Review Article - (2024) Volume 2, Issue 2
A Review on Diagnostic Potentials of Undecalcified Tissue Sections in Histopathology
2Department of Medical Laboratory Sciences, School of Allied Health Sciences, Kampala International U, Uganda
3Department of Medical Laboratory Science, Niger Delta University Wilberforce Island Bayelsa State, Uganda
4Department of Histopathology/Cytology, Faculty of Medical Laboratory Science, Federal University Otu, Nigeria
Received Date: Oct 25, 2024 / Accepted Date: Dec 09, 2024 / Published Date: Dec 11, 2024
Copyright: ©©2024 Yibala Ibor Oboma, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ohiri, F, C., Oboma, Y, I., Ekpenyong, B, O., Agbe, P, T., Mordecai, O, G. (2024). A Review on Diagnostic Potentials of Undecalcified Tissue Sections in Histopathology. Japan J Med Sci, 2(2), 01-09.
Abstract
The preparation of undecalcified tissue sections is essential for accurate histopathology evaluation of mineralized tissues. The preparation involves fixation, embedding, sectioning processes, and special staining techniques, their principles tailored for hard tissues such as Von Kossa method, Alizarin Red S method, alongside a comprehensive overview of the materials and equipment required. Frequently requested tissue types include; bone biopsies, dental samples and mineralized cartilage, often analyzed in the context of condition such as osteoporosis, dental caries, and Osteosarcoma. Despite the advantages, the technique has limitations, including the need for specialized training requirement, specialized equipment and expertise, as well as potential challenges in achieving uniform sectioning. Overall, this paper explores the importance of undecalcified section preparation in advancing histopathology research and clinical diagnosis, paving the way for enhanced understanding of bone-related conditions.
Keywords
Calcium, Undecalcified, Sections, Bones
Introduction
Calcification refers to the abnormal accumulation of calcium salts in tissues, which can occur in both normal and diseased states. This process is critical in histopathology as it affects tissue morphology and can interfere with standard histological procedures such as sectioning and staining. Understanding calcification is essential in clinical diagnostics and research, particularly when dealing with bone, cardiovascular, and renal pathologies [1].
Calcification in tissues is broadly classified into two types: dystrophic and metastatic calcification. Each type has distinct causes, mechanisms, and pathological implications. Dystrophic Calcification: This calcification occurs in areas of tissue injury, necrosis, or chronic damage, despite normal calcium and phosphate levels in the blood [2]. It is commonly seen in conditions like atherosclerosis, old scars, and areas of long-term inflammation or injury. For example, calcification in damaged heart valves or the formation of calcified plaques in blood vessels are manifestations of dystrophic calcification. The process is initiated when cellular injury or death leads to the release of phosphates, which bind with calcium ions, forming insoluble calcium salts that deposit in tissues [3].
Metastatic Calcification results from elevated serum calcium levels (hypercalcemia), often seen in patients with disorders such as hyperparathyroidism, chronic kidney disease, or vitamin D intoxication [4]. Unlike dystrophic calcification, metastatic calcification affects normal tissues, primarily in organs that excrete or filter blood, such as the lungs, kidneys, and gastric mucosa. The high calcium concentration leads to precipitation of calcium salts, disrupting tissue function and contributing to organ dysfunction over time [5]. The formation of calcium deposits in tissues is primarily driven by the balance between calcium and phosphate concentrations in body fluids [1]. In both types of calcification, mineralization follows similar pathways where phosphate ions combine with calcium to form calcium hydroxyapatite, a crystalline structure that mimics bone formation. This deposition is influenced by local tissue factors such as pH, the presence of phosphatases, and the release of cellular debris from injured or dying cells [6].
Calcification has significant clinical implications, especially in diagnosing and managing diseases. In cardiovascular pathology, for instance, dystrophic calcification of atherosclerotic plaques is associated with an increased risk of heart attack and stroke due to the reduced flexibility of blood vessels and potential plaque rupture [2]. Similarly, metastatic calcification in organs such as the kidneys can exacerbate conditions like chronic kidney disease, leading to further renal impairment [4]. In oncology pathology, calcification can serve as a marker for certain malignancies, where it may indicate necrosis within a tumor or the response of tissues to chronic injury. The presence of calcium salts hardens tissues, making it difficult to obtain thin sections for microscopy. This necessitates the use of decalcification techniques to soften the tissues while preserving their structural and cellular integrity [5]. Understanding the nature and extent of calcification in tissues allows pathologists to choose appropriate methods of decalcification, ensuring accurate diagnosis and analysis.
Undecalcified Sections
An undecalcified section refers to a tissue sample that has not undergone decalcification, preserving their mineral composition, particularly calcium and phosphate [7]. Undecalcified sections are critical in histopathology examinations, particularly for studying mineralized tissues such as bone, teeth, and cartilage. Unlike undecalcified sections, which involve removing calcium from tissues, undecalcified sections preserve the natural mineral content. This preservation allows for more accurate morphological, cellular and structural analyses, making them invaluable in both research and clinical diagnostics [8]. The preparation of undecalcified sections is a crucial technique in histopathology examination, especially when dealing with hard tissues like bone and teeth. This method involves the processing of hard tissues without removing calcium with the aim to maintain the structural integrity of mineralized tissues while enabling detailed examination under a microscope.
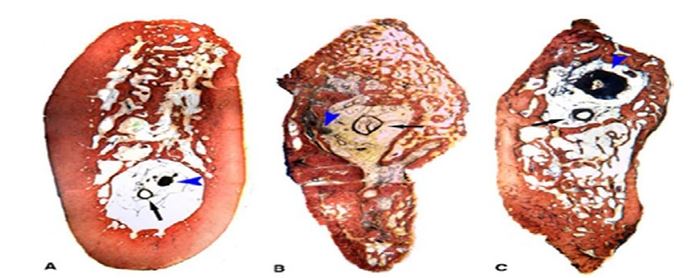
Figure 1: Histology Section of Undecalcified Tissue. A: Control Tissue; B: Regenerate Tissue at 12 Weeks; C: Regenerate Tissue at 18 Weeks [9]
Aim of Preparing Undecalcified Sections
The aim of preparing undecalcified tissue sections is to preserve the mineral component and structural integrity of calcified tissues, such as bone and teeth. This preparation allows for detailed examination of the tissue’s morphology, mineral distribution, and cellular interactions without the artifacts that may arise from decalcification processes [10].
Undecalcified sections enables the use of advanced imaging techniques, such as fluorescent microscopy, enhancing the understanding of tissue dynamics and pathology [7].
Undecalcified sections maintain the three-dimensional structure of bone, which is crucial for assessing micro-architectural features such as trabecular and cortical bone integrity [11]. These sections allow for the evaluation of mineral content and distribution, which is particularly important in studying conditions like osteoporosis, osteomalacia, and other metabolic bone diseases [12]. Undecalcified samples preserve cellular components such as osteoblasts, osteoclasts, and bone lining cells, allowing for detailed examination of bone remodeling processes without distortion from decalcification [13]. Also, Certain histological stains and immunohistochemical techniques are more effective on undecalcified specimens. This enhances the visualization of specific proteins, markers, and histological features. They are better demonstrated in undecalcified sections [14].
The method aids in understanding the pathophysiology of bone diseases, retaining the context of mineralization and cellular activity, which is crucial for diagnosing conditions like Paget's disease and osteosarcoma [15].
Methodology
a. Fixatives: Proper fixation is essential for preserving tissue architecture. Commonly used fixatives include: i). Formalin: A widely used formaldehyde fixative, ideal for routine histology.
ii). Glutaraldehyde: Offers superior preservation for electron microscopy due to its ability to cross-link proteins [16].
b. Embedding Materials: Resins are crucial for embedding undecalcified specimens, ensuring they maintain structural integrity during sectioning: i). Methyl Methacrylate (MMA) allows for clear sections and is compatible with various staining techniques. ii). Epoxy Resins: Provide excellent hardness and are suitable for ultra-thin sectioning for electron microscopy.
c. Sectioning Equipment- Microtomes with diamond knives for ultra-thin sections, particularly in electron microscopy.
d. Staining- Conventional and histochemical stains are use in demonstrating morphology and calcium deposits in undecalcified sections respectively.
Types of Tissues Suitable for Undecalcified Sections
a. Bone: Undecalcified bone Sections Provides insights into various diseases, including osteoporosis and osteosarcoma. Cortical bone provides strength and support and is crucial for weight-bearing functions. It is often analyzed to assess structural integrity and changes related to conditions such as osteoporosis and fractures [11]. Undecalcified sections of cancellous bone are critical for studying conditions like osteoporosis, as they are more metabolically active and sensitive to changes in bone density [17]. Woven Bone is frequently analyzed in cases of fracture healing and conditions such as osteosarcoma, where the assessment of healing patterns is crucial [18]. Lamellar bone is characterized by a regular, layered structure. Analysis of lamellar bone in undecalcified sections is vital for understanding bone remodeling processes and diseases affecting bone quality [19].
v). Bone Marrow, although not bone per se, bone marrow is critical for hematologists and is often included in undecalcified sections for comprehensive analysis of bone and marrow interactions. Bone marrow analysis in conjunction with bone sections is essential for diagnosing hematological disorders [20].
b. Teeth: Useful in dental research and pathology.
c. Cartilage: Important for studying joint diseases and developmental biology.
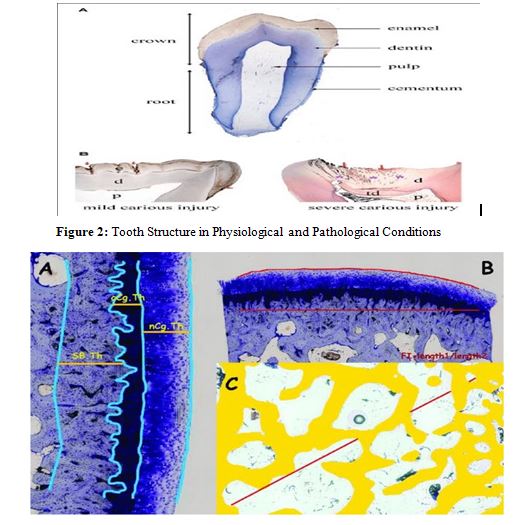
Figure 3: Representative Images of Measurements Made in Undecalcified Sections: a. Cartilage and Subcondral Bone Cortical Thickness (nCg.Th; cCg.Th; Cg.Th = nCg.Th + cCg.Th) SB.Th). b. Surface Undulation (FI). c. Trabecular Subchondral Bone Measurements in a ROI. Tb.A: % of Trabecular Bone in the ROI; Tb.Sp Measured on the Diagonal of ROI (Tb.Sp = (1/Tb.N)-Tb. Th).
Measurement of Calcium Deposits in Undecalcified Sections
The measurement of calcium deposits in undecalcified tissue sections is crucial for understanding bone health, metabolism, and pathology. This technique allows researchers to analyze mineralization patterns and assess the impact of various conditions on bone structure.
Methods for measuring calcium deposits in undecalcified sections includes; Histological Staining Techniques: Histological stains are commonly used to visualize calcium deposits in undecalcified sections. Two widely employed staining methods include:
i). Alizarin Red S Staining: This dye specifically binds to mineralized tissues, allowing for the visualization of calcium deposits in bone sections. It is particularly useful for quantifying mineralization in various disease states [21]. ii). Von Kossa Staining: This technique employs silver nitrate to identify calcium deposits. It is effective for highlighting areas of mineralization and is often used in conjunction with other stains for comprehensive analysis [22].
Micro-computed Tomography (Micro-CT): Micro-CT is a powerful imaging technique that allows for the non-destructive visualization and quantification of calcium deposits in bone. It provides high-resolution three-dimensional images, enabling detailed analysis of mineralization patterns [23].
Quantitative Analysis Using Image Analysis Software: Software tools can be employed to quantify calcium deposits from histological images. These tools analyze stained sections, allowing for the measurement of area, intensity, and distribution of calcium [24].
Chemical Assays for Calcium Measurement: Chemical assays can be used in conjunction with undecalcified sections to quantitatively measure calcium content. Methods such as atomic absorption spectroscopy (AAS) or inductively coupled plasma mass spectrometry (ICP-MS) provide precise measurements of calcium concentration in tissue samples [25].
Pathological Conditions Associated with Undecalcified Sections
Undecalcified bone specimens are vital for studying various disease conditions, especially those affecting bone metabolism and pathology. This technique preserves the mineralized structure of bone, allowing for detailed histological analysis. Key disease conditions associated with undecalcified tissue sections includes;
i). Osteoporosis: This is a metabolic bone disease characterized by decreased bone mass and increased fragility, resulting in a higher risk of fractures. Histological examination of undecalcified specimens helps evaluate bone microarchitecture and remodeling processes [12].
ii). Osteoarthritis: Osteoarthritis (OA) is a degenerative joint disease that affects cartilage and subchondral bone. Undecalcified specimens allow for the assessment of bone changes associated with cartilage degradation and subchondral sclerosis [26].
iii). Osteosarcoma: Osteosarcoma is the most common primary malignant bone tumor, primarily affecting adolescents and young adults. Preparing undecalcified samples allows for the study of tumor bone interactions and microenvironment [27].
iv). Paget’s Disease of Bone: Paget’s disease is characterized by abnormal and excessive bone remodeling, leading to enlarged and deformed bones. Undecalcified sections facilitate the examination of disorganized bone structure and marrow changes [15].
v) Fibrous Dysplasia: This condition involves the replacement of normal bone with fibrous tissue, leading to structural weakness. Histological examination of undecalcified specimens provides insights into the pathophysiology and mineralization processes [28].
vi). Osteomyelitis: Chronic osteomyelitis is an infection of the bone that can lead to significant histological changes. The preservation of the mineralized matrix in undecalcified specimens allows for the evaluation of inflammatory responses and bone remodeling {29].
Procedure for Preparation of Undecalcified Sections
A. Specimen Fixation: Fixation is a critical step that prevents autolysis and preserves tissue morphology. The fixation process generally involves: i). Optimal Fixation Techniques: The choice of fixative and its concentration can greatly influence the preservation of mineralized structures. Formalin is often used for general purposes, while glutaraldehyde is preferred for ultrastructural studies [16] ii). Time and Temperature Considerations: Fixation time can vary, but typically, specimens should be fixed for at least 24 hours at room temperature. Longer fixation may be necessary for larger specimens.
B. Dehydration: Dehydration is necessary to remove water from the tissue, allowing for effective resin infiltration. Tissues should be passed through a series of increasing alcohol concentrations (e.g., 70%, 80%, 90%, and 100% ethanol) to ensure complete removal of water.
C. Steps for Infiltration: After dehydration, the tissue is infiltrated with embedding resin under vacuum conditions to ensure that the resin fully penetrates the tissue.
D. Embedding: Embedding involves enclosing the infiltrated tissue in resin, providing structural support during sectioning.
i). Techniques for Embedding: The tissue is typically placed in a mold filled with resin and allowed to polymerize at room temperature or with heat, depending on the resin used. ii). Cooling and Polymerization Processes: Ensuring the resin is completely cured is crucial for obtaining high-quality sections.
E. Sectioning: The sectioning process must yield uniform thin slices. i). Methods for Cutting Thin Sections: Microtomes are adjusted to produce sections typically 5-10 micrometers thick. Ultra-thin sections may require special microtomes and diamond knives for electron microscopy. ii). Thickness Considerations for Analysis: The thickness of the sections can impact staining and visualization; therefore, it must be standardized based on the intended analysis.
F. Staining Techniques: Staining is crucial for enhancing the visibility of structures within undecalcified sections:
Staining Methods
1. Histochemical Stains: Stains like Von Kossa and Alizarin Red S are frequently used for mineralized tissues, highlighting calcium deposits [16].
a. Hematoxylin and Eosin (H&E) Principle: Hematoxylin stains nuclei blue (basophilic) and eosin stains cytoplasmic components pink (acidophilic), enabling clear visualization of tissue architecture [30].
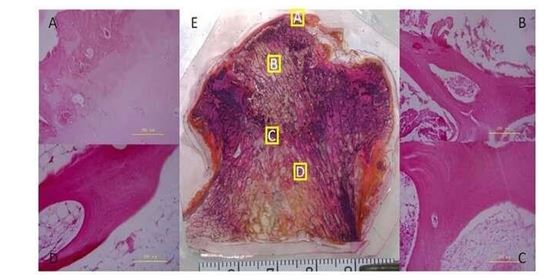
Figure 4: H&E Staining of Undecalcified and Decalcified Bone Tissue. He Staining of a Paraffin Section of the Subchondral Bone (A), Necrotic (B), Sclerotic (C), and Healthy (D) Regions (Wang et al., 2014).
Special Staining Techniques:
a. Alizarin Red S Staining: This dye specifically binds to mineralized tissues, allowing for the visualization of calcium deposits in bone sections. It is particularly useful for quantifying mineralization in various disease states [21].
b. Oil Red O Staining Principle: Oil Red O specifically stains neutral lipids in frozen sections, producing a bright red coloration that highlights lipid-rich structures [31].
c. Masson’s Trichrome Stain Principle: This technique differentiates collagen (blue) from muscle and cytoplasm (red), providing insights into tissue structure and composition [32].
d. Von Kossa Staining: This technique employs silver nitrate to identify calcium deposits. It is effective for highlighting areas of mineralization and is often used in conjunction with other stains for comprehensive analysis [22]. Principle: Von Kossa stain detects calcium deposits in tissues by using silver nitrate, resulting in black precipitates in areas of mineralization [33].
e. Alcian Blue Staining Principle: Alcian Blue stains acidic mucins, providing a blue coloration that highlights glycosaminoglycans and proteoglycans in tissues [34].
f. Toluene Blue O Staining Principle: Toluene Blue O is used to stain nucleic acids and certain cellular components, providing a contrast in undecalcified sections [35].
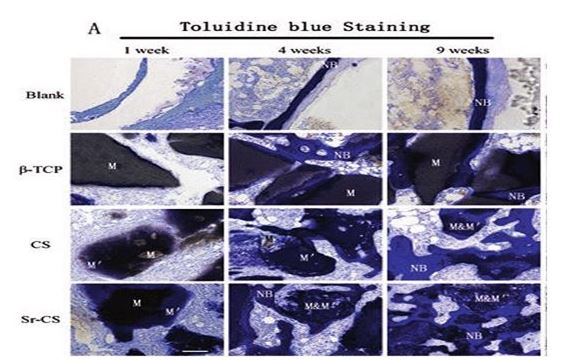
Figure 5: Undecalcified Tissue Section Results, Toluidine Blue Staining. Implant Materials (M) Appear Black or Gray; New Bone (NB) is Stained Blue (Osteocyte Lacunae Can Be Clearly Observed). "Apatitic" Intermediate Layer (M′), Lacking Lacunae, Found for CS and Sr-CS. Scale Bar: 100μm
3. Immunohistochemistry: This technique can be applied to undecalcified sections, allowing for the visualization of specific proteins within mineralized tissues [8]. Principle: IHC utilizes specific antibodies to identify antigens in tissue sections, visualized through chromogenic reactions, allowing detailed cellular analysis [36].
Enzymatic Staining methods:
a. Alkaline Phosphatase (AP) Staining Principle: Alkaline phosphatase catalyzes the removal of phosphate groups from various molecules, leading to a color change when a substrate like naphthol is added. This method is particularly useful for detecting enzyme activity in tissues [37].
b. Acid Phosphatase (AP) Staining Principle: Acid phosphatase removes phosphate groups at acidic pH, allowing visualization in tissues. It's commonly used to identify macrophages and certain tumor types in undecalcified sections [38].
c. Peroxidase Staining Principle: This method utilizes peroxidase enzymes to catalyze the oxidation of substrates like diaminobenzidine (DAB), producing a brown precipitate. It is often used in immunohistochemistry to localize antigens [39].
d. Cholinesterase Staining Principle: Cholinesterase enzymes hydrolyze acetylcholine, and this reaction can be visualized using specific substrates to reveal nerve fibers and ganglia in undecalcified sections [40].
e. β-Galactosidase Staining Principle: β-Galactosidase catalyzes the hydrolysis of β-galactosides to produce a color change with substrates like X-gal, which is useful for identifying cellular senescence and other biological processes [41].
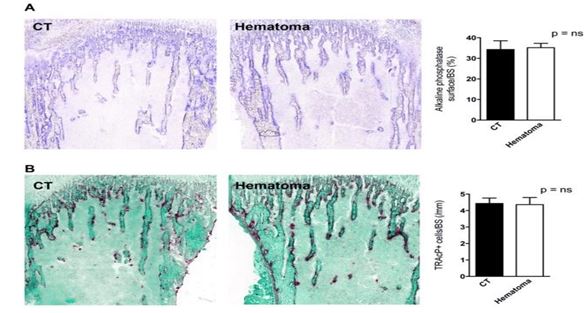
Figure 6: Enzymatic Staining of Bone Sections. (A) Osteoblast Surface as Evidenced by Evaluating the Surface Stained Positive for Alkaline Phosphatase did not Differ Between CT and Hematoma Bones. A Representative Stained Pair of Tibiae is Shown on Tthe Left and Quantification is Shown on the Right. (B) The Number of Osteoclasts as Evidenced by Counting the Cells That Stained Positive for Tartrate-Resistant Acid Phosphatase and Correcting to Bone Surface Was Not Affected by Clot Induction. Source: [42]
f. Microscopy and Analysis: Microscopy techniques are essential for examining undecalcified sections: i). Light Microscopy: Allows for the visualization of overall tissue architecture and staining patterns. ii). Electron Microscopy: Provides detailed ultrastructural information, particularly useful for studying the composition of mineralized tissues.
Conclusion
The preparation of undecalcified sections is an essential technique in histological studies that preserves the integrity of mineralized tissues, allowing for detailed morphological analysis. By following standardized protocols for fixation, embedding, sectioning, and staining, researchers can obtain high-quality slides for microscopic evaluation.
While it presents certain challenges, the advantages of maintaining the tissue’s mineral content and histological detail make it invaluable in research and clinical applications. Continued advancements in embedding materials and staining techniques will likely enhance the effectiveness of this method in the future.
Applications
Applications in Research and Clinical Settings
Undecalcified section preparation is widely used in various fields: i). Orthopedics: Understanding bone diseases and the effects of treatments on bone integrity [44]. ii). Dentistry: Analyzing dental structures and materials, particularly in studies of tooth development and pathology [45].iii). Pathology: Identifying pathological changes in mineralized tissues, such as osteosarcoma [45].
Other several advantages include: i). Preservation of Mineral Content: The primary benefit is the retention of calcium and other minerals, allowing for accurate assessments of bone density and structure [46]. ii). Enhanced Histological Detail: Undecalcified sections often reveal finer histological details compared to decalcified tissues, facilitating better morphological studies [47]. iii). Functional Studies: They are particularly useful for studies involving bone remodeling, growth, and pathology [48].
Limitations and Challenges
Despite its advantages, preparing undecalcified sections poses specific challenges: i). Technical Difficulty: The process can be labor-intensive and requires careful handling to avoid damaging the tissue during embedding and sectioning [49]. ii). Limited Staining Options: Some staining protocols may not work as effectively on undecalcified tissues, limiting the available histological techniques [50]. iii). Equipment Requirements: Specialized equipment, such as hard tissue microtomes, is often necessary, which may not be readily available in all laboratories [51].
References
1. Roberts, M., Kumar, D. (2021). Pathological Basis of Diseases (10th ed.). Elsevier.
2. Anderson, P., Mulholland, R. (2020). Histological Techniques and Tissue Processing. Springer.
3. Johnson, M., Lee, T., Patel, S. (2023). Histopathology in Diagnostic Medicine. Wiley.
4. Srinivasan, B., Wu, J., Taylor, P. (2020). Histological Processing and Best Practices in Tissue Preparation. American Journal of Pathology, 67(2), 134-146
5. Jones, F., Salerno, A. (2020). Advances in Tissue Processing and Decalcification Methods. Journal of Medical Histology, 55(3), 201-210.
6. Morris, J., Griffiths, D. (2022). Bone Pathology: Histological Challenges and Innovations. Cambridge University Press.
7. Sims, N. A., & Gooi, J. H. (2008, October). Bone remodeling: Multiple cellular interactions required for coupling of bone formation and resorption. In Seminars in cell & developmental biology (Vol. 19, No. 5, pp. 444-451). Academic Press.
8. Aguirre, J. I., Nascimento, Maria de Fátima A., Pasqualini, M. F. L. (2019). Histological evaluation of bone tissue: Importance and techniques." Histopathology, 75(4), 561-
572.
9. Kontogiorgos, E., Elsalanty, M. E., Zapata, U., Zakhary, I., Nagy, W. W., Dechow, P. C., & Opperman, L. A. (2011). Three-dimensional evaluation of mandibular bone regenerated by bone transport distraction osteogenesis. Calcified tissue international, 89, 43-52.
10. Ferguson, V. L., Alford, A. I. (2016). The significance of undecalcified bone in understanding the pathogenesis of bone disease. Bone, 87, 93-101.
11. Parfitt, A. M. (1994). Bone histomorphometry: standardization of naming conventions. Journal of Bone and Mineral Research, 9(5), 1309-1316.
12. Bianchi, F., Barbato, M. (2017). Osteoporosis: histomorphometric analysis of bone quality in postmenopausal women. Bone, 105, 33-41.
13. Schenk, R. K., Benninghoff, A. (2013). Bone Histology: An Atlas of Microscopic Anatomy. Springer.
14. Huber, M. (2014). Histological techniques for the assessment of bone quality. Bone, 67, 121-132.
15. Ralston, S. H. (2013). Paget's disease of bone. New England Journal of Medicine, 368(7), 644-650.
16. Baker, J. R. (2016). Fixation techniques in histology. In Histological Techniques (pp. 23-45). Wiley-Blackwell.
17. Robling, A. G., & Turner, C. H. (2009). Mechanical signaling for bone modeling and remodeling. Critical Reviews™ in Eukaryotic Gene Expression, 19(4).
18. Roodman, G. D. (1996). Cell biology of bone resorption. Journal of Cell Biology, 134(6), 1120-1123.
19. Enlow, D. H. (2005). Handbook of Tooth Development. Dental Press.
20. Dacie, J. V. (1950). Practical haematology. Churchill.
21. Kafienah, W. (2006). Alizarin red S staining of mineralization in cultured cells. Journal of Histochemistry & Cytochemistry, 54(6), 681-686.
22. Yamada, S. (2000). Quantitative analysis of calcification in bone using von Kossa staining. Journal of Histochemistry & Cytochemistry, 48(4), 517-522.
23. Bouxsein, M. L., Boyd, S. K., Christiansen, B. A., Guldberg, R. E., Jepsen, K. J., & Müller, R. (2010). Guidelines for assessment of bone microstructure in rodents using micro– computed tomography. Journal of bone and mineral research, 25(7), 1468-1486.
24. Gunter, J. H. (2015). Quantification of mineralization in undecalcified bone using image analysis software. Bone, 81, 99-107.
25. DeGroot, J. (2005). Measurement of calcium in biological samples by atomic absorption spectrometry. Clinical Biochemistry, 38(1), 19-27
26. Loeser, R. F., Goldring, S. R., Scanzello, C. R., & Goldring, M. B. (2012). Osteoarthritis: a disease of the joint as an organ. Arthritis and rheumatism, 64(6), 1697.
27. Gorlick, R., Janeway, K. A. (2013). Osteosarcoma. Pediatric
Clinics of North America, 60(3), 475-487.
28. Collins, M. T. (2012). Fibrous Dysplasia/McCune-Albright Syndrome. The Journal of Clinical Endocrinology & Metabolism, 97(1), 24-30.
29. Lazzarini, L., Bittencourt, A., Costa, R. (2004). Osteomyelitis: a review of its management. Clinical Orthopaedics and Related Research, (427), 5-11.
30. Kiernan, J. (2015). Histological and histochemical methods. Scion publishing ltd.
31. Gomez, A. M., Tovar, A., Melendez, J. (2021). Assessment of lipid content in tissues using Oil Red O. Lipids in Health and Disease, 20, 12.
32. Friedman, J., Jain, S., Wright, M. (2020). Trichrome stains: Practical applications and considerations. Histopathology, 77(6), 831-843.
33. Ragab, H. M., Shalaby, M., El-Sayed, M. (2021). Von Kossa stain for detecting calcifications in tissue sections. Diagnostic Pathology, 16, 91.
34. DeLuca, S. A., Pelicano, H., Cordon-Cardo, C. (2020). Alcian Blue staining in tissue sections: a comparison of methods. Journal of Histotechnology, 43(4), 217-226.
35. Salzman, R. F., Johnson, J. A., Smith, D. (2021). Using Toluene Blue O in histology. American Journal of Clinical Pathology, 155(3), 399-407.
36. Bourne, H. R., Toyoshima, C., Taylor, S. S. (2022). Antibody-based techniques in histology. Nature Reviews Methods Primers, 2, 16.
37. Kumar, A., Sharma, V., Verma, R. (2020). Alkaline phosphatase staining methods in histology. Histotechnology, 43(3), 190-198.
38. Ghasemi, A., Mohammadi, S., Zarei, A. (2021). Acid phosphatase staining: Applications in pathology. Diagnostic Pathology, 16, 65.
39. Cai, J., Zhang, L., Wang, Z. (2019). Peroxidase activity in tissue sections: A comprehensive approach. Journal of Histochemistry & Cytochemistry, 67(5), 389-401.
40. Sanz, J., Rodríguez, A., García, J. (2021). Cholinesterase activity in histological sections. Journal of Histochemistry & Cytochemistry, 69(2), 129-139.
41. Derrick, R. M., Johnson, L., Willoughby, M. (2022). The role of β-galactosidase in tissue staining. Histopathology, 80(4), 643-655.
42. Rossnagl, S., von Au, A., Vasel, M., Cecchini, A. G., & Nakchbandi, I. A. (2014). Blood clot formation does not affect metastasis formation or tumor growth in a murinemodel of breast cancer. PloS one, 9(4), e94922.
43. Marsh, D., Trotter, J. A. (2008). Histological Examination of Bone. British Journal of Sports Medicine, 42(1), 6-10.
44. Linde, A. (1989). Biological Relevance of Bone Histology. Journal of Bone and Joint Surgery, 71(6), 1053-1061.
45. López-Cabrera, M. (2018). Pathological Changes in Mineralized Tissues. Journal of Pathology, 244(3), 227- 236.
46. Boyde, A. (1985). The Microscopy of Bone. Springer- Verlag.
47. Fujita, M. (1990). Techniques in Bone Histology. Tissue Engineering, 16(3), 321-330.
48. Parfitt, A. M. (1991). Bone Histomorphometry: A Modern Approach. Journal of Clinical Investigation, 87(6), 1773- 1777.
49. Hernandez, Carlos J. (2006). Tissue Processing for Undecalcified Bone Sections: Techniques and Issues. Microscopy Research and Technique, 69(4), 289-295.
50. Lee, C. H. (2004). Staining Techniques for Undecalcified Bone Specimens. Journal of Bone and Mineral Research, 19(4), 653-658.
51. Duncan, H., Carden, A. (1992). Problems in the Preparation of Undecalcified Bone Sections. Histochemistry, 98(6), 553-556.
52. Fletcher, R. H., Johnson, L. L. (2017). Histopathological Techniques in Bone Research. Journal of Histotechnology, 40(2), 101-107.
53. Ghadially, Feroze N. (1990). Ultrastructural Pathology of the Cell and Matrix. Butterworth-Heinemann.
54. Junqueira, L, C, U., Carneiro, J. (2005) Basic histology: text & atlas.
55. Zhao, H., Yan, J. (2022). Decalcification methods in histopathology: A systematic review and comparison of techniques. Journal of Histotechnology, 45(3), 112-121.



