Research Article - (2025) Volume 9, Issue 2
A Microscopic Investigation on Atomic Transport Properties for Liquid Al
2Department of Theoretical Physics, University of Dhaka, Bangladesh
3Department of Applied Mathematics and Physics, Valdosta State University, Valdosta, Georgia, United States
4National High Magnetic Field Laboratory, Tallahassee, Florida, United States
5Department of Electrical Engineering, Tokyo University of Science, Japan
6Department of Electrical and Electronic Systems Engineering, Faculty of Engineering, Saitama University, Japan
7Faculty of Social Science, University of Dhaka, Bangladesh
8Department of Ceramic & Metallurgical Engineering and Technology (RUET), Rajshahi-6204, Bangladesh
Received Date: Mar 12, 2025 / Accepted Date: May 20, 2025 / Published Date: Jun 03, 2025
Copyright: ©Â©2025 Fysol Ibna Abbas, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Abbas, F. I., Chakraborty, S., Sugiyama, M., Samad, M., Khan, A. R. (2025). A Microscopic Investigation on Atomic Transport Properties for Liquid Al. J App Mat Sci & Engg Res, 9(2), 01-05.
Abstract
This work focuses on studying the static and dynamic effects of liquid Al. Static method involves interionic pair interaction, static structure factor, pair correlation function, and excess entropy. The dynamic effects are examined using universal scaling laws (USLs) to analyze properties of the liquid state, like shear viscosity and diffusion coefficient. Inter-ionic interaction is the main ingredient to describe the liquid state feature proposed by Brettonet–Silbert (BS) pseudopotential has been utilized for both static and dynamic nature. BS potential procedure employing as a self-consistent microscopic technique that involves the electronic theory of metals and statistical mechanics. The liquid structure is characterized by the use of thermodynamic perturbation theory referred to as Linearized Weeks–Chandler–Andersen (LWCA) theory. We find excellent agreement between theory and experiment for both static and dynamic properties.
Keywords
Al, Liquid Noble Metal, Effective Bretonnet-Silbert Pseudopotential, Lwca Theory
Introduction
Liquid Al, and aluminum-based alloys have excellent physical properties for metallurgical and industrial applications. This usefulness of the Al based alloys urges us to spend efforts to understand their thermophysical properties from the point of view of both theory and experiment [1-9]. The system we are interested in to work on theoretically is the liquid Al at a thermodynamic state T=1200 K. The cause of choosing this system is fourfold. First, the alloy forming elements Al is heavy polyvalent metal, and are sometimes difficult to handle theoretically, particularly, in the framework of pseudopotentials. Second, liquid Al liquid has not been studied yet for T=1200 K within the same framework, to our knowledge, from any microscopic theory, although some attempts are made from the empirical or, semi empirical semi-empirical models [1–3]. Third, experimental data for static structure factors for elemental liquid Al is available in the literature at the thermodynamic state in question [4]. Fourth, the dynamic properties we are interested in investigating theoretically are already measured by different experimentalists [5].
In this work, we study the atomic diffusion and shear viscosity coefficient of liquid Al. Universal Scaling Laws (USLs) have recently gained prominence in the theoretical calculation of transport coefficients, particularly for high-temperature melts [10,11]. In USLs, the thermodynamic variable known as excess entropy is associated with the structure, particularly the pair correlation function of liquid metals. Conversely, the fundamental component of HS theory is the HS diameter, which is derived from the major peak point of the pair correlation function. In addition to the aforementioned concerns, proposed a novel parameter that signifies the hardness and softness of the interaction for the calculation of transport properties [12]. Besides using theoretical and modeling methods, also carried out experiments to study how the self-diffusion coefficients of liquid pure Al change with temperature, using incoherent quasi-elastic scattering theory [13]. Subsequently, they juxtaposed the data with theoretical values derived from the Sutherland-Einstein relation rather than the Stokes-Einstein relation. used coherent quasi-elastic neutron scattering to investigate the diffusion coefficients of liquid aluminum [14]. They looked at their experimental findings and compared them to theoretical results that came from combining the SE relation with MD data from different potentials. In the studies ,we observed that even though existing theories for calculating how atoms move are being looked at in new ways, the effects of temperature and the changes in interactions between ions from pseudopotentials on these movement properties have not been discussed, especially when considering USLs, HS theories, and the Bretonnet and Silbert (BS) potential [10-15]. Therefore, we, in the present article, intend to study for the variational effects of interionic pair interaction obtained from BS local pseudopotential with temperature, on atomic transport coefficients at thermodynamic state T=1200 K.
Theorey
The Effective Partial Pair Potential
The local pseudopotential for the ith component of a metallic alloy may be modeled as a superposition of two terms, one inside and another outside the core,
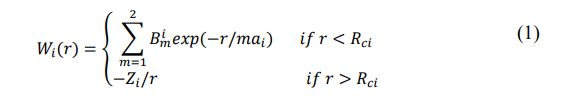
where a, Rci and Z denote the softness parameter, core radius and the effective s-electron occupancy number, respectively [15]. The term outside the core is just the bare Coulomb interaction (in atomic units) between a conduction electron and an ion. The contribution inside the core is contributed by the first two terms of the Dirichlet series arising from the inverse scattering approach. For details, see Ref [4,6]. The coefficients of expansion in the core depends on the parameters ai, Rci, and Zi. Finally, the partial interionic interaction between ith and jth ions is [16,17] .
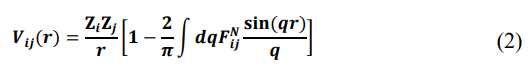
where the normalized energy wave number characteristics [18].
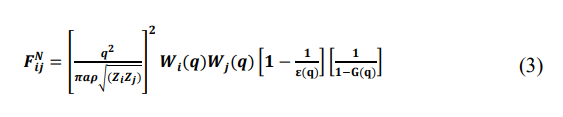
Wi(q), in Equation (11), denotes the unscreened form factor of the ith component obtained from the Fourier transform of Wi(r) (see Eq. (9)), ε(q) and G(q) are dielectric function and the local field factor in momentum space, respectively, with q as the amount of momentum transferred. These functions are taken from Ichmaru and Utsumi because their theory satisfies both the compressibility sum rule and the short-range correlation condition [19].
Pair Distribution Function
In order to have numerical values for partial correlation function, we first calculate the Ashcroft-Langreth (AL) partial static structure factors, Si j(q), and then take a Fourier transform of it,
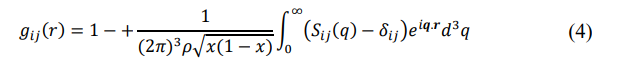
where, ρ is the ionic density of alloys [20]. We note that calculation of Si j(q) requires the knowledge of the effective hard sphere diameters σi j, which is obtained by using the linearized WCA thermodynamic perturbation theory [21].
Scaling Laws
To compute the transport properties of liquid metals and alloys, scaling rules put forth by Rosenfeld and Dzugutov are utilized as a link between structural and thermodynamical qualities [10,11]. In order to determine transport coefficients, Dzugutov and Li [11] took into account microscopic reduction parameters, collision frequency, and HS diameter, while Rosenfeld took into account macroscopic reduction parameters, temperature, and density [11]. suggested a comparable scaling relationship for viscosity after Dzugutov. computed transport coefficients numerically for a variety of systems with varying potentials using the scaling law put forward by Dzugutov [11]. These theories' specifics have been covered in [11]. This study intends to focus for interionic pair interaction obtained from BS model using the USLs with the combination of Dzugutov framework [10,11]. The equation involved for these calculations are mentioned below:

Result and Discussion
This section presents the results of calculations for the static and dynamic properties of liquid Al. We obtained these results from detailed microscopic studies of the ionic transport properties, including diffusion coefficients and shear viscosity. We performed the calculations to investigate the dynamic properties at 1200 K. On the other hand, the static properties were investigated of T = 1323 K. In Figure. 1(b.c) the BS parameters were fitted to the experimental data of static structure factor S(q), and pair correlation function,
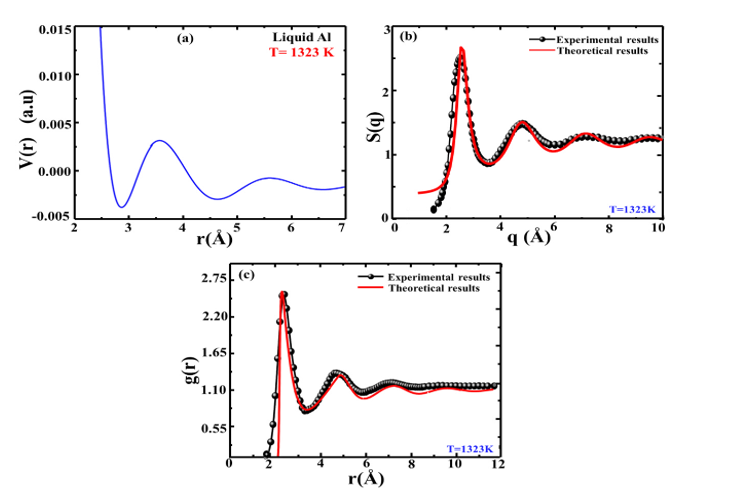
Figure 1: (a) BS Potential Profile, (b) Static Structure Factor, (c) Pair Correlation Function For Liquid Al at T=1323 K. Solid line, and Circle with Black Line Represents the Theoretical and, Experimental Results Respectively.
|
Valency, Z |
Core radius, Rc (a.u.) |
Softness parameter, a (a.u.) |
Number density, r |
Viscosity, h (mPa.s) hexp hSc |
Diffusion, D (109m2s1) Dexp DSc |
||
|
3.0 |
1.91 |
0.49 |
0.05157 |
0.865[9] |
0.91 |
12.65 [14,11] |
14.812 |
Table 1: Input Parameters and Calculated Results
g(r), respectively. The BS fitted parameters were listed in Table 1, namely chemical valence Z, core radius Rc, and softness parameter a. It is noted that the pair correlation function, g(r), is the key ingredient of this study [4, 6]. The estimated effective partial pair potential V(r) (a.u.) for liquid aluminum is shown in Figure. 1(a), and using this potential, the related microscopic inter-ionic correlation g(r) is calculated and displayed in Figure. 1(c). The experimental data perfectly matches this, demonstrating the accuracy of the current work. In the end, these findings about structure and interactions are used to figure out the atomic transport properties based on the electronic theory of metals. No free parameter or anything irrelevant to the interionic interaction enters in this process laterally. From Table 1, it is seen that the values of ÃÃÂ??? and D, calculated using the USLs of Dzugutov with a local field correction, match the experimental data more closely than those obtained using the Li et al. method [21,22]. A few studies claimed that Al has an s-d hybridization effect that is exhibited in the Al cluster [23]. This study has been conducted without consideration of the hybridization effect and found an excellent correlation between the theoretical study with USL's and experimental results. In the branch of material science study, may extend this method to uncover the microstructure properties of other liquid metals, semi-metals, and transition metals.
Conclusion
The results of this study demonstrate that the utilization of USLs is a particularly appropriate approach for analyzing the liquid state of the material attributes. As an additional material property inquiry, this study might also be taken into consideration.
Acknowledgements
The authors (especially FIA) express their sincere appreciation to Tokyo University of Science for giving the study environment to finish this project.
Funding
This work was conducted by personal funding requisition
Conflict of interest
The authors of this work declare that they have no conflicts of interest.References
- Odusote, Y. A., Popoola, A. I., & Oluyamo, S. S. (2016). Bulk and surface properties of demixing liquid Al–Sn and Sn–Tlalloys. Applied Physics A, 122, 1-9.
- Popescu, C. A., & Taloi, D. (2007). Thermodynamic calculation in liquid Al-Sn alloys systems. UPB Sci Bull Ser B, 69(3), 77-84.
- Prasad, L. C., & Mikula, A. (2000). Concentration Fluctuations and Interfacial Adhesion at the Solid-Liquid Interface Between Al2O3 and Αl-Sn Liquid Alloys. High Temperature Materials and Processes, 19(1), 61-69.
- Abbas, F. A., Bhuiyan, G. M. (2018). A study of thermodynamics of mixing for Al1-xSnx liquid binary alloy, J. N. Crys. Solids.481, 391-396.
- Abbas, F. A., Bhuiyan, G. M. (2020). Critical Properties of Segregation for Al1xBix Liquid Binary Alloys, J. Physical Society of Japan. 89, 114004 (1-10).
- Abbas, F. A., Bhuiyan, G. M. (2022). A study of thermodynamics of mixing for Al1-xZnx liquid binary alloy, Physica B: Physics of Condensed Matter. 647,414365(1-6).
- Waseda, Y. (1984). The Structure of Non-Crystalline Materials, McGraw-Hill.
- Hultgren, R., Desai, R.D., Hawking, D.T., Gleiser, M., Keluey, K.K. (1973). Selected Values of the Thermodynamics Properties of Binary Alloys, American Society of Metals.
- Assael, M. J., Kakosimos, K., Banish, R. M., Brillo, J., Egry, I., Brooks, R., ... & Wakeham, W. A. (2006). Reference data for thedensity and viscosity of liquid aluminum and liquid iron. Journal of physical and chemical reference data, 35(1), 285-300.
- Yokoyama, I., & Tsuchiya, S. (2002). Excess entropy, diffusion coefficient, viscosity coefficient and surface tension of liquid simple metals from diffraction data. Materials Transactions, 43(1), 67-72.
- Islam, M. A. M., Gosh, R.C., Abbas, F. I., Bhuiyan, G. M. (2021). Effects of interionic pair interactions on atomic transport properties of liquid Al. Indian J Phys 96(3):697-706.
- Iida, T., Tripathi, N., Isac, M., Guthrie, R.L. (2007). Models and equations for atomic transport coefficients of liquid metals: Viscosity and self-diffusivity. Materials Science Forum 539-543, 2509-2517.
- Kargl, F., Weis, H., Unruh, T., & Meyer, A. (2012, February). Self diffusion in liquid aluminium. In Journal of Physics: Conference Series (Vol. 340, No. 1, p. 012077).
- Demmel, F., Szubrin, D., Pilgrim, W. C., & Morkel, C. (2011). Diffusion in liquid aluminium probed by quasielastic neutro scattering. Physical Review B—Condensed Matter and Materials Physics, 84(1), 014307.
- Bretonnet, J. L., & Silbert, M. (1992). Interionic interactions in transition metals. Application to vanadium. Physics and Chemistryof Liquids, 24(3), 169-176.
- Abbas, F. A., Bhuiyan, G. M. (2019). Local minimum in pair potentials of polyvalent metals: A limitation of pseudopotential theory,International Journal of Modern Physics B. 33, 1950049.
- Abbas, F. A., Bhuiyan, G. M., Ahmed, A. Z. Z. (2016). Energy of mixing and entropy of mixing for CuxAl1-x liquid binary alloys. Materials Science (condmat.mtrl-sci) [cond-mat.mtrl-sci].
- Shimoji, M. (1977). Liquid Metals. An Introduction to the Physics and Chemistry of Metals in the Liquid State. Academic Press London. 1977, 391 p.
- Ichimaru, S., Utsumi, K. (1981) Analytic expression for the dielectric screening function of strongly coupled electron liquids at metallic and lower densities. Physical Review B, 24(12), 7385.
- Ashcroft, N. W., Langreth, D. C. (1967). Structure of binary liquid mixtures. I. Physical Review, 156(3), 685.
- Do Phuong, L., Pasturel, A., & Manh, D. N. (1993). Effect of sd hybridization on interatomic pair potentials of the 3d liquid transition metals. Journal of Physics: Condensed Matter, 5(13), 1901.
- Li, X., Wu, H., Wang, X. B., & Wang, L. S. (1998). s-p hybridization and electron shell structures in aluminum clusters: Aphotoelectron spectroscopy study. Physical review letters, 81(9), 1909.
- Gantefor, G., Chem, W. M. (1994). Shell structure and s-p hybridization in small aluminum clusters. Phys. Lett. 217 600.


