Research Article - (2025) Volume 8, Issue 4
A Physical Theory based on the Barycenter Frame of Reference III: Principles of Particle Statistics
Received Date: Oct 13, 2025 / Accepted Date: Nov 10, 2025 / Published Date: Nov 28, 2025
Copyright: ©2025 Zhong Cheng Liang. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Liang, Z. C. (2025). A Physical Theory based on the Barycenter Frame of Reference III: Principles of Particle Statistics. Adv Theo Comp Phy, 8(4), 01-20.
Abstract
In the barycenter frame of reference, the motion of a real (elastic) particle consists of three modes: translational, rotational, and vibrational. The three modes of energy of a thermodynamic system form a vector space, and the macroscopic state of the system is expressed by a vector in the energy space. The equilibrium state principle proposed in this article generalizes the classical laws of thermodynamics, thus bringing statistical physics into a unified axiomatic framework. The relationship between the order degree and the interaction is found by describing the microscopic states of the system through a vector of particle distribution. A complete set of the thermodynamic functions, the state equations, and the differential equations is derived with the help of particle statistics at two structural levels. The connection between the entropy and the association degree is established by defining the thermal entropy with the state function of thermal energy. The physical mechanism and properties of the phase transition are elucidated based on the structural analysis of the energy space. The results show that the real-particle statistical theory based on the barycenter reference frame reveals the universal laws of thermodynamic systems.
Keywords
Statistical Physics, Real-Particle Statistics, Barycenter Frame of Reference, Thermodynamic Relations, Phase Transition
Introduction
What are particles? Real particles in the physical sciences are small spatially localized objects with mass, volume and shape. They vary greatly in size or quantity, from subatomic particles like protons and electrons, to microscopic particles like atoms and molecules, and to macroscopic particles like powders and other granular materials [1]. In order to study the laws of motion of particles, classical mechanics reduced particles to point masses (point particles) without volume and shape [2]. To explain the aggregation phenomena, properties of interactions, such as electric charge, color charge, and spin, are assigned to point particles. In the Standard Model (SM) of particle physics, there are 61 kinds of elementary particles, which form composite particles through fundamental interactions (gravitational, electromagnetic, strong, and weak forces) [3]. However, the SM does not include gravitational interactions and cannot address the fundamental problems of condensed matter physics. There is no doubt that point particles are the approximation of real particles, and physical theories based on point particles are incomplete descriptions of the real world. In order to account for the spatial extension and size effects of particles, a paradigm shift in particle theory must be realized [4].
How do particles motion? Classical mechanics uses orbits to describe the movement of point particles. Knowing the force and initial conditions of the particles, the trajectory of the particles can be predicted according to Newton's laws [2]. In a tiny spatial range, it is impossible to predict the trajectory of particles because it is impossible to simultaneously determine their position and velocity. This limitation led to the birth of quantum mechanics. Quantum mechanics uses the state function to describe the motion of particles, and the motion of particles in tiny space is controlled by the Schrödinger equation. By determining the form of interaction, the probability of particle position or momentum occurrence can be predicted without knowing the initial position and velocity of the particle [5]. Classical mechanics and quantum mechanics describe particle motion in different ways, which leads to significant differences in concept and cognition. For example, quantum mechanics interprets the spatial extension of the state function as a property of the particle itself (wave-particle duality), which gives rise to the concept of non-localized particles such as the electron clouds and the quantum fields. In fact, both of classical and quantum particles describe the same entity, and the nonlocality of the quantum is only a reflection of the spatial extension of the particle trajectories [6,7].
Can the motion laws of macro and micro particles be unified? The real particle theory created by the author gives an affirmative answer [4,8,9]. Real (elastic) particles are objects with mass, volume and shape. From atoms to galaxies, they all share the common characteristics regardless of their scale. Unlike classical and quantum mechanics, real particle theory uses modes to describe motion. In a center-of-mass (barycenter) frame of reference, the motion of a real particle has three modes: translational, rotational, and vibrational. The translation is the displacement of the center of mass of the particle, the rotation is the spin of the particle, and the vibration is the change in the density of the particle. The orbital motion of point particles belongs to the translational mode of real particles and is suitable to be described by the orbital method; the motion of quantum belongs to the vibrational and rotational modes of real particles and is suitable to be described by the state function. The theory of real-particle flow fields shows that the action quantities of the flow fields include gradient, curl, and divergence, and the equation of the action field is a set of Poisson's equations about the action quantities [10-13]. The theory of real-particle dynamics shows that particles are driven by forces of gradient, curl, and divergence, and their motion follows the generalized Newton's second law, the modified energy theorem, and the angular momentum theorem, as well as the newly discovered curlity theorem [14,15]. The stationary state of the quantum corresponds to the equilibrium state of particle dynamics, and the quantization of energy is essentially a manifestation of the energy conservation theorem.
What are the statistics of particles? Starting from the common properties of real particles and based on the theory of particle flow-fields and real-particle dynamics, particle statistics uses statistical methods to calculate the average result of the motion of a large number of particles to infer the macroscopic properties of thermodynamic systems. Particle statistics is statistical physics or statistical mechanics based on the laws of motion of real particles [16,17]. In this paper, the basic principles of particle statistics are presented from the viewpoint of equilibrium of motion [18]. The particle statistical theory characterizes the macroscopic state of the system by the vector in energy space, describes the microscopic state of the system by the vector of particle distribution, presents the interaction of particles by the order degree of energies, and derives a complete set of thermodynamic functions and equations, thereby integrating statistical physics into a unified axiomatic theoretical system.
Fundamentals of Real-Particle Theory
Basic Postulates
The real particle theory takes five axioms as the starting point of logical deduction [4,10]. (1) Object: an object is a finite system composed of discrete real particles and has a hierarchical nested structure. (2) Real particles: real particles are objects with mass, volume, and shape. (3) Real space: real space is a three-dimensional place filled with moving particles. (4) Real time: real time is an irreversible process of object motion. (5) Motion: real particles move endlessly through interactions. Based on the above postulates, the author constructed the real particle theory, which contains the theories of particle flow-fields, particle dynamics, and particle statistics.
Object Structure
Objects consist of discrete real particles. There are only two primordial particles in nature, the proton and the electron, both of which have a certain mass and do not have to be broken down further. The mass of the proton is Mp = 1.6726216 × 10-27 kg and the mass of the electron is Me = 9.1093821 × 10-31kg. A proton and an electron make up a neutron, neutrons and protons make up atomic nuclei, nuclei and electrons make up atoms, atoms make up molecules, molecules make up supra-molecules, and so on to form composite particles (objects) with hierarchically nested structure.
The nested structure of an object can be expressed in terms of a family of particle sets as [10,18].
Topson ⊇ Midson ⊇ Bason ⊇ Hidson
Topson, midson, bason, and hidson are coined to name the particles located at the top level, middle level, basic level, and hidden level, respectively. An upper-level particle contains the lower-level particles, and the lower-level particles are the subsets of the upper-level particle. To study the nature of water, for example, consider the water system as a topson and the water molecules as the basons, then the midsons are the clusters of water molecules (water clusters) and the hidsons are the hydrogen and oxygen atoms that make up the water molecule. To study the structure of a galaxy, consider the galaxy as a topson and the stars as the basons, then the midsons are the clusters of stars and the hidsons are all celestial bodies within the stars.
Object Motion
The spatial state of an object consists of position, profile, and posture. Position is represented by the position vector of the object's center of mass (barycenter), profile by the eigenvalues of the inertia matrix (principal inertias), and posture by the eigenvectors of the inertia
matrix (inertia principal axes). Using the barycenter as a reference, the object's motion consists of three modes: translational, vibrational, and rotational, which correspond to the temporal changes in the object's position, profile, and posture, respectively [10,15].
At time t, let Oc be the barycenter of the object, (I1, I2 , I3) be the principal inertias, and (e1 , e2 , e3) be the inertia principal axes, from which we can establish the barycenter frame of reference [Oc e1 e2 e3]. After unit time ts, the barycenter of the object moves to Oc', the principal inertias changes to (I1' , I2', I3' ), and the principal axes changes to (e1' , e2' , e3' ), then the barycenter frame of reference at the moment of t' = t + ts becomes [O'c e'1 e'2 e'3 ],′ - If the position of Oc ' in [e1 , e2 , e3] is ξ = (ξ1 , ξ2 , ξ 3), then the translational velocity of the barycenter is

object's barycenter, rotation is the fixed-point rotation of the object around its barycenter, and vibration is the radial oscillation of the particles inside the object with respect to the barycenter. Each mode has three degrees of freedom, a total of nine degrees of freedom of motion for an object.
Measurement Principle
The principle of measurement is expressed as follows: The measurement of any physical quantity has precision limitation, and the limit precision is constrained by physical relationships.
The limit precision of a physical measurement is called the scale, and the physical quantity expressed in terms of the scale is called the

The measurement principle is subversive in that it reveals the nature of quantum, circumvents the infinity of mathematics, and opens the door to nonlinear calculus. The measurement principle has been referred to as the principle of measurement relativity and the principle of objectivity [8,9,19].
Principles of Particle Statistics
State of Particle Systems
Consider a real particle system with a volume of V containing N basons. Labeling the bason's mass Mi, the principal inertia Iiα, the compression modulus Yi , and the volume Vi , we can express the bason's translation energy Ki , rotation energy Li , and vibration energy Hi as

The statistical quantities {K,L,H,E} are always greater than zero and represent the macroscopic state of the particle system.
Definition of Energy Space

Define the derived energy according to cyclic symmetry: entire energy Ez (front-energy plus back-energy), potential energy Jz (back- energy minus main-energy), thermal energy Qz (main-energy plus front-energy), and chemical energy Gz (front-energy minus back- energy).
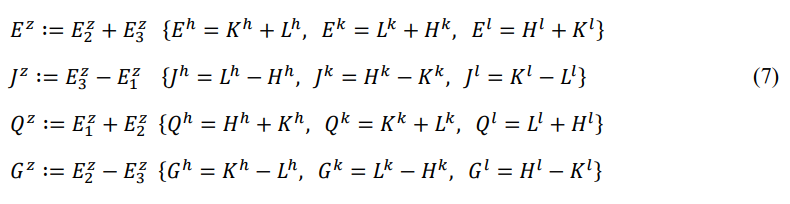
Define the order degree: front-order degree az (front-energy divided by main-energy), and back-order degree bz (back-energy divided by main-energy).
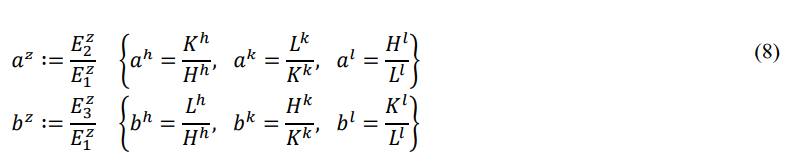
The front and back energies defined in the order of “HKLHKL” are called the right cycle, and the front and back energies defined in the order of “HLKHLK” are called the left cycle. This article uses the right cycle, while the previous papers of the author used the left cycle [15]. In both cycles, the entire energy is equal, the potential energy is always negative, the thermal energy is always positive, and the chemical energy is equal in value and opposite in sign. The calculation method for the two cycles is the same, the distinction is that the front and back energies represent different modes of motion.
Geometry of Energy Space
The structure of energy space can be represented by a Cartesian coordinate system containing {H, K, L} axes. For easy of illustration, we use the set symbol E[*] to denote the three-dimensional space, F[*] denote the two-dimensional planes, S[*] denote the two-dimensional curved surfaces, L[*] denote the one-dimensional lines, P[*] denote the zero-dimensional points, and the symbol “*” denote the condition or sign of the set. As shown in Figure 1, the three coordinate axes of the energy space are {H, K, L} and the three coordinate planes are {F [H = 0], F [K = 0], F[L = 0]}. The energy space is confined to the first octant (+,+,+) since the motion energy is always positive.

The six phases have six interfaces, which are divided into two classes of J-type and G-type. The J-type interfaces are the planes between
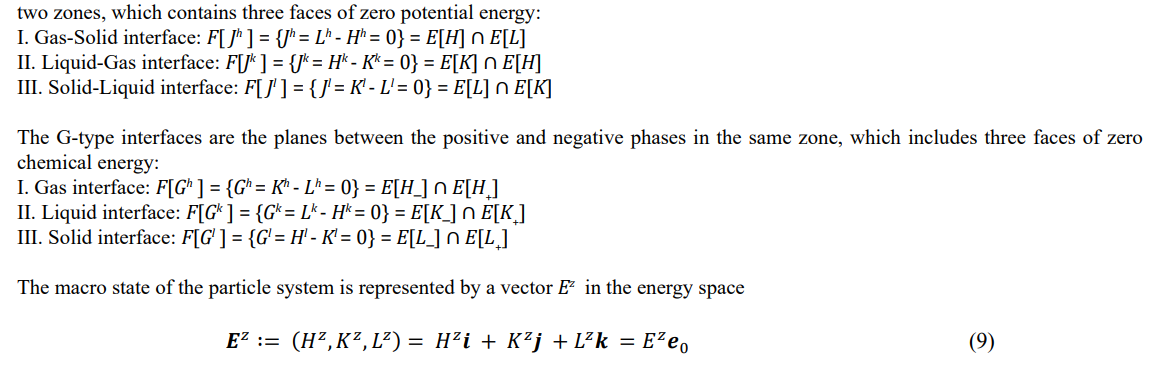
E z is called the energy vector, where (i , j , k) are the coordinate basis vectors, e0 is the unit vector, and E z is the full energy. In energy space, the spherical surface of equal full-energy is known as the isoenergetic surface and is denoted by S[Ez ] = {S[Eh ], S[Ek ], S[El ]}.
Note that Ez = (Hz, Kz, Lz ) expresses a vector in Cartesian space, with the three components fixedly combined with the basis vectors (i, j, k). The ordered triplet {H, K, L} does not represent the energy vector but indicates the vibration zone {Hh, Kh, Lh }. To express the translational and rotational zones, the triplet elements must be rearranged by even permutations.
Principle of Equilibrium State
The equilibrium state principle is the fundamental principle of particle statistics, which generalizes and extends the laws of classical thermodynamics. The principle of equilibrium state is formulated as follows:
There are equilibrium states in an isolated system, and the length and direction of the energy vector in the equilibrium state remain stable. The relaxation process of the system from non-equilibrium to equilibrium is irreversible.
The principle contains following aspects:
• Definitions of the Equilibrium State and the Relaxation Process: A system in which the length and direction of the energy vector remain constant for a long time is called an equilibrium state, otherwise it is non-equilibrium. The process by which a particle system moves from a non-equilibrium state to an equilibrium state is called the relaxation process, and the time it takes is called the relaxation time. Long time means much longer than the relaxation time.
• Existence Hypothesis of Equilibrium States: There exist equilibrium states for isolated systems. The length of the energy vector of the isolated system is unchanged, and the direction of the energy vector will inevitably stabilize through the relaxation process. A equilibrium system can be divided into subsystems, each with the same direction of the energy vector. This is a fact stated by the zero-th law of thermodynamics.
• Energy Conservation and Transformation Theorem: The length of the energy vector of an isolated system is constant and the full energy is conserved. The energy vector changes direction during relaxation, which means that the motion energy is transformed between three modes. This is the implication of the first law of thermodynamics.
• Irreversibility Theorem: The relaxation process of the isolated system is irreversible. The energy vector of an isolated system will not lose its stability after its direction is stable, otherwise it will violate the definition of equilibrium states. This is the content of the second law of thermodynamics.
• Fluctuation Theorem: The length and direction of the energy vector of the equilibrium system fluctuate instantaneously as a result of surrounding influences. Instantaneous means much less than the relaxation time, and fluctuation means the change in the energy vector is within the allowable range of the energy scale.
Equations of Equilibrium State
Both the motional energy and the derived energy of the equilibrium state are state functions of the system. By equating the full energy and the entire energy

Each equilibrium surface contains three regions, where {S[H1 ], S[K1 ] , S[L1 ]} are stable equilibrium regions and the rest are sub-stable equilibrium regions.
The stable regions represent normal states. {S[H1 ], S[K1 ], S[L1 ]} show three basic states of matter: gas, liquid, and solid. The sub-stable regions represent super-normal states. The sub-stable regions include the super-vibration surface S[H * ] = S[L2 ] U[K3 ] located in the gas zone, the super-translation surface S[K * ] = S[H2 ] US[L3 ] located in the liquid zone, and the super-rotation surface S[L * ] = S[K2 ] U S[H3 ] located in the solid zone.
Scales of Thermodynamic System
Specify the energy scale as the average of the main energy
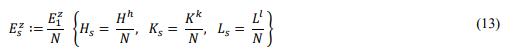

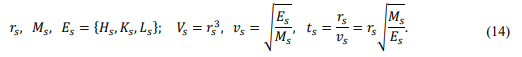
Vs ,vs , ts are the volume scale, velocity scale, and time scale, respectively. This theory adopts an all-variable scale system. Since no specific scale base value is prescribed, the conclusions apply to any thermodynamic system under extreme conditions.
In order to standardize the energy units of different zones, the energy scale is expressed in the following form

Where {D, T, B} are the motion strengths, and {h, k, l} are coefficients for unit transformation. In the SIU system, the energy unit is joule (J). If the vibration strength uses the divergence D (in Hertz, Hz), the coefficient h = 6.6260693 × 10-34 J/Hz is the Planck constant [10]. The translation strength uses the thermodynamic temperature T (in Kelvin, K), and the coefficient k = 1.3806506 × 10-23 J/K is the Boltzmann constant. The rotation strength uses the magnetic induction strength B (in Tesla, T), then, the coefficient l = 9.274 ×10-24 J/T is the constant of Bohr magneton. Since the motion energies {H, K, L} are positive definite, the following conclusions are obviously
![]()
It shows that the motion strength cannot be zero, where T > 0 is a fact stated by the third law of thermodynamics (absolute temperature zero is not reachable).
According to the general quantization rules of particle dynamics, the digits of the motion energy in terms of the scale Esz take only non- negative integers, i.e [14].

It can be inferred that the digits of the derived energies are all integers, where the entire (full) and thermal energies are constantly positive, the potential energy is constantly negative, and the chemical energy can be positive, negative, or zero. The order degrees are constantly positive and are rational numbers. The quantization condition indicates that the states on the equilibrium surface are not continuously but discretely distributed.
Interaction of Particles
The current dominant theory in statistical physics is the Gibbs ensemble theory [16,17]. Gibbs' theory gives the microcanonical, canonical, and grand canonical distributions of energy in the Γ-space for the isolated, closed, and open systems, respectively. For quantum systems, Gibbs statistics requires transition to quantum distributions (Fermi-Dirac and Bose-Einstein distributions). The difficulty of Gibbs statistics lies in the integration of the potential energy function in the Γ-space. Potential energy represents the interaction of particles, and due to the lack of a unified form of interaction, it is impossible to draw universal conclusions. Real particle theory has constructed the energy space, determined the equilibrium surface and its quantum properties, and the introduction of particle interactions is the next step in the completion of particle statistics.
Interaction is the relationship of mutual influence and restraint between particles, which is the fundamental cause of object motion and state changes. Interaction can be expressed in different forms. Classical physics expresses interaction in terms of potential energy and force, while modern physics expresses interactions in the quantity of action. Particle statistics studies the general laws of systems consisting of a large number of particles. It avoids dealing with specific forms of potential energy or action quantity and understands interactions in terms of the motion of particles [4]. For example, the form of potential energy cannot be given for complex systems such as ionic liquids, but general transport properties of ions can be extracted by analyzing the processes of association and dissociation [20]. Molecular dynamics simulations have shown that the structural features of ionic liquids can also be inferred from the correlation coefficients of the motion of particles [21,22].
Particle statistics treats association and dissociation as the form of particle interaction and connects them to the order degree of the particle energy. On the one hand, particle association characterizes attraction and dissociation characterizes repulsion. The process of particle association-dissociation reflects the dynamic nature of chemical reactions, and the equilibrium of association-dissociation is closely related to the change in the state of matter. On the other hand, the order degree is a measure of the correlation of motion modes. According to the idea that motion and action are mutually causal, the order degree certainly characterizes the interaction of particles. The order degree consists of the front-order degree a and the back-order degree b. The equilibrium equation ab = 1⁄2 shows that they are inversely proportional to each other and represents opposite effects of attraction and repulsion. Since there are two prescriptions for the front and back energies, the front/back may correspond to either association/dissociation or dissociation/association. The process of association or dissociation should be analyzed according to the motion modes represented by the front and back energies.
Statistics of Midsons
The theory of particle statistics describes the microscopic states of a system in terms of the distribution of midsons. In the nested model of objects, basons have stable structure and a definite number, while midsons are clusters composed of a few basons with a dynamic structure and indefinite number. The association of basons forms midsons, and the dissociation of midsons returns to the basons. The number of midsons is the result of the competitive balance between association and dissociation. The relationship between the micro- state and macro-state can be finally established through the midson statistics.
According to the measurement principle, the volume of the particle system can be expressed as V=![]() Vs, and the unit volume Vs = (rs)3 is called the space cell. If a space cell contains n basons, it forms an n-midson, denoted by C(n). Since each midson occupies the volume Vs, the total number of midsons is C =
Vs, and the unit volume Vs = (rs)3 is called the space cell. If a space cell contains n basons, it forms an n-midson, denoted by C(n). Since each midson occupies the volume Vs, the total number of midsons is C = ![]() Intercept the system's configuration at time t = τ . ts and count the number of C(n) in the configuration, denoted as Cn. The N-dimensional vector Cτ = (C1, C2 , C3 , ..., CN ) is known as the distribution vector of midsons. Figure 3 illustrates an instantaneous configuration of a system where the number of basons N = 14, the number of midsons V =
Intercept the system's configuration at time t = τ . ts and count the number of C(n) in the configuration, denoted as Cn. The N-dimensional vector Cτ = (C1, C2 , C3 , ..., CN ) is known as the distribution vector of midsons. Figure 3 illustrates an instantaneous configuration of a system where the number of basons N = 14, the number of midsons V = ![]() = 9, and the midson's distribution vector Cτ = (5,3,1,0,0,0,0,0,0,0,0,0,0,0).
= 9, and the midson's distribution vector Cτ = (5,3,1,0,0,0,0,0,0,0,0,0,0,0).
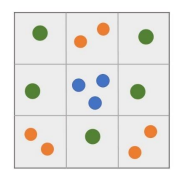
Figure 3: Two-Dimensional Schematic Diagram of Midson Distribution
The vector of midson distribution characterizes the micro state of the system. The distribution vector has the following properties:
• The sum of the vectoral components is equal to the total midson's number of the system

• The vectoral components are subject to the constraint of the conservation of bason's number

• The distribution vector determines two probability functions

ρn is the occurrence probability of the n-midson, and bn is the dissociation degree of the n-midson
• The probability distribution functions have the following properties

Summing over ρn gives the probability normalization, and summing over bn gets the dissociation degree b of the system. The larger b is, the closer the number of midsons is to the number of basons. The b = 1 indicaties that all midsons are dissociated, and a = 1 indicates that all basons are associated. This paper specifies the back-order degree as the degree of dissociation and the front-order degree as the degree of association, or vice versa if necessary.
The midson's vibrational, translational, rotational, entire, potential, thermal, and chemical energies are labeled with the Greek letters ηn,

Fundamentals of Thermodynamics
Equations of State
Pressure
Denote the volume by V z and define the motion pressure as the density of the motion energy

Their specific forms are
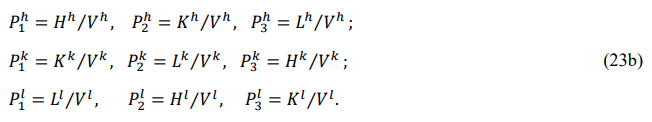
p1z,p2z,p3z are called the main, front, and back pressures, respectively. The full pressure of the system is defined as

The full pressure subjects to the equilibrium constraint

The full pressure can also be calculated using the definition of entire energy according to the following equation
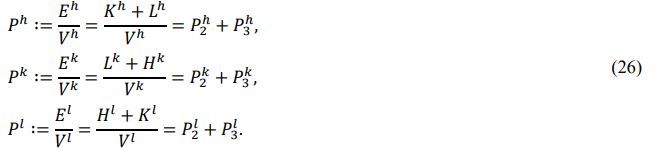
Therefore, the full pressure is also equal to the sum of the front and back pressures. Note that the pressure defined by particle statistics is different from that defined in textbooks.
State Equations
Express the main energy in terms of the energy scale as
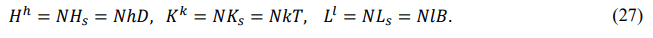
Therefore
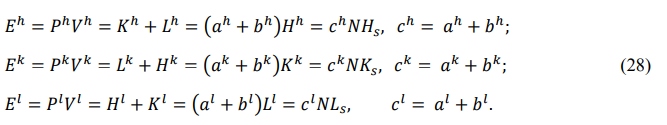
They are a set of state equations applied to the vibration surface S[H], the translation surface S[K], and the rotation surface S[L], respectively. The cz = az + bz are referred to as the mode coefficients in the equations.
Equipartition Theorem
According to the definitions of the back-order degree and the potential energy, there are

On the J-type interface, the potential energy is J z = 0, so the back-order degree is bz = 1 the front-order degree is az = 1⁄ (2bz ) = 1⁄2, and the mode coefficient is cz = 3⁄2. Therefore, the equations of state become
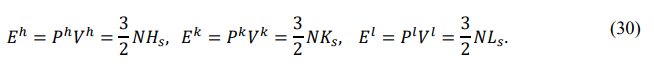
A potential energy of zero indicates that the midsons are all dissociated (bz = 1) and the basons are statistically independent (az = 1⁄2). The average energy of each bason is then (3 ⁄ 2) Esz, and the energy of each degree of freedom is (1⁄2) Esz. This is known as the theorem of energy equipartition for independent particle systems.
Functions of State
Energy, volume, pressure, and motion strength are all state functions of an equilibrium system. Since the ordered triplet{H,K,L} has cyclic symmetry, we analyze the state functions in the translation zone as the example, and the calculations in vibration and rotation zones can be carried out with reference. For simplicity, we omit the index k in the translation zone.
Motion Energy
The main energy in the translation zone is the translation energy K, the front energy is the rotation energy L, and the back energy is the vibration energy H. The energy scale is Ks = kT, and the order degree and the equilibrium conditions are

The motion energy is decomposed according to the digital factors as follows
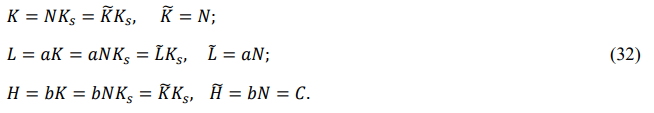
The motion energy can be decomposed in terms of the number of midsons
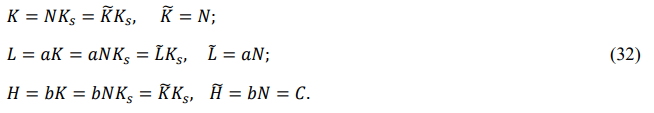
They can also be factorized by the volume V = CVs as
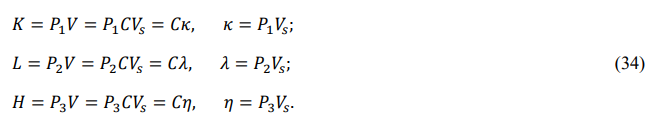
Derived Energy
The derived energy can be decomposed by digital factor as
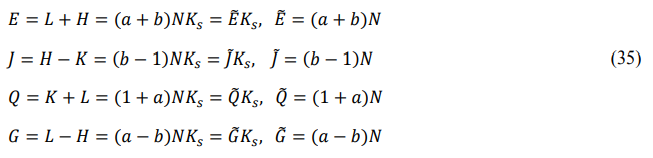
They can also be decomposed by the midson number as
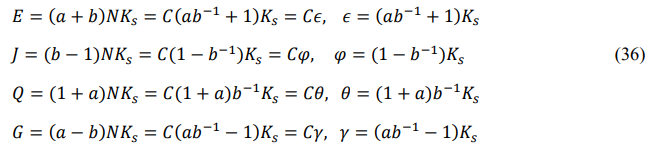
The γ is the midson average of chemical energy and is known as chemical potential.
Thermal Entropy
Entropy is an important concept connecting thermodynamics and statistical mechanics. In thermodynamics, the heat Q* is a process quantity. Through the analysis of the reversible Carnot cycle, Clausius discovered the entropy function S that describes the macro state of the system (dS = δQ*⁄T). In statistical mechanics, it is the state number Ω that describes the micro state, and it is the Boltzmann formula S = k. ln Ω that links the macro and micro parameters. In particle statistics, entropy can be defined directly by the thermal energy, and the parameter that links entropy to the micro state is the degree of association.
Particle statistics defines the ratio of the thermal energy to the temperature as the thermal entropy, i.e.

The midson average of the thermal entropy is
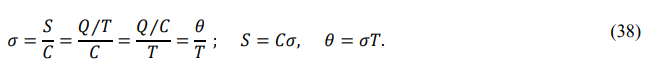
Equation (37) shows that the thermal entropy is proportional to the front-order degree (association degree) a. In translation zone, a = L ⁄ K indicates that the greater the thermal entropy, the greater the ratio of the rotation energy to the translation energy. From the viewpoint of the heat engine energy conversion, the larger the thermal entropy, the smaller the translational energy (or the larger the rotational energy) of the working fluid, and the weaker the ability to do external work. This result is consistent with the conclusion of thermodynamics. However, there is a clear difference between the thermal entropy and the classical entropy. Statistical mechanics interprets classical entropy as the degree of disorder; the greater the entropy, the more disordered the system. Particle statistics indicates that thermal entropy represents the association degree and reflects the particle interaction. The greater the thermal entropy, the higher the degree of association of basons and the lower the degree of dissociation of midsons.
Internal and Enthalpic Energies
The other two derived functions, internal energy U and enthalpic energy Y, are defined as follows
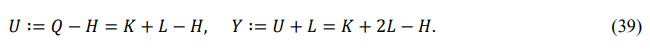
Their digital factors are
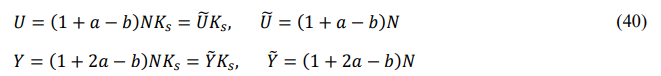
Their midson averages are
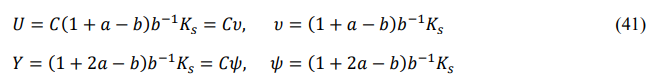
The energy variation in a cycle of heat engine can be explained by using the internal energy function. The amount of change in internal energy of the working fluid after a cycle is ![]() U =
U = ![]() Q -
Q - ![]() H, which is equal to the heat absorbed
H, which is equal to the heat absorbed ![]() Q minus the change in the mechanical energy
Q minus the change in the mechanical energy ![]() H. The fluid potential energy remains constant
H. The fluid potential energy remains constant ![]() J = 0 after the cycle, and the change in mechanical energy
J = 0 after the cycle, and the change in mechanical energy ![]() H =
H = ![]() K +
K + ![]() J =
J = ![]() K equals the work done by the system to the outside. The cycle efficiency of the heat engine is
K equals the work done by the system to the outside. The cycle efficiency of the heat engine is ![]() K /
K / ![]() Q = (
Q = (![]() Q −
Q − ![]() L)/
L)/![]() Q = 1−
Q = 1− ![]() L /
L / ![]() Q. After cycling, the molecular association increases and the rotational energy of the working fluid increases (
Q. After cycling, the molecular association increases and the rotational energy of the working fluid increases (![]() L > 0), so the efficiency of heat engine is less than one.
L > 0), so the efficiency of heat engine is less than one.
Response Functions
The equation of state in translation zone is
![]()
The equation of state gives the relationship between {P,V,T,c}, from which we can calculate the response functions of thermodynamics.
• Isobaric volume response RP: The rate of volume change caused by temperature change while maintaining constant full-pressure.

• Isovolumetric pressure response RV: the rate of full-pressure change caused by temperature change while maintaining constant volume.

• Isothermal volume response RT: the rate of volume change caused by full-pressure change while maintaining constant temperature.

• Adiabatic volume response RE: the rate of volume change caused by full-pressure change while maintaining constant full-energy.

Response functions reflect the property of continuous change in the order degree. A jump in the order degree will cause RP , RV , RT to diverge.
Total Differential of Energy
According to equations (34) and (38), the rotational and thermal energies of a midson are λ = P2 Vs and θ = σT, respectively. Since κ = θ - λ, we express the differential of the translation energy as
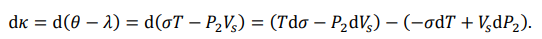
Also, since κ = υ - γ and dκ = dυ - dγ, we obtain the differentials of the internal energy and chemical energy
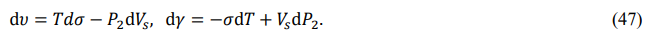
Therefore, the differential of the chemical energy of the system can be calculated by
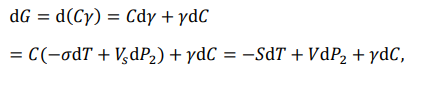
Briefly

This is the total (complete) differential equation for chemical energy, which is called the basic differential equation.
Subtracting the basic differential by dG = d(Cγ) = Cdγ + γdC yields an identical formula
From the basic differential, other total differentials of energies can be obtained by Legendre transformations. For example, substituting SdT = dQ - TdS into the basic differential yields the differential equation for enthalpy dY = TdS + VdP2+ γdC. Table 1 lists the various energy relations and full differentials for the translation zone, which are equivalent to each other.
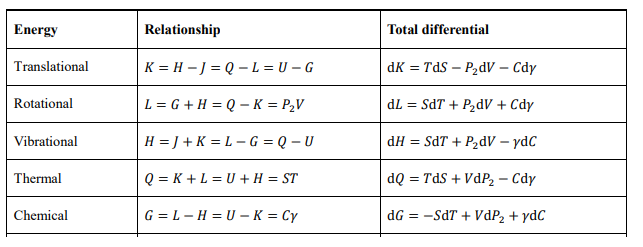
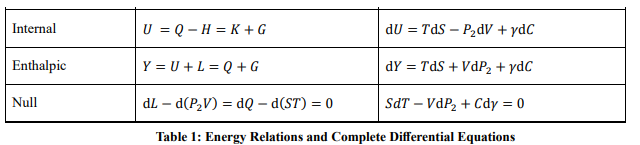
The above conclusions apply to closed systems. The closed system has a definite bason number N, and the variation of the midson number C reflects physicochemical processes. For open systems, the midson number can be regarded as the mole number of fixed components ni, and if the chemical potential per mole is γi, then G = ∑ ni γi . At this point the basic differential equation of the open system can be expressed in the common form

Comparison with the classical thermodynamics shows that in this paper G is the Gibbs free energy, H the mechanical (vibrational) energy, -H the Helmholtz free energy, -L the Landau free energy, and the identity SdT - VdP2 + Cdγ = 0 is comparable to the Gibbs-Duhem equation [16]. A significant difference is that the front pressure P2 replaces the full pressure in the differential equations. Moreover, the conclusions of this section apply only to the translation zone. The cases in the rotational and vibrational zones require transformations according to the cyclic symmetry.
Relations of Partial Derivatives
Partial derivative relations between state functions are important tool in the study of thermodynamic processes. According to the sufficient conditions for complete differentiation, each total differential equation corresponds to a set of partial differential relations called Maxwell relations [17]. For example, the total differential of the thermal energy is

The corresponding Maxwell relations are
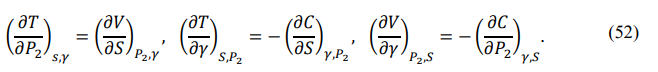
If {P,V,T} are used as independent parameters, the full differentials of {S,P2,γ} are given by
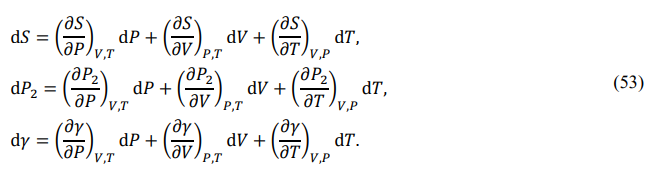
Substituting into Eq. (51) and rearranging yields the following set of partial differential relations
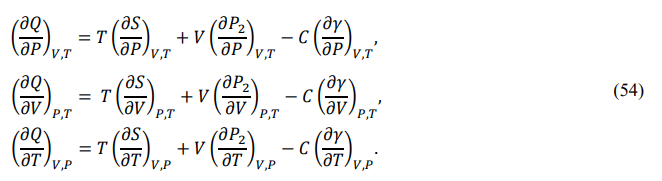
Fundamentals of Phase Transition
Classification of Phase Transition
Phase transitions usually refer to physical processes in which a medium changes between three basic states: gas, liquid, and solid. Particle statistics systematically describes phase transition phenomena in terms of energy space. As described in Section 3, the energy of the particle system is located in the first actant of the Cartesian coordinate system with {H,K,L} as axes. The energy space is divided into gas zone E[H], liquid zone E[K], and solid zone E[L] by three J-type interfaces, and each zone is divided into positive and negative phases by a G-type interface, making a total of six phases in the energy space. The energy space has three equilibrium surfaces, each with one stable region and two sub-stable regions. The stable region represents normal states and the sub-stable region represents super- normal states.
A phase transition occurs when the state of the system changes from one stable region to another. This phase transition occurs at a J-type interface and is therefore called a J-type phase transition.
A phase transition occurs when the state of the system changes between the positive and negative phases of a stable region. The state before and after this phase transition crosses the G-type interface, so it is called a G-type phase transition.
A system may move from a stable region on an equilibrium surface to a sub-stable region, i.e., from the normal state to the super-normal state. The phenomenon of changing the state of matter of a super-normal system is called a non-equilibrium phase transition.
J-Type Phase Transition
Transition Lines
A straight line where two equilibrium surfaces intersect is called a phase transition line, and the J-type phase transition occurs on the transition line. There are three phase transition lines, each with two names according to the direction of the phase transition process. Right cycle: gas-liquid line L[HK] = S[H] ∩ S[K], liquid-solid line L[KL] = S[K] ∩ S[L] and solid-gas line L[LH] = S[L] ∩ S[H]. Left cycle: gas-solid line L[HL], solid-liquid line L[LK], and liquid-gas line L[KH]. The gas-liquid (liquid-gas) line is included in the gas-liquid interface, the liquid-solid (solid-liquid) line is included in the liquid-solid interface, and the solid-gas (gas-solid) line is included in the solid-gas interface.
The J-type phase transition encompasses the six basic phase transitions of nature, and their names and basic characteristics are summarized in Table 2.
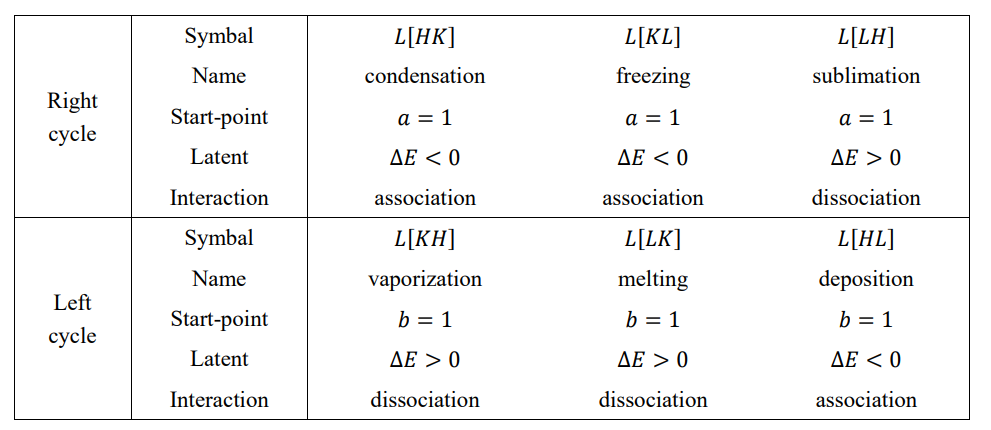
Table 2: Basic Features of J-Type Phase Transition
Transition Mechanism
The process of J-type phase transition is described as follows: the system starts from point P[A] after arriving at the transition line, and then moves continuously along the line until it reaches point P[B] to complete the phase transition. P[A] is the starting point of the phase transition, P[B] is the ending point of the phase transition, and there are states of two-phase coexistence between P[A] and P[B]. The energy vectors at P[A] and P[B] are EA and EB , respectively
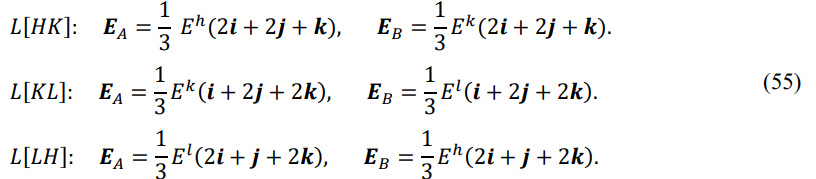
From the starting point to the ending point, the length of the energy vector changes while the direction remains constant. The amount of change in the length ![]() E = EB - EA is called the latent energy of the phase transition.
E = EB - EA is called the latent energy of the phase transition. ![]() E > 0 indicates a process of absorbing energy for particle dissociation;
E > 0 indicates a process of absorbing energy for particle dissociation; ![]() E < 0 indicates a process of releasing energy for particle association. The J-type transition is equivalent to the first-order phase transition in condensed matter theory [16].
E < 0 indicates a process of releasing energy for particle association. The J-type transition is equivalent to the first-order phase transition in condensed matter theory [16].
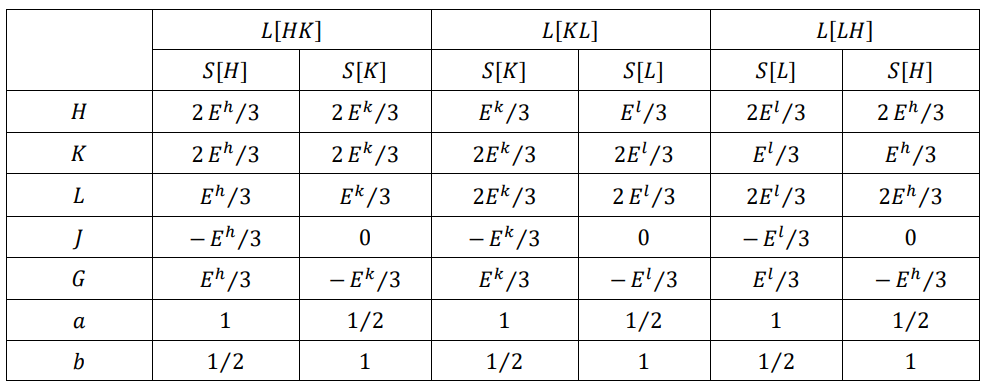
Table 3: The Energy and the Order Degree in the Phase Transition Line
Table 3 lists the motional energy, potential energy, chemical energy, and order degree at the starting and ending points of the phase transition. As can be seen from the table, the starting point of the right cycle transition is at a = 1, with a negative potential energy and a positive chemical energy; the starting point of the left cycle transition is at b = 1, with a zero potential energy and a negative chemical energy. The change in sign of the chemical energy is a common property of all phase transitions. The J-type phase transition is characterized by the finite amount of latent energy and the 1/2 leap or drop in the order degree.
Liquid-Gas Transition
Knowing the phase transition temperature T0 , the molar latent energy ![]() E, and the molar volumes V0 k and V0 h , the phase transition parameters can be calculated according to the relationships listed in Table 4.
E, and the molar volumes V0 k and V0 h , the phase transition parameters can be calculated according to the relationships listed in Table 4.
|
|
Liquid zone |
Gas zone |
|
Energy scale |
Ks = kT0 = Kk ⁄ N0 |
Hs = hD = Hh ⁄ N0 |
|
Molar volumes |
V0k |
V0h |
|
Translation energy |
Kk = 2Ek⁄3 |
Kh = 2Eh⁄3 |
|
Vibration energy |
Hk = 2Ek⁄3 |
Hh = 2Eh⁄3 |
|
Rotation energy |
Lk = Ek⁄3 |
Lh = Eh⁄3 |
|
Full pressure |
Pk = Ek⁄V0k |
Ph = Eh⁄V0h |
Table 4: Parameters and Relationships of Liquid-Gas Phase Transition
Taking the vaporization of water as an example, the liquid-gas transition temperature of water at standard atmospheric pressure is T0 = 373.15K. It is known that the latent energy (heat of vaporization) per mole ![]() E = Eh - Ek=40626 J, the molar volume of liquid water V0k = 18.612 × 10-6m3, the molar volume of water vapor V0h = 30114 × 10-6m3, and Avogadro constant N0 = 6.02214179 × 1023 mol-1. Thus, we yield following results
E = Eh - Ek=40626 J, the molar volume of liquid water V0k = 18.612 × 10-6m3, the molar volume of water vapor V0h = 30114 × 10-6m3, and Avogadro constant N0 = 6.02214179 × 1023 mol-1. Thus, we yield following results
G-Type Phase Transition
Symmetric State
The energy vector on the G-type interface F[G z] can be expressed as
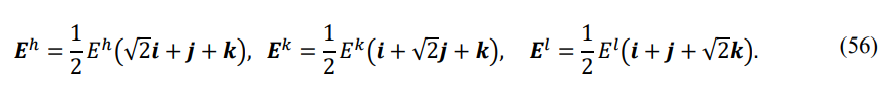
Stable symmetry states are very sparse compared to excited states. Table 5 shows the five pairs of stable symmetry states with minimum full energy. It can be seen that the order degrees of the same pair of symmetry states are unequal, with differences of less than 1/2. The differences in the front-order degrees of the five pairs are 1⁄12,1⁄12,7⁄30,1⁄12,7/40, respectively, with the third pair having the largest difference in order degrees.
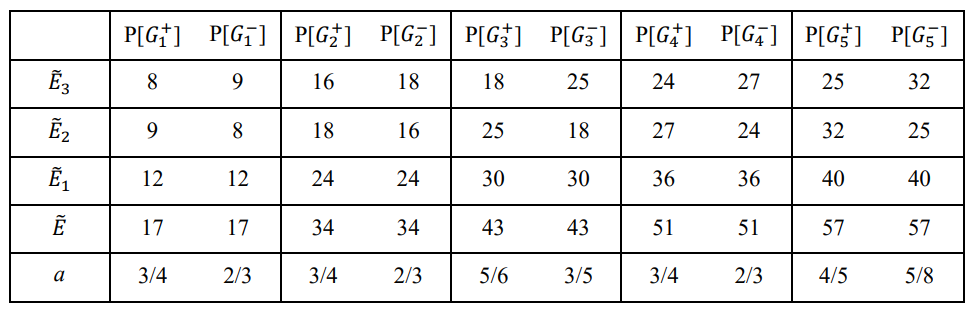
Table 5: Five Pairs of Symmetric States with Minimum Full Energy
Transition Mechanism
A G-type phase transition is a change in the state between P[Gi + ]and P[Gi - ]. Because they occur in the same zone, G-type transitions have three subclasses: gas-zone transition, liquid-zone transition, and solid-zone transition. Since there is no change in the state of matter, the physical mechanism for this type of transition is a change in the structure of midson. If G-type transitions are referred to as structural phase transitions, J-type transitions can be referred to as non-structural phase transitions. G-type phase transitions can also be categorized into symmetric and asymmetric phase transitions. Symmetric transitions occur when the system switches between the same pair of symmetric states P[Gi - ] and P[Gi + ] During symmetric transitions the direction of the energy vector changes, the length remains constant, and no energy is exchanged with the surroundings. Symmetric transitions include second- order phase transitions (continuous phase transitions) in condensed matter physics, such as the phase transitions of low-temperature superfluidity and low-temperature superconducting [16].
Asymmetric phase transitions occur as a transfer between different pairs of symmetry states P[Gi - ] and P[Gi + ]. Both the length and i j direction of the energy vector change before and after the asymmetric transition. Such phase transition includes alloy phase transition and liquid-crystal phase transition. As seen in Table 5, the G-type transition is accompanied by a leap or drop in the order degree, so the response function diverges during the transition process.
Liquid Crystal Phase Transition
Liquid crystal (LC) is a state of materials whose properties lie between those of conventional liquids and those of solid crystals [23]. There are two main categories of liquid crystals: thermotropic liquid crystals and solvatropic liquid crystals. Typical thermotropic liquid crystals consist of rod-shaped organic molecules that can exhibit smectic, nematic, and isotropic phases as the temperature changes. The structure of liquid crystal materials is characterized by the orders of the molecular position and molecular orientation. The molecules of smectic phase have ordered positions and ordered orientations; the molecules of nematic phase have disordered positions and ordered orientations; and the molecules of isotropic phase have neither ordered positions nor ordered orientations.
Liquid crystals are in the liquid zone, and their energy scale is Ks = kT. The main energy of LC is the translation energy K, the front energy is the rotation energy L, and the back energy is the vibration energy H. The positional order of the LC molecules decreases because of the increase of the translation energy, and the directional order decreases because of the increase of the rotation energy. During the heating process, both the translational and rotational energies increase, and the positional and directional orders gradually decrease, so that the smectic-nematic transition and the nematic-isotropic transition occur sequentially. The inversion of the vibrational energy during the heating process is the critical point of the phase transition. As seen in Table 5,![]() represent two 3 4 4 5 processes of the asymmetric transition, in which both the main and front energies are increasing only the back energy is decreasing. They are consistent with the mechanism of the LC phase transition described above. The author believes that the phenomenon of high- temperature superconduction also belongs to the asymmetric phase transition.
represent two 3 4 4 5 processes of the asymmetric transition, in which both the main and front energies are increasing only the back energy is decreasing. They are consistent with the mechanism of the LC phase transition described above. The author believes that the phenomenon of high- temperature superconduction also belongs to the asymmetric phase transition.
The concept of order degree in real-particle statistics is different from the that of order parameter in mean field theory [16]. The order degree contains two scalars (a,b), only one of which is independent because there is a constraint (2ab = 1). The order parameter has different forms in different systems. For example, the order parameter for the liquid-gas phase transition is a scalar (density difference), the order parameter for the ferromagnetic phase transition is a vector (magnetization intensity), and a tensor order parameter is used to describe the LC phase transition [23].
Conclusion and Prospect
In particle statistics, the energy vector E = (H,K,L) represents the macroscopic state of the system, the midson distribution vector Cτ = (C1, C2,C3,.., CN) represents the microscopic state of the system, and the order degrees (a,b) are the macroscopic parameters representing the particle interactions. In this paper, the complete thermodynamic function relationships are derived by the midson statistics, and the state change rules and phase transition mechanism are studied according to the energy space structure.
The theory of real particles is based on barycenter frames of reference. The barycenter reference frames are finite systems that can be applied to different levels of objects. The motion of objects in the barycenter frame has unique modes and rules. The particle flow-field theory provides a unified form of interacting forces, particle dynamics theory provides the dynamic law of a few particles, and particle statistics provides the thermodynamic law of a large number of particle systems. The consistency and universality of the real particle theory determine its wide application prospects in the fields of physics and chemistry.
The material world is both complex and simple. The complexity lies in its ever-changing forms; the simplicity lies in its obedience to the unified physical laws. The real particle theory tells people that nature needs only two primitive particles (protons and electrons) to create a colorful material world. The unity of physical laws is embodied in the mathematical space of higher dimensions: the scale concept proposed by the measurement principle expands the dimension of mathematical analysis, the mode concept derived from the motion principle creates the dimension of energy space, and the nesting concept contained in the structure principle demonstrates a structural dimension. The cyclic symmetry of energy space centrally reflects the unity of the matter world.
References
- Wikipedia contributors. Particle. Wikipedia, The Free Encyclopedia (Retrieved 4 Mar. 2025).
- Jin, S. N., & Ma, Y. L. (2002). Theoretical Mechanics. Higher Education Press.
- Du, D. S., & Yang, M. Z. (2023). Introduction to particle physics.
- Liang, Z. C. (2022). The paradigm shifts of particle theories: Exemplars of sola r and atomic systems. Res Gate Preprint.
- Qian, B. C. (2006). Quantum Mechanics. Higher Education Pres.
- Liang, Z. C., & Chen, N. N. (2023). The distortion and quantization of the Earth’s orbit. J. Phys. Chem. Biophys, 13, 361.
- Liang, Z. C., & Xie, L. H. (2023). Wave or particle? Quantum behavior of solar system. Glob. J. Sci. Front. Res, 23(9).
- Liang, Z. C. (2020). Outline of real physics. Glob. J. Sci. Front. Res. A, 20(3), 9-27.
- Liang, Z. C. (2019, November). Modeling of real particles. In Journal of Physics: Conference Series (Vol. 1391, No. 1, p. 012026). IOP Publishing.
- Liang, Z. C. (2024). A physical theory based on barycenter reference frames I. Principles of particle flow fields.
- Liang, Z. C. (2021). Dark matter and real-particle field theory. Glob. J. Sci. Front. Res. A, 21(6), 27-39.
- Liang, Z. C. (2019). The origin of gravitation and electromagnetism. Theoretical Physics, 4(2), 85-102.
- Liang, Z. C. (2018, September). Essence of light: particle, field, and interaction. In Photonic Fiber and Crystal Devices: Advances in Materials and Innovations in Device Applications XII (Vol. 10755, pp. 80-93). SPIE.
- Liang, Z. C. (2024). A physical theory based on barycenter reference frames II. Principles of particle dynamics.
- Liang, Z. C. (2019). Motion, energy, and state of body particle system. Theoretical Physics, 4(2), 67.
- Su, R. K. (2004). Statistical Physics. Higher education press.
- Liang, X. X., & Ban, S. L. (2023). Statistical Thermodynamics. Science Press.
- Liang, Z. C. (2019). Cluster ensemble statistics of body particle system. New Horiz. Math. Phys, 3, 53.
- Liang, Z. C (2015). Physical principles of finite particle system. Scientific Research Publishing, Inc. USA.
- Liang, Z. C. (2021). Ion-cluster model and charge-transport properties of ionic liquids.
- Bi, L., & Liang, Z. C. (2015). Features of velocity-cross-correlations in ionic liquids. Journal of Molecular Liquids, 212, 775-784.
- Bi, L., & Liang, Z. C. (2015). Investigation on dynamics and self-diffusion coefficient of [BMIM][PF6] via molecular dynamicssimulations. Russian Journal of Physical Chemistry B, 9(6), 978-982.
- De Gennes, P. G., & Prost, J. (1993). The physics of liquid crystals (No. 83). Oxford university press.