2022) Volume 3, Issue 1
A Wake-Up Call: Determination of Antibiotics Residue Level in Raw Meat in Abattoir and Selected Slaughterhouses in Five Local Government in Kano State, Nigeria
2Department of Community Medicine, Environmental Health Unit, Faculty of Clinical Sciences, Niger Delta University, Wilberforce Island, Bayelsa State, Nigeria
Received Date: Jan 01, 2022 / Accepted Date: Jan 07, 2022 / Published Date: Jan 21, 2022
Copyright: ©Copyright: ©2022 Habeeb Modupe Lateefat, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Habeeb Modupe Lateefat, Opasola Afolabi Olaniyi, Garba Misbahu, Olalekan Morufu Raimi. (2022). A Wake-Up Call: Determination of Antibiotics Residue Level in Raw Meat in Abattoir and Selected Slaughterhouses in Five Local Government in Kano State, Nigeria. J Vet Heal Sci, 3(1), 54-61
Abstract
The frequent use of antibiotics may result in drug residues that can be discovered at varying quantities in animal products such as milk or meat. Various persons have perceived the presence of pharmaceutical residues in food above the MRLs globally. Antibiotics residues are present in food, which can endanger human health by causing antibiotic sensitivity, allergic reactions, microflora imbalance, bacterial resistance to antibiotics in microorganisms, and financial loss to the food industry. Farmers around the world utilize them on a sporadic basis for both preventative and curative purposes. This study assessed the antibiotics residues in raw meat sold in 6 slaughter houses in Kano States. The study is a descriptive cross-sectional study involving six (6) major slaughterhouse in Kano state. Muscle, Kidney and liver samples were collected from each slaughterhouse. The antibiotic residues in the meat samples were analysed using high performance liquid chromatography (HPLC) for tetracycline, ciprofloxacin and oxytetracycline residue results were presented in charts and tables. Out of a total of 18 beef samples analyzed during this study, 15 (83%) of the total samples had detectable levels of tetracycline residues from which 6(33.3%) had tetracycline residues at violative levels above the WHO/FAO maximum residue limits (MRLs), out of those 18 beef samples analyzed during this study, 6(33%) of the total samples had detectable levels of oxytetracycline residues from which 3(17%) had oxytetracycline residues at violative levels above the WHO/FAO maximum residue limits (MRLs) and out of those 18 beef samples analyzed during this study, 12(67%) of the total samples had detectable levels of ciprofloxacin, all levels are below the WHO/FAO maximum residue limits (MRLs). This high level of tetracycline and oxytetracycline residues in greater proportion of meat destined for human consumption at violative levels could be as a result of the indiscriminate use and misuse of veterinary drugs as commonly practiced among livestock producers and marketers without observing withdrawal period prior to slaughter. These results indicate that consumers may be predisposed to health hazards and hinder international meat trade from Nigeria. Regulatory authorities should therefore ensure compliance with good agricultural practices including withdrawal period of drugs used for treatment of food animals.
Keywords
Antibiotics, Tetracyclin. Oxytetracycline, Ciprofloxacin, Slaughterhouse, Kano State
Introduction
Rachel Carson stated fifty (50) years ago for the first time that the widespread use of pesticides is hazardous not only to wild-life but also to humans. This is still an ongoing concern, as ev-idenced by recent studies by that pesticides are contributing to and causing massive adverse effects, necessitating the restric-tion of a wide range of chemical products and uses, and the use of many chemicals in commerce with far greater prudence and caution [1-8]. The authors claimed that humankind is heading beyond a "safe operating area" as the size of these consequenc¬es approaches or exceeds certain thresholds that indicate global system tipping points or natural limits for processes with no ap¬parent limits (so-called "hazardous levels"). Numerous research- ers have reported that human health is generally influenced by their environment and specifically, the nature and quality of the food [9-37].Nature cattle products is broadly concerning public health body throughout the globe since veterinary medications have assumed a significant part in the field of animal farming and agro-industry, and increasing event of drug residues, and re¬sistance have become issues of concern [38]. Veterinary medica¬tions are expected to address the difficulties in provision of suf¬ficient measures of food to the increasing global population as medications enhance pace of weight gain, further enhances feed proficiency, or forestall and treat infections in food producing animals (American Veterinary Medication Affiliation (AVMA), 2015).
Veterinary Anti-infection agents (VAs) are mainly used by cattle rearers and poultry farmers, that might prompt antibiotic residues from food animals to human, and consequently cause dangerous health hazards to the consumers [39]. High degree of antibiot¬ics residues consumption from animal products to human might affect immunological reactions and can unfavorably influence digestive microbiota in susceptible people [40]. Nonetheless, farmers utilize the VAs for various purposes such as prophylac¬tic, healing, growth promoters and at times both prophylactic and healing purposes throughout the globe [41]. In Canadian poultry industry for prophylaxis and growth enhancers are used for various types of antibiotics are used [42]. Enormous level of antibiotics are utilized in Bangladesh yearly. A large proportion of this are used unreasonably under conditions of lacking or no skilled supervision and as a rule without earlier testing on docu-mentation of the microorganisms and resilience of its sensitivity of the antibiotics recommended [43].
Fluoroquinolones including ciprofloxacin, enrofloxacin, nalidix-ic corrosive, and so on have been largely used for treatment and prophylaxis [44]. Ciprofloxacin is observed viable where micro¬organisms are resistant to aminoglycosides, tetracycline, mac-rolides and β-lactams [45]. Tetracycline serves various function such as treating infections and as a growth enhancer in animals owing to the fact that it is a broad-spectrum antibiotic. Around 60% of an ingested portion of oxytetracycline is assimilated from the gastrointestinal tract and broadly spread in the body [46-48]. As of late, many examinations have shown that antibi¬otics injected to poultry and domesticated animals were amassed in liver, kidney, muscle and bones surpassing the Maximum Re¬sidual Limits (MRL) (see table 1 below) [49]. The often use of antibiotics might bring about drug residues that can be found at various levels in animal products like milk or meat. Differ¬ent people have perceived presence of medications residues in food beyond the MRLs worldwide [50]. Presence of antibiotics residues in foodstuff can present risks to human wellbeing e.g., sensitivity to antibiotics, allergy responses, microflora imbal¬ance, bacterial resistance to antibiotics in microorganisms and loss to the food business. Notwithstanding, farmer suse them oc¬casionally for both prophylactic and remedial purposes globally [41]. Thus, the aim of the research is to aimed at evaluating of chemical level and antibiotic residue in raw meat and having the following objectives which are: to determine the level of chem¬ical and antibiotic residues in meat samples and to compare the level of chemical and antibiotic residues in the different meat from different abattoirs.
Table 1: Maximum Residual Limits (MRLS) Of Tetracyclines and Oxytetracycline in Animal Derived Foods According To the JECFA (2013)
|
S/N |
Antibiotic |
Tissue |
MRLs |
|
1 |
Tetracyclines (Oxytetracycline, |
Muscle |
200 μg/kg |
|
2 |
Tetracycline, chlortetracyclines) |
Liver |
600 μg/kg |
|
3 |
|
Kidney |
1200 μg/kg |
Study Area
Kano is the state capital of Kano State in North West, Nigeria. It is situated in the Sahelian geographic region, south of the Saha-ra. Kano is the commercial nerve centre of Northern Nigeria and is the second largest city in Nigeria. The Kano metropolis ini-tiallycovered 137 square kilometres (53 square miles), and com-prised six local government areas (LGAs) Kano Municipal, Fag-ge, Dala, Gwale, Tarauni and Nasarawa; However, it now covers two additional LGAs - Ungogo and Kumbotso. The total area of Metropolitan Kano is now 499 square kilometres (193 square miles), with a population of 2,828,861 as of the 2006 Nigerian census; the latest official estimate (for 2016) is 3,931,300. The principal inhabitants of the city are the Hausa people. However, there are many who speak Fulani language. As in most parts of northern Nigeria, the Hausa language is widely spoken in Kano. The city is the capital of the Kano Emirate.
Field Sample Collection
Table 2: Location and Number of Samples Collected From Kano Abattoir and Other Slaughter House within the State
|
LOCATION |
SAMPLE |
|
Kano Abattoir Limited |
1 |
|
Unguwa Uku Slaughter House |
2 |
|
Dawanau Slaughter House |
3 |
|
Badarawa Slaughter House |
4 |
|
Madubi Slaughter House |
5 |
|
Warawa Slaughter House |
6 |
All of the samples were obtained during the daytime.
Sampling Technique
Kidney, liver and muscle samples were randomly selected from six slaughterhouses in Kano state. Totaling 18 samples.
Sample Collection
A total of 18 samples (kidney, muscle and liver) were collected from Kano abattoir limited and other slaughterhouses from dif¬ferent local government of Kano state. Each sample was placed into a separate plastic container. The collected samples were stored in Freezer at -20°C and transferred to laboratory in a cool-er of ice and until extraction.
Apparatus
JASCO HPLC system with pump PU1580, a Rheodyne loop in-jector with injection volume 20uL, JASCO UV detector 1575 operated at 365nm, JASCO BORWIN software version 1.50 with Hercule-200 chromatography interface for results inte-gration and recording, HIQ Sil C18 LC column (main column size: 4.6mm internal diameter x 250mm length and guard col-umn size: 4.6mm internal diameter x 75mm length) and mobile phase made of 50mM aqueous oxalic acid solution containing 13% (V/V) each of methanol and acetonitrile were used.
Reagent
HPLC grade water filtered through 0.2µm with maximum impu-rities of 0.0003% and minimum transmission of 100% at 200 and 250 nm procured from Qualigens Fine chemicals, Glaxo-Smith-kline Pharmaceutical Ltd was used.
Extraction
2g of raw meat product was homogenized in a blender for 2 min and then 0.1g citric acid was added. To this mixture, 1ml nitric acid (30%), 4ml methanol and 1ml deionised water were added, respectively. The suspension with solid particles was put in a vortex for good mixing, kept in an ultrasonic bath for 15 min and then centrifuged for 10 min at 5300 rPM. After filtering through a 0.45µm nylon filter, 20µl of solution was injected into HPLC for analysis.
Data Analysis
The data was analyzed using mean, percentage and chart.
Results
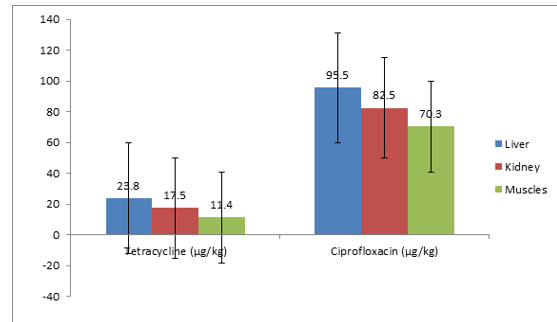
Figure 1: Mean Distribution of Tetracycline and Ciprofloxacin Residues in Tissues of Slaughtered Cattle in Sample 1
The result of sample 1 above of 1 slaughtered cattle. A total of 3 tissues (liver = 1; kidney = 1; muscle = 1) were screened for the presence of antibiotics residue. Tetracycline and ciprofloxa-cin were detected. The mean residue levels of tetracycline were 17.57 μg/kg ± 6.20 μg/kg and 82.77 μg/kg ± 12.60 μg/kg for ciprofloxacin respectively. The result shows multi-residues of antibiotics
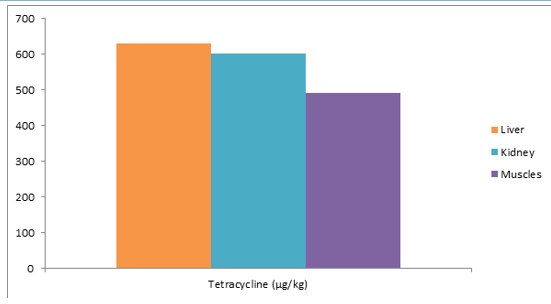
Figure 2: Mean Distribution of Tetracycline Residues in Tissues of Slaughtered Cattle in Sample 2
It can be observed that from the sample 2 above of 1 slaughtered cattle. A total of 3 tissues (liver = 1; kidney = 1; muscle = 1) were screened for the presence of drugs residue. Only tetracy¬cline was detected. The mean residue levels of tetracycline were 575.37 μg/kg ± 73.23 μg/kg.
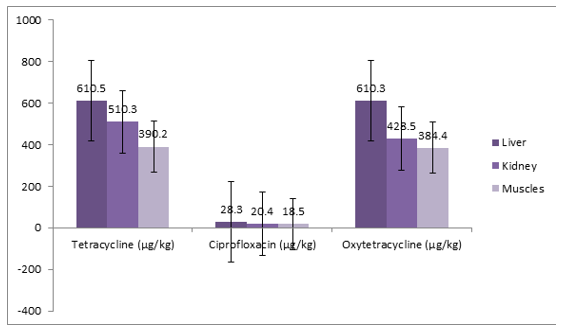
Figure 3: Mean Distribution of Tetracycline, Ciprofloxacin and Oxytetracycline Residues in Tissues of Slaughtered Cattle in Sample 3
From the above samples 3 of 1 slaughtered cattle. A total of 3 tissues (liver = 1; kidney = 1; muscle = 1) were screened for the presence of antibiotics residue. Tetracycline, ciprofloxacin and oxytetracycline were detected. The mean residue levels of tetracycline were 503.67 μg/kg ±110.30 μg/kg, ciprofloxacin 22.4 μg/kg ± 5.20 μg/kg and 474.4 μg/kg ± 119.74 μg/kg for oxytetracycline respectively. The result shows multi-residues of antibiotics.
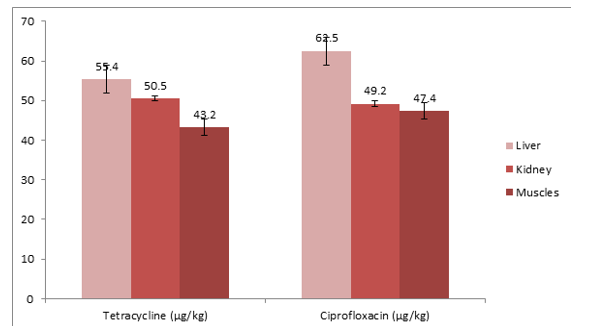
Figure 4: Mean Distribution of Tetracycline and Ciprofloxacin Residues in Tissues of Slaughtered Cattle in Sample 4
The sample 4 above tested shows positive for the two antibiot-ics tetracycline and ciprofloxacin residues. A total of 3 tissues (liver = 1; kidney = 1; muscle = 1) from 1 slaughtered cattle were screened for the presence of antibiotics residue. The result shows multi-residues of antibiotics. The mean residue levels of tetracycline were 49.7 μg/kg ± 6.14 μg/kg and 53.03 μg/kg ± 8.25 μg/kg for ciprofloxacin respectively
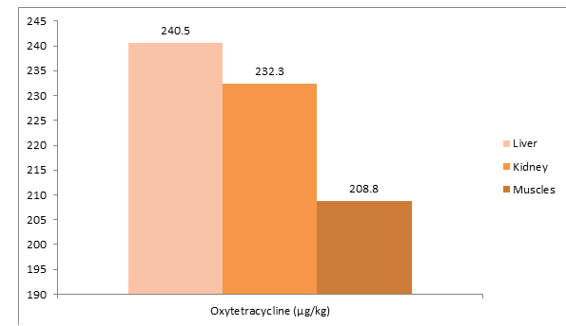
Figure 5: Mean Distribution of Oxytetracycline Residues in Tissues of Slaughtered Cattle in Sample 5
It can be observed from the above samples 5 of 1 slaughtered cattle. A total of 3 tissues (liver = 1; kidney = 1; muscle = 1) were screened for the presence of drugs residue. Oxytetracycline were detected. The mean residue levels of Oxytetracycline were 227.2 μg/kg ± 16.45 μg/kg.
Figure 6: Mean Distribution of Tetracycline and Ciprofloxacin Residues in Tissues of Slaughtered Cattle in Sample 6
The result of analysis in sample 6 above shows positive for the two antibiotic tetracycline and ciprofloxacin residues respective¬ly. A total of 3 tissues (liver = 1; kidney = 1; muscle = 1) from 1 slaughtered cattle were screened for the presence of antibiotics residue. The mean residue levels of tetracycline were 594 μg/ kg ± 47.71 μg/kg and 49.6 μg/kg ± 6.42 μg/kg for ciprofloxacin respectively. The result shows multi-residues of antibiotics.
Table 3: Result of Meat/Organ with Tetracycline, Ciprofloxacin and Oxytetracycline Residue from Kano Abattoir
|
Sample |
N |
Tetracycline |
Ciprofloxacin |
Oxytetracycline |
>MRLs |
|
Liver |
6 |
5 (83%) |
4 (67%) |
2 (33%) |
4 (22%) |
|
Kidney |
6 |
5 (83%) |
4 (67%) |
2 (33%) |
0 (0%) |
|
Muscles |
6 |
5 (83%) |
4 (67%) |
2 (33%) |
5 (28%) |
|
Total |
18 |
15 (249%) |
12 (201%) |
6 (99%) |
9 (50%) |
|
Where: N is the number of samples, MRL is maximum residue limit |
|||||
Discussion
Antibiotic use contributes to the emergence of drug resistant or-ganisms, these important drugs must be used judiciously in both animal and human medicine to slow the development of resis¬tance. The results of this study show the presence of residues of tetracycline, ciprofloxacin and oxytetracycline, which are some of the leading antimicrobials used in Nigeria, in all the tissues screened, with the highest concentrations found in the liver, fol¬lowed by the kidney and then the muscle, with the lowest levels. This is incomparable with a study completed by which found that the excessive concentrations of antibiotics are within the muscular tissues, accompanied by the liver and kidney with low¬est concentration [51]. This finding further gives proof that abuse of antimicrobial use, such as when used on food animals meant for almost immediately slaughter, ends in residues in tissues ob¬tained from table 3 above, out of 18 samples that were analyzed for tetracycline residues 15(83.3%) had detectable tetracycline residues, of those 15 samples, 5(83%) were from liver, 5(83%) were from kidney, and 5(83%) were from muscle. This study does not concur with findings of where out of the 250 samples that were analyzed for tetracycline residues, 114(45.6%) had de- tectable tetracycline residues. Of those 114 samples, 60(24%) were from liver, 35(14%) from kidney, and 19(7.6%) from mus¬cle. Although the concentration levels obtained in this study were above the maximum residual limits (100) recommended (JECFA, 2013) for some of the samples [52].
The result reported in figure 1 above comprising of liver = 1; kidney = 1; and muscle = 1 were screened for the presence of antibiotics residue. Tetracycline and ciprofloxacin were detect¬ed. The mean residue levels of tetracycline were 17.57 μg/kg ± 6.20 μg/kg and 82.77 μg/kg ± 12.60 μg/kg for ciprofloxacin respectively. The result shows multi-residues of antibiotics. This is consistent with finding of in Kenya with the mean tetracy-cline levels of cattle samples from the five slaughterhouses in the study were as follows: Athi River, 1,046 μg/kg; Dandora, 594 μg/kg; Ngong, 701 μg/kg; Kiserian, 524 μg/kg; and Dagoretti, 640 μg/kg. The mean levels of the detected tetracyclines were higher than the recommended maximum levels in edible tissues except in kidney of this present finding [53].
It can be observed that from the figure 2 above comprising of liver = 1; kidney = 1; and muscle = 1 were screened for the pres-ence of drugs residue. Only tetracycline was detected. The mean residue levels of tetracycline were high (575.37 μg/kg ± 73.23 μg/kg). The residue level in muscle is above permissible levels recommended by JECFA (2013). This is in contrast with work done by on title: Investigation of the risk of exposure to antimi¬crobial residues present in marketed milk in Tanzania with mean residue of 404.72μg/kg ± 382.01 μg/kg [54].
Mean whilst, From the figure 3 above comprising of liver = 1; kidney = 1; and muscle = 1 were screened for the presence of antibiotics residue. Tetracycline, ciprofloxacin and oxytetra-cycline were detected. The mean residue levels of tetracycline were 503.67 μg/kg ±110.30 μg/kg, ciprofloxacin 22.4 μg/kg ± 5.20 μg/kg and 474.4 μg/kg ± 119.74 μg/kg for oxytetracycline respectively. The result shows multi-residues of antibiotics. This study disagrees with research by Sarker et al., (2018) where, out of the 160 samples that were analyzed for antibiotics cipro-floxacin residues. The percentage of the samples were 52% and 42% for liver and muscle respectively. The concentration levels obtained in this study were below the maximum residual limits recommended (WHO, 1999; JECFA, 2013) except for the high concentration of Tetracyclin and oxytetracyclin in the muscles. The figure 4 above detected two antibiotics tetracycline and cip-rofloxacin residues. The mean residue levels of tetracycline were 49.7 μg/kg ± 6.14 μg/kg and 53.03 μg/kg ± 8.25 μg/kg for cipro-floxacin respectively. This concurs with a report by where out of the 180 samples that were analyzed for oxytetracycline residues, 98(54.44%) comprising of 48(80%) liver, 33(55%) kidney and 17(28.3%) muscle samples had detectable level of oxytetracy-cline residues [55]. Out of the positive sample, 62(63.2%) had oxytetracycline residue at violative levels while 36(36.8%) had residues level below the WHO/FOA recommended MRLs for oxytetracycline in meat, liver and kidney with mean residue lev-els of 1197.0 μg/kg; 372.7 μg/kg and 51.80 in μg/kg in liver, kidney and muscles and 503.94 μg/kg, 492.07 μg/kg, 259.52 μg/ kg for ciprofloxacin respectively.
It can be observed from figure 5 above comprising of liver = 1; kidney = 1; and muscle = 1 were screened for the presence of drugs residue. Oxytetracycline were detected. The mean residue levels were 227.2 μg/kg ± 16.45 μg/kg which is below the max-imum permissible levels except in muscle. This is in agreement a study by in south-western Nigeria. The findings revealed res-idues of oxytetracycline (kidney: 9.47 μ/kg ± 3.24 μ/kg; liver: 12.73 μ/kg ± 4.39 μ/kg; muscle: 16.17 μ/kg ± 5.52 μ/kg). Al-though finding from these studies revealed that concentration of Oxytetracyclin in muscles where a little above the permissible limits [56].
Results from figure 6 above detected two antibiotic tetracycline and ciprofloxacin residues respectively. This include liver = 1; kidney = 1; and muscle = 1 were screened for the presence of an¬tibiotics residue. The result shows multi-residues of antibiotics. The mean residue levels of tetracycline were 594 μg/kg ± 47.71 μg/kg and 49.6 μg/kg ± 6.42 μg/kg for ciprofloxacin respective¬ly. This is consistent with finding done by in Nairobi slaughter house in Kenya except in kidney which is below permissible lev¬els with mean residue of 366.8 μg/kg ± 421.7 μg/kg for tetracy¬cline. Although, concentration of Tetracycline in this study was above the WHO recommended Maximum Residual Limits [57].
The concentration level of Tetracycline in muscle samples in sample 2, oxytetracycline and tetracycline in muscles in sample 3, and tetracycline in muscles in samples 6 obtained in this study were above the maximum residual limits recommended [58]. This is so within the light of bioaccumulation whilst these resi¬dues present in the animal tissues collect continuously over the lifespan of the individuals through extended intake. This is par¬ticularly of potential problem in Nigeria in which cattle are the most regularly ingested animals and consumption of uncooked unpasteurized milk is normal among cattle owners, which can have a potentially bad impact on children, who are expected to consume more milk. Moreover, tetracycline, ciprofloxacin and oxytetracycline can input the human frame from other means, which includes poultry meat, given the uncontrolled abuse of antibiotics amongst food animals in Nigeria [59-61].
The low levels detected in some of the tissues sampled could be attributed to the probably low doses of antimicrobials commonly administered by livestock traders in order to maximize the num¬ber of doses available, an assertion supported by an earlier re¬port that nomadic herdsmen administer chemotherapeutic agents without veterinary prescription and most likely at incorrect dos¬ages [54]. In another study conducted in southwestern Nigeria, strains of E. coli resistant to tetracycline and penicillin-G from both animal and human samples were reported [62]. In general, reports from different parts of Nigeria have observed temporal trends in the prevalence of resistance amongst enteric organisms such as E. coli and Shigella [63]. This practice by livestock trad¬ers and herders therefore becomes an issue that needs urgent at-tention if efforts towards stamping out the menace of antimicro¬bial resistance in animals and humans.
Conclusion
The combination of several substances with diverse use as well as emission patterns, affecting a host of diverse endpoints in a plethora of exposed species in the vastly diverse ecosystems of the world, plus human health consideration, makes the deriva¬tion of a single quantitative antibiotic’s residue level in raw meat a daunting, if not impossible task. However, antibiotic traces have been found in the meat of cattle slaughtered for human con¬sumption in Kano state [64]. The findings in this study elucidat¬ed the importance of antibiotics usage in cattle and poultry and provided the quantitative analysis of the prevalence and levels of antibiotic residues. This study also provided quantitative data on the prevalence of oxytetracycline, tetracycline and ciprofloxacin residue in cattle meat that were being consumed in city of Kano, Nigeria. The results of this study revealed that greater proportion of the meat being consumed in Kano contained oxytetracycline and tetracycline residues above international food safety stan¬dards. The high proportion of cattle meat samples containing residues of antibiotics could be due to the misuse and lack of strict regulation and control of antimicrobial use Nigeria live¬stock production. This therefore requires urgent public health attention by the consumers and appropriate regulatory agencies in the country.
Recommendations
The misuse of antibiotics may result in the different health haz-ards. Thus, the reduction of antibiotic use constitutes a challenge for the world. In order to achieve such a reduction, the following nine steps should possibly be considered with regard to all an-tibiotics:
1. Routine inspection should be adopted by environmental health officers to ensure drugs residues in animal product.
2. Health education and sensitization of herdsmen and live-stock industry should be established by environmental health officer for the negative impact of antibiotics residue on human health.
3. The effective prevention of infectious diseases and the adoption of strict hygiene standards and rearing skills may reduce our need for antibiotics, particularly in the veterinary field.
4. The use of alternatives to antibiotics, such as plant-derived antimicrobial substances and probiotics, may represent a promising option; vaccination against some bacterial dis-eases may be of great value in the near future.
5. Strict national legislation must be passed in Nigeria to avoid the unnecessary use of antibiotics.
6. National monitoring of antibiotic residues in foods and up-dating of the maximum permissible limits of these residues should be undertaken. In Table 1, we state the maximum limits on commonly observed antibiotic residues in food-stuffs.
7. Avoid using antibiotics in the veterinary field without a vet-erinarian’s prescription
8. The heat treatment of meat, milk, and eggs may inactivate antibiotic contaminants in feedstuffs.
9. The freezing of animal-derived foods may also contribute to the reduction of some antibiotic contamination.
References
- Raimi, M., Muhammadu, I. H., UDENSI, L. O., & Akpojubaro, E. H. (2020). Assessment of safety practices and farmers behaviors adopted when handling pesticides in rural Kano state, Nigeria. Olalekan RM, Muhammad IH, Okoronkwo UL, Akopjubaro EH (2020). Assessment of safety practices and farmer’s behaviors adopted when handling pesticides in rural Kano state, Nigeria. Arts & Humanities Open Access Journal, 4(5), 191-201.
- Muhammad, I. H., Olalekan, R. M., Olawale, S. H., Emmanuel, O. O., Getso, B. B., & Haladu, S. (2020). Qualitative Adverse Health Experience Associated with Pesticides Usage among Farmers from Kura, Kano State, Nigeria. Merit Research Journal of Medicine and Medical Sciences (ISSN: 2354-323X) Vol, 8(8), 432-447.
- Isah, H. M., Raimi, M. O., & Sawyerr, H. O. (2021). Probabilistic Assessment of Self-Reported Symptoms on Farmers Health: A Case Study in Kano State for Kura Local Government Area of Nigeria. Available at SSRN.
- Isah, H. M., Raimi, M. O., & Sawyerr, H. O. (2021). Patterns of Chemical Pesticide Use and Determinants of Self-Reported Symptoms on Farmers Health: A Case Study in Kano State for Kura Local Government Area of Nigeria. Research on World Agricultural Economy, 2(01).
- Isah, H. M., Sawyerr, H. O., Raimi, M. O., Bashir, B. G., Haladu, S., & Odipe, O. E. (2020). Assessment of Commonly Used Pesticides and Frequency of Self-Reported Symptoms on Farmers Health in Kura, Kano State, Nigeria. Journal of Education and Learning Management (JELM),HolyKnight, 1, 31-54.
- Raimi, M. O., Vivien, O. T., Alima, O., Akpojubaro, E. H., & Williams, E. A. (2021). Articulating the Effect of Pesticides Use and Sustainable Development Goals (SDGs): The Science of Improving Lives through Decision Impacts.
- Morufu Olalekan Raimi. (2021).“Self-reported Symptoms on Farmers Health and Commonly Used Pesticides Related to Exposure in Kura, Kano State, Nigeria”. Annals of Community Medicine & Public Health, 1(1), 1002.
- Olalekan MR, Abiola I, Ogah A, Dodeye EO. (2021). Exploring How Human Activities Disturb the Balance of Bio-geochemical Cycles: Evidence from the Carbon, Nitrogen and Hydrologic Cycles. Research on World Agricultural Economy. Volume 02, Issue 03.
- Khattab, W. O., Elderea, H. B., Salem, E. G., & Gomaa, N.F. (2010). Transmission of administered amoxicillin drug residues from laying chicken to their commercial eggs. J Egypt Public Health Assoc, 85(5-6), 297-316.
- Adewuyi, GO, Olatoye, OI, Abafe, AO, Otokpa, MO, Nku-kut, NK. (2011). High Performance Liquid Chromatograph-ic Methods for Evaluation of Two Antibiotic Residues in Liver and Muscle of Broilers in Ibadan City, Southern Nigeria. Journal of Pharmaceutical and Biomedical Sciences, 11(6), 1–4.
- Attari, V. E., Abbasi, M. M., Abedimanesh, N., Ostadra-himi, A., & Gorbani, A. (2014). Investigation of enrofloxacin and chloramphenicol residues in broiler chickens carcasses collected from local markets of Tabriz, Northwestern Iran. Health Promotion Perspectives, 4(2), 151.
- Karmi, M. (2014). Detection and presumptive identification of antibiotic residues in poultry meat by using FPT. Glob J Pharmacol, 8(2), 160-5.
- Lawal, J. R., Jajere, S. M., Geidam, Y. A., Bello, A. M., Wakil, Y., & Mustapha, M. (2015). Antibiotic residues in edible poultry tissues and products in Nigeria: A potential public health hazard. International Journal of Animal and Veterinary Advances, 7(3), 55-61.
- Raimi MO, Sabinus CE. (2017). An Assessment of Trace Elements in Surface and Ground Water Quality in the Ebo-cha-Obrikom Oil and Gas Producing Area of Rivers State, Nigeria. International Journal for Scientific and Engineering Research (Ijser): Volume 8, Issue 6, July Edition. ISSN: 2229-551
- Raimi, M. O., Pigha, T. K., & Owobi, O. E. (2017). Water-Related Problems And Health Conditions In The Oil Producing Communities In Central Senatorial District Of Bayelsa State. Imperial Journal of Interdisciplinary Research (IJIR) Vol-3, Issue-6.
- Raimi, M., Nimisngha, D., Odipe, O. E., & Olalekan, A. S. (2018). Health risk assessment on heavy metals ingestion through groundwater drinking pathway for residents in an oil and gas producing area of Rivers State, Nigeria. Open Journal of Yangtze Oil and Gas, 3, 191-206.
- Suleiman, R. M., Raimi, M., & Sawyerr, O. H. (2019). A deep dive into the review of national environmental standards and regulations enforcement agency (nesrea) act. Suleiman Romoke Monsurat, Raimi Morufu Olalekan and Sawyerr Henry Olawale (2019) A Deep Dive into the Review of National Environmental Standards and Regulations Enforcement Agency (NESREA) Act. International Re-search Journal of Applied Sciences. pISSN, 2663-5577.
- Raimi, M., Adedotun, A. T., Emmanuel, O. O., & Anu, B. (2019). An analysis of bayelsa state water challenges on the rise and its possible solutions. Raimi Morufu Olalekan., et al. An Analysis of Bayelsa State Water Challenges on the Rise and Its Possible Solutions'. Acta Scientific Agriculture, 3(2019), 110-125.
- Raimi MO, Bilewu OO, Adio ZO, Abdulrahman H. (2019). Women Contributions to Sustainable Environments in Nigeria. Journal of Scientific Research in Allied Sciences. 5(4), 35-51. ISSN NO. 2455-5800.
- Raimi, M., Suleiman, R. M., Odipe, O. E., Tolulope, S. J., Modupe, O., Olalekan, A. S., & Christianah, M. B. (2019). Women role in environmental conservation and development in Nigeria. Ecol Conserv Sci, 1(2).
- Raimi, M., Ayibatonbira, A. A., Anu, B., Odipe, O. E., & Deinkuro, N. S. (2019). 'Digging Deeper'Evidence on Water Crisis and Its Solution in Nigeria for Bayelsa State: A Study of Current Scenario. Int J Hydro, 3(4), 244-257.
- Okoyen, E., Raimi, M., Oluwatoyin, O., & Williams, E. A. (2020). Governing the environmental impact of dredging: Consequences for marine biodiversity in the niger delta region of Nigeria.Insights Mining Science and technology, 2(3), 555586.
- Raimi, M. (2020). A review of environmental, social and health impact assessment (Eshia) practice in Nigeria: a panacea for sustainable development and decision-making. MOJ Public Health, 9(3-2020).
- Raimi, M. (2020). A Critical Review of Health Impact Assessment: Towards Strengthening the Knowledge of Decision Makers Understand Sustainable Development Goals in the Twenty-First Century: Necessity Today; Essentiality Tomorrow. Research and Advances: Environmental Sciences, ISSN, 2652-3655.
- Raimi, M. O., Odipe, O. E., Anu, B., Omini, D. E., Akpojubaro, E. H., & Owobi, O. E. (2020). Leaving no One Behind? Drinking Water Challenge on the Rise in Niger Delta Region of Nigeria: A Review. Drinking Water Challenge on the Rise in Niger Delta Region of Nigeria: A Review (May 2, 2020).
- Raimi, M. O., Oluwatoyin, O. A., & Olalekan, A. (2020). Health Impact Assessment: A tool to Advance the Knowledge of Policy Makers Understand Sustainable Development Goals:A Review. ES Journal of Public Health, 1(1), 1002.
- Raimi, M., Adindu, I. B., Udensi, E. O., Funmilayo, A. A., Opufou, T., Deinkuro, N. S.,& Adeniji, A. O. (2020). Health Impact Assessment: Expanding Public Policy Tools for Promoting Sustainable Development Goals (SDGs) in Nigeria. EC Emergency Medicine and Critical Care.
- Raimi, M. O., Emeka, C. L., Okoyen, E., Clement, A., Og-bointuwei, C., & Atoyebi, B. (2021). Making Better Informed, More Confident COVID-19 Decisions: Vaccine Hesitancy, Its Barriers and Impact Studies: Taking Bayelsa State as an Example. Int J Vaccine Immunizat, 5(1).
- Raimi, M. O., Mcfubara, K. G., Abisoye, O. S., Ezekwe, C. I., Sawyerr, O. H., & Gift, R. A. A. (2021). Responding to the Call through Translating Science into Impact: Building an Evidence-Based Approach to Effectively Curb Public Health Emergencies [COVID-19 Crisis].
- Raimi, M. O., Ezekwe, C. I., & Sawyerr, H. O. (2021). Problematic groundwater contaminants: impact of surface and ground water quality on the environment in ebocha-obri-kom oil and gas producing area of rivers state, nigeria. Nigeria. (September 17, 2021).
- Raimi, M. O., Gift, R. A. A., & Adias, T. C. (2021). ‘Si-lent Pandemic’: Evidence-Based Environmental and Public Health Practices to respond to the COVID-19 Crisis (pre-print).
- Raimi, M. O., Ezekwe, C. I., & Abiodun, B. (2021). Statistical And Multivariate Techniques To Trace The Sources Of Ground Water Contaminants And Affecting Factors Of Groundwater Pollution In An Oil And Gas Producing Wetland In Rivers State, Nigeria. medRxiv.
- Raimi, O. M., Sawyerr, O. H., Ezekwe, C. I., & Salako, G. (2021). Many Oil Wells, One Evil: Potentially toxic metals concentration, seasonal variation and Human Health Risk Assessment in Drinking Water Quality in Ebocha-Obrikom Oil and Gas Area of Rivers State, Nigeria. medRxiv.
- Raimi, M. O., Sawyerr, O. H., Ezekwe, C. I., & Olaniyi,O. A. (2021). Quality water not everywhere: Exploratory Analysis of Water Quality across Ebocha-Obrikom Oil and Gas Flaring Area in the Core Niger Delta Region of Nigeria.
- Morufu Olalekan Raimi, Tonye Vivien Odubo & Adedoyin Oluwatoyin Omidiji (2021) Creating the Healthiest Nation: Climate Change and Environmental Health Impacts in Nigeria: A Narrative Review. Scholink Sustainability in Environment. ISSN.
- Odipe, O. E., Raimi, M., Deinkuro, N. S., Funmilayo, A. A., innocent Edewor, O. P., Lateefat, H. M., & Fadeyibi,M. (2019). Assessment of Environmental Sanitation, Food Safety Knowledge, Handling Practice among Food Handlers of Bukateria Complexes in Iju Town, Akure North of Ondo-State, Nigeria. Acta Scientific Nutritional Health, 3(6).
- Segun, A. A., & Raimi, M. O. (2021). When Water Turns Deadly: Investigating Source Identification and Quality of Drinking Water in Piwoyi Community of Fed-eral Capital Territory, Abuja Nigeria. Online Journal of Chemistry, 1(1), 38-58.
- Ramos, M., Aranda, A., de Pozuelo, M. M., & Reuvers, T. (2003). Chloramphenicol residues in food samples: their analysis and stability during storage. Journal of liquid chromatography & related technologies, 26(15), 2535-2549.
- Chanda, R. R., Fincham, R. J., & Venter, P. (2014). Review of the Regulation of Veterinary Drugs and Residues in South Africa. Critical reviews in food science and nutrition, 54(4), 488-494.
- Ramatla, T., Ngoma, L., Adetunji, M., & Mwanza, M. (2017). Evaluation of antibiotic residues in raw meat using different analytical methods. Antibiotics, 6(4), 34.
- Guetiya Wadoum, R. E., Zambou, N. F., Anyangwe, F. F., Njimou, J. R., Coman, M. M., Verdenelli, M. C., ... & Colizzi, V. (2016). Abusive use of antibiotics in poultry farming in Cameroon and the public health implications. British poultry science, 57(4), 483-493.
- Chapman, H. D., & Jeffers, T. K. (2014). Vaccination of chickens against coccidiosis ameliorates drug resistance in commercial poultry production. International Journal for Parasitology: Drugs and Drug Resistance, 4(3), 214-217.
- Sarker, Y. A., Hasan, M. M., Paul, T. K., Rashid, S. Z., Alam, M. N., & Sikder, M. H. (2018). Screening of antibiotic residues in chicken meat in Bangladesh by thin layer chromatography. Journal of Advanced Veterinary and Animal Research, 5(2), 140-145.
- Modi, C. M., Patel, H. B., & Mody, S. K. (2013). Animal husbandry practice to contaminants and residues of chemical in animal origin foods and health hazard. International Journal of Molecular Veterinary Research, 3(1).
- Van Boeckel, T. P., Gandra, S., Ashok, A., Caudron, Q., Grenfell, B. T., Levin, S. A., & Laxminarayan, R. (2014). Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. The Lancet infectious diseases, 14(8), 742-750.
- Ekuttan, C. E., Kang’ethe, E. K., Kimani, V. N. and Randolph, T. F. (2007). Investigation on the prevalence of antimicrobial residues in milk obtained from urban smallholder dairy and non-dairy farming households in Dagoretti Division, Nairobi, Kenya. East Afr. Med. J., 84(11): 87-91
- Eltayb, A., Barakat, S., Marrone, G., Shaddad, S., & Stålsby Lundborg, C. (2012). Antibiotic use and resistance in animal farming: a quantitative and qualitative study on knowledge and practices among farmers in Khartoum, Sudan. Zoonoses and Public Health, 59(5), 330-338.
- Mund, M. D., Khan, U. H., Tahir, U., Mustafa, B. E., & Fayyaz, A. (2017). Antimicrobial drug residues in poultry products and implications on public health: A review. International Journal of Food Properties, 20(7), 1433-1446.
- Kempe, M., Cederfur, J., Zihui, M., Pei, Y., Wistrand, U. J., Fiaccabrino, G. C., & van Rhijn, H. (2000). CREAM—Car-tridges with Molecularly Imprinted Recognition Elements for Antibiotic residues Monitoring in Milk. In Proceedings of the EuroResidues IV Conference, Veldhoven, the Netherlands.
- Adesokan, H. K., Agada, C. A., & Adetunji, V. O. (2013). Oxytetracycline and penicillin-G residues in cattle slaughtered in southwestern Nigeria: Implications for livestock disease management and public health. Journal of the South African Veterinary Association, 84(1), 1-5.
- Muriuki, F. K., Ogara, W. O., Njeruh, F. M., & Mitema, E.S. (2001). Tetracycline residue levels in cattle meat from Nairobi salughter house in Kenya. Journal of Veterinary Science, 2(2), 97-101.
- Kurwijila, L. R., Omore, A., Staal, S., & Mdoe, N. S. Y. (2006). Investigation of the risk of exposure to antimicrobial residues present in marketed milk in Tanzania. Journal of food protection, 69(10), 2487-2492.
- Olufemi, O. I., & Agboola, E. A. (2009). Oxytetracycline residues in edible tissues of cattle slaughtered in Akure, Nigeria. Internet Journal of food safety, 11, 62-66.
- Joint, F. A. O., WHO Expert Committee on Food Additives, & World Health Organization. (1999). Evaluation of certain veterinary drug residues in food: fiftieth report of the Joint FAO/WHO Expert Committee on Food Additives. World Health Organization.
- Adetunji, M. O., & Rauf, M. O. (2012). Analysis of household demand for meat in southwest, Nigeria. Global Journal of Science Frontier Research, 12(1).
- Gambo, P., Maguda, A. S., Adole, J. A., Dyek, D. Y., Ifende,V. I., Bot, C., & Adedeji, A. J. (2018). A survey of viral diseases of livestock characterized by skin lesions in Kanam Local Government Area of Plateau State, Nigeria. Nigerian Veterinary Journal, 39(3), 250-262.
- Cadmus, S., Palmer, S., Okker, M., Dale, J., Gover, K., Smith, N., & Gordon, S. V. (2006). Molecular analysis of human and bovine tubercle bacilli from a local setting in Nigeria. Journal of clinical microbiology, 44(1), 29-34.
- Aibinu, I., Adenipekun, T., Adelowotan, T., Ogunsanya, T., & Odugbemi, T. (2007). Evaluation of the antimicrobial properties of different parts of Citrus aurantifolia (lime fruit) as used locally. African Journal of Traditional, Complementary, and Alternative Medicines, 4(2), 185.
- Okeke, I. N., Laxminarayan, R., Bhutta, Z. A., Duse, A. G.,Jenkins, P., O'Brien, T. F., & Klugman, K. P. (2005). Antimicrobial resistance in developing countries. Part I: recent trends and status. The Lancet infectious diseases, 5(8), 481-493.
- American Veterinary Medicine Association (AVMA). (2015).Antimicrobial use and antimicrobial resistance.
- Joint FAO/WHO Expert Committee on Food Additives (JECFA). (2013). Residue evaluation of certain veterinary drugs. 8th Meeting, FAO/ JECFA Monographs, pp. 15
- Olufemi, O. I., & Agboola, E. A. (2009). Oxytetracycline residues in edible tissues of cattle slaughtered in Akure, Nigeria. Internet Journal of food safety, 11, 62-66.
- Raimi MO, Sawyerr HO and Isah HM. (2020). Health risk exposure to cypermethrin: A case study of kano state, Nigeria. Journal of Agriculture. 7th International Conference on Public Healthcare and Epidemiology. September 14-15, 2020 | Tokyo, Japan
- Sattar, S. Hassan, MM.Islam, SKMA. Alam, M. Faruk, MSA. Chowdhury, S. Saifuddin, AKM (2014). Antibiotic Residues in Broiler and Layer Meat in Chittagong District of Bangladesh. Veterinary World, 7(9), 738–743.


