Research Article - (2025) Volume 4, Issue 1
A Comparative Study on the Relationship Between Soil Organic Carbon and Soil Microbial Communities Under Alpine Tree and Shrub Vegetation
Received Date: Apr 17, 2025 / Accepted Date: May 20, 2025 / Published Date: May 30, 2025
Copyright: ©Â©2025 Honglin Li, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Yao, J., Li, X., Wu, F., Wei, G. (2025). A Comparative Study on the Relationship Between Soil Organic Carbon and Soil Microbial Communities Under Alpine Tree and Shrub Vegetation. World J Forest Res, 4(1), 01-06.
Abstract
This study investigates the relationship between soil organic carbon (SOC) and soil microbial communities under various vegetation types within alpine ecosystems. Soils from three tree and three shrub communities in the Maixiu National Forest Park, Qinghai, China, were analyzed for SOC content and microbial community composition. Tree soils exhibited higher SOC content, particularly in the surface layers, which is associated with deeper root systems and greater belowground biomass. In contrast, shrub soils had lower SOC content, indicating faster carbon cycling. Fungal communities, particularly Ascomycetes, were positively correlated with SOC, while bacterial communities, including Proteobacteria and Actinobacteria, were linked to rapid organic matter turnover. These findings highlight the role of vegetation type in regulating microbial communities and carbon dynamics in alpine soils.
Keywords
Soil Organic Carbon, Soil Microbial Communities, Alpine Ecosystems, Vegetation Types
Introduction
Global climate change, driven primarily by greenhouse gas emissions such as carbon dioxide, poses a significant challenge to humanity [1]. Soil, a pivotal carbon reservoir, influences the atmospheric carbon balance through its role in the global carbon cycle [2]. Alpine regions, characterized by unique climates and ecological conditions, host distinct forest ecosystems that significantly impact soil properties [3]. Microorganisms within these soils play a critical role in biogeochemical processes, including organic matter decomposition, nutrient cycling, and carbon dynamics. Particularly in alpine regions, where cold climates slow organic matter decomposition, the distinctive carbon accumulation and release processes exert a substantial impact on the global carbon balance [4,5]. Despite this, our comprehension of the interactions between soil organic carbon (SOC) and soil microorganisms across different alpine forest communities is limited, impeding precise assessments and predictions of alpine ecosystem balances.
Materials and Methods
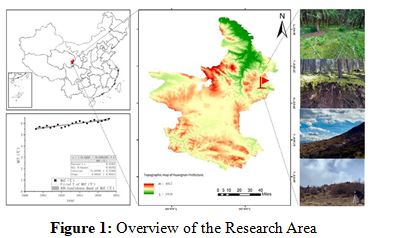
The study area is located in Maixiu National Forest Park (35°8'N–35°21'N, 101°46'E–102°04'E), situated in the Huangnan Tibetan Autonomous Prefecture of Qinghai Province, China. The park lies on the eastern edge of the Qinghai-Tibet Plateau, with an elevation range of 2699–4971 m (Figure 1). The experimental area falls within the park's managed zone and is characterized by a cold and humid climate, with an average annual temperature of approximately 5.8°C, annual precipitation of around 600 mm, and total annual sunshine duration of about 2600 hours [6,7].
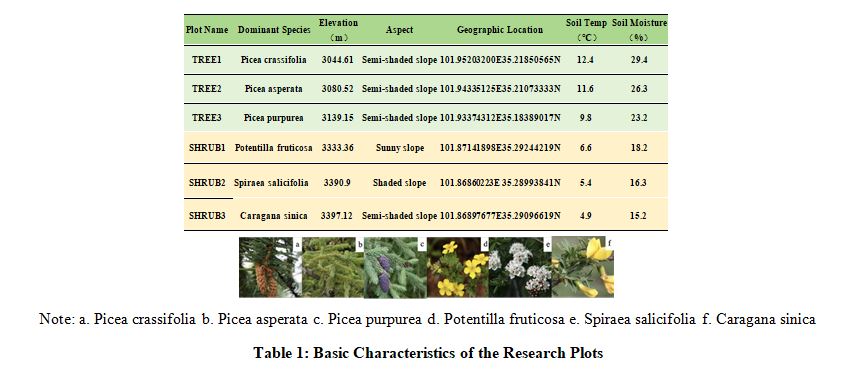
This study examines the interactions between soil microorganisms and SOC in distinct alpine tree and shrub communities within Maixiu National Forest Park. Field and laboratory methods were employed to collect and analyze environmental data (Table 1). SOC was measured using standard volumetric methods, and Illumina sequencing assessed soil microbial diversity and composition. The research explores SOC dynamics, focusing on the regulatory role of microbial communities in organic matter decomposition and carbon transformation. This work aims to elucidate micro-scale carbon dynamics in alpine ecosystems, informing predictions of carbon storage changes and supporting ecosystem management under climate change.
Results and Discussion
Belowground Biomass and Soil Organic Carbon
Content Figure 2 delineates pronounced disparities in belowground biomass and soil organic carbon (SOC) content between tree (TREE) and shrub (SHRUB) communities across soil depths. In the surface layer (0–10 cm), tree soils (e.g., TREE1: 912 ± 50 g/ m²) harbor significantly greater belowground biomass than shrub soils (e.g., SHRUB1: 411 ± 30 g/m²). This trend continues with decreasing depth; at 10–20 cm, TREE1's biomass reduces to 204
± 20 g/m², and SHRUB1 to 104 ± 15 g/m², while at 20–30 cm, values are 106 ± 10 g/m² for TREE1 and 52 ± 5 g/m² for SHRUB1. Correspondingly, SOC content is markedly higher in tree soils in the surface layer (TREE1: 262 ± 20 g/kg vs. SHRUB1: 153 ± 15 g/ kg), decreasing with depth to 52 ± 5 g/kg for TREE1 and 42 ± 5 g/ kg for SHRUB1 at 20–30 cm.
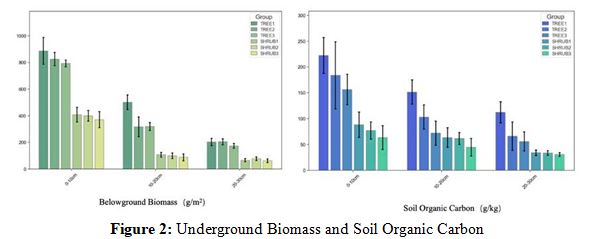
The elevated belowground biomass and SOC in tree communities suggest a more substantial contribution to carbon fixation and organic matter input, attributed to their extensive root systems [8]. Conversely, shrubs, with their shallower root systems, exhibit lower biomass and SOC, indicating reduced carbon sequestration and faster turnover rates [9]. The positive correlation between belowground biomass and SOC underscores the pivotal role of vegetation in carbon cycling, with greater biomass enhancing organic matter input and carbon sequestration potential [10].
Summary: Trees, with their deeper root systems and higher biomass, are more effective in promoting SOC storage and mitigating carbon loss, particularly in surface soils [11]. Shrubs contribute less to long-term SOC accumulation. These findings highlight the decisive influence of vegetation type on soil carbon dynamics, emphasizing the importance of trees in carbon sequestration and soil fertility enhancement [12].
Composition of Soil Fungal and Bacterial Communities
Analysis of Soil Fungal and Bacterial Composition
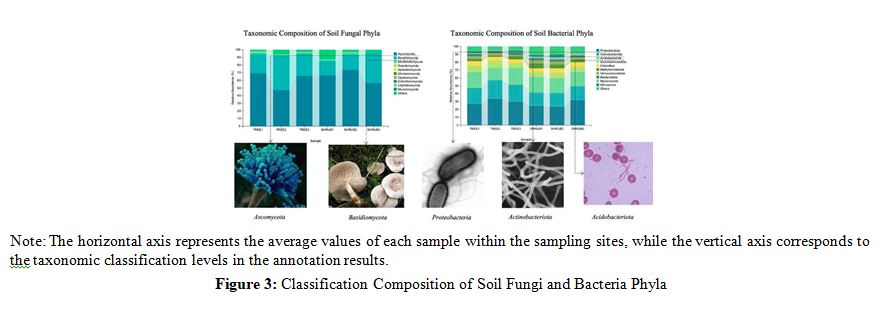
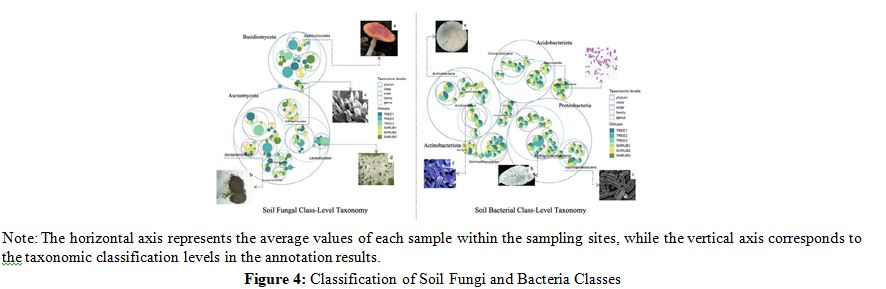
As can be seen from Figures 3 and 4, the soils under trees and the soils under shrubs have different microbial community compositions, with implications for soil organic carbon (SOC) content [13]. Fungal community analysis revealed comparable Ascomycota abundances in tree (TREE1: 0.6949 ± 0.024) and shrub (SHRUB1: 0.6635 ± 0.018) soils, yet SHRUB2 showed the highest Ascomycota abundance at 0.7399 ± 0.025. Basidiomycota was more abundant in tree soils, exemplified by TREE1 (0.2469 ± 0.018) versus SHRUB1 (0.1877 ± 0.016). Bacterial community variations were subtle; Proteobacteria abundance was consistent across tree (TREE1: 0.2687 ± 0.021) and shrub (SHRUB1: 0.2437 ± 0.017) soils, while Actinobacteriota was more prevalent in TREE1 (0.2033 ± 0.019) than SHRUB1 (0.1695 ± 0.015). Acidobacteriota peaked in TREE3 (0.2109 ± 0.018) and was less in SHRUB3 (0.1822 ± 0.015). Correlative analyses indicated that higher Ascomycota abundances, particularly in SHRUB2 and TREE1, coincided with greater SOC content. Proteobacteria and Actinobacteriota abundances also tracked SOC levels; TREE2 and SHRUB2, with elevated Proteobacteria, showed SOC contents of 119.09 ± 5.12 g/kg and 50.11 ± 4.32 g/kg, respectively. These correlations emphasize the microbial taxa's pivotal role in SOC dynamics.
Synthesis and Implications: Significant disparities in the compo- sition of fungal and bacterial communities between tree and shrub soils directly influence soil organic carbon (SOC) accumulation [14]. Ascomycota and Basidiomycota are pivotal in decomposing and transforming organic carbon, with Ascomycota particularly adept at degrading complex organic materials like lignin and cel- lulose [15]. Proteobacteria and Actinobacteriota contribute signifi- cantly to soil carbon cycling by rapidly decomposing fresh organ- ic matter, thereby enhancing SOC content [16]. These microbial taxa, under varying vegetation types, act as key regulators of SOC dynamics, reflecting the distinct ecological strategies of trees and shrubs in modulating soil carbon processes and, consequently, the carbon dynamics within alpine forest ecosystems [17].
Analysis of Soil Fungal and Bacterial Diversity Differ- ences
In the analysis of alpha diversity indices for soil fungi (Figure 5.1) and soil bacteria (Figure 5.2), it was observed that the shrub community (SHRUB1) demonstrated significant advantages across multiple diversity metrics. Specifically, the Chao1 index for the SHRUB1 group was 462±20 for fungi and 4613±220 for bacteria, both significantly higher than those of other communities, reflecting a higher species richness. The Simpson index was 0.98±0.01 for fungi and 0.99±0.01 for bacteria, indicating a higher species diversity within the SHRUB1 group. The Shannon index further confirmed the higher species diversity and evenness in the SHRUB1 group, with values of 6.5±0.5 for fungi and 11.0±0.5 for bacteria. These results suggest that the shrub community may provide a more favorable ecological environment for soil microorganisms, thereby promoting an increase in microbial diversity. Statistical analysis revealed that these differences were significant at the p<0.05 level, underscoring the crucial role of shrub communities in maintaining soil microbial diversity.Bacteria, with their higher diversity and uniformity, are likely central to rapid organic matter decomposition and short-term carbon release [18]. In contrast, fungi, despite lower diversity, are crucial for long-term carbon stabilization, particularly in decomposing complex organic materials [19]. Shrub soils, with their higher microbial diversity, may hasten carbon cycling, while tree soils, with richer bacterial communities and greater biomass inputs, are more significant for long-term carbon sequestration [20].
Summary: The differential diversity of bacteria and fungi significantly impacts soil carbon dynamics, with bacteria driving short-term carbon flux and fungi focusing on long-term carbon fixation [21]. The interplay between vegetation types and microbial community structure is pivotal in shaping carbon cycling processes in alpine forest ecosystems [22].
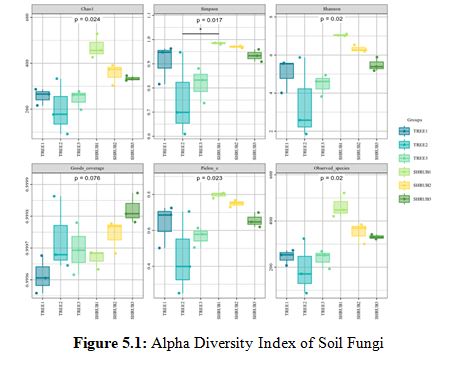
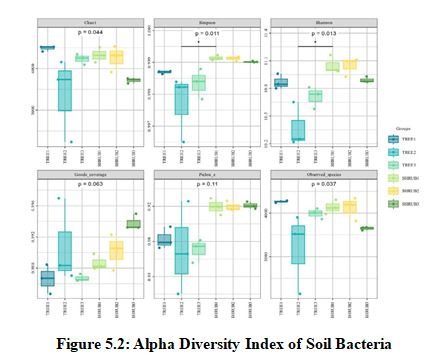
Note: The horizontal axis represents the average values of each sample within the sampling sites, while the vertical axis corresponds to the values of the respective alpha diversity indices. In the boxplot, the symbols have the following meanings: the top and bottom lines of the box represent the upper and lower quartiles (Interquartile Range, IQR); the line within the box indicates the median; the upper and lower edges represent the maximum and minimum values (outliers within 1.5 times the IQR); points outside the upper and lower edges indicate outliers.
Conclusion
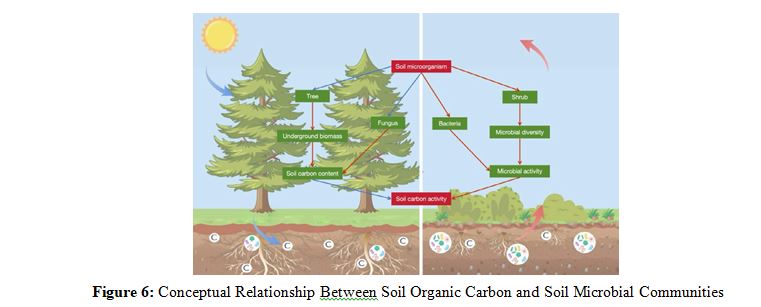
As depicted in Figure 6, This study provides a comprehensive analysis of the interactions between soil organic carbon (SOC) and soil microbial communities under various vegetation types in alpine ecosystems. Soils dominated by trees, particularly coniferous species, exhibit substantial potential for carbon sequestration due to their deeper root structures and greater belowground biomass, which enhances the input of organic matter and promotes long-term SOC accumulation. In contrast, shrub soils, with their lower biomass, display significantly lower SOC content, suggesting less efficiency in the long-term stabilization of SOC. Fungal communities, especially those belonging to the Ascomycota, and bacterial communities, including Proteobacteria and Actinobacteriota, play crucial roles in regulating SOC content, with the latter being associated with rapid organic matter turnover and short-term carbon fluxes. The study underscores the significance of vegetation type in influencing the structure and function of soil microbial communities, thereby affecting carbon dynamics in alpine soils. It suggests that forest management strategies should prioritize tree species with deep root systems for effective carbon storage. Understanding the relationship between soil microbial diversity, SOC content, and vegetation type is essential for predicting future changes in carbon storage in response to climate change, particularly in high-altitude regions.
References
1. Yoro, K. O., & Daramola, M. O. (2020). CO2 emission sources, greenhouse gases, and the global warming effect. In Advances in carbon capture (pp. 3-28). Woodhead Publishing.
2. Li, C., Wang, Y., Yi, Y., Wang, X., Santos, C. A. G., & Liu, Q. (2024). A review of reservoir carbon Cycling: Key Processes, influencing factors and research methods. Ecological Indicators, 166, 112511.
3. Canedoli, C., Ferrè, C., El Khair, D. A., Comolli, R., Liga, C., Mazzucchelli, F., ... & Padoa-Schioppa, E. (2020). Evaluation of ecosystem services in a protected mountain area: Soil organic carbon stock and biodiversity in alpine forests and grasslands. Ecosystem services, 44, 101135.
4. Ramm, E., Liu, C., Ambus, P., Butterbach-Bahl, K., Hu, B., Martikainen, P. J., ... & Dannenmann, M. (2022). A review of the importance of mineral nitrogen cycling in the plant-soil- microbe system of permafrost-affected soils—changing the paradigm. Environmental Research Letters, 17(1), 013004.
5. Schuur, E. A., Bockheim, J., Canadell, J. G., Euskirchen, E., Field, C. B., Goryachkin, S. V., ... & Zimov, S. A. (2008). Vulnerability of permafrost carbon to climate change: Implications for the global carbon cycle. BioScience, 58(8), 701-714.
6. Rao, H. X., Yu, J., Li, S. J., Song, X. P., & Li, D. M. (2021). Gene polymorphisms of Bartonella species in small mammals in Maixiu National Forest Park in the Qinghai-Tibet plateau, China.
7. Xie, M., Chen, Z., & Han, F. (2021). Three new species of eriophyoid mites from Maixiu national forest park, Qinghai Province, China (Acari: Eriophyoidea). å?¨ç?©å??ç±»å¦æ?¥,
46(3), 200-207.
8. Alberti, G., Vicca, S., Inglima, I., Belelli-Marchesini, L., Genesio, L., Miglietta, F., ... & Cotrufo, M. F. (2015). Soil C: N stoichiometry controls carbon sink partitioning between above-ground tree biomass and soil organic matter in high fertility forests. iForest-Biogeosciences and Forestry, 8(2), 195.
9. Tariq, A., Graciano, C., Sardans, J., Zeng, F., Hughes, A. C., Ahmed, Z., ... & Peñuelas, J. (2024). Plant root mechanisms and their effects on carbon and nutrient accumulation in desert ecosystems under changes in land use and climate. New Phytologist, 242(3), 916-934.
10. Forfora, N., Azuaje, I., Vivas, K. A., Vera, R. E., Brito, A., Venditti, R., ... & Gonzalez, R. (2024). Evaluating biomass sustainability: Why below-ground carbon sequestration matters. Journal of Cleaner Production, 439, 140677.
11. Shi, L., Feng, W., Xu, J., & Kuzyakov, Y. (2018). Agroforestry systems: Metaâ?analysis of soil carbon stocks, sequestration processes, and future potentials. Land Degradation & Development, 29(11), 3886-3897.
12. Rodrigues, C. I. D., Brito, L. M., & Nunes, L. J. (2023). Soil carbon sequestration in the context of climate change mitigation: A review. Soil Systems, 7(3), 64.
13. Yan, J., Wang, L., Hu, Y., Tsang, Y. F., Zhang, Y., Wu, J., ...
& Sun, Y. (2018). Plant litter composition selects different soil microbial structures and in turn drives different litter decomposition pattern and soil carbon sequestration capability. Geoderma, 319, 194-203.
14. Wang, H., Liu, S., Chang, S. X., Wang, J., Shi, Z., Huang, X.,... & Cai, D. (2015). Soil microbial community composition rather than litter quality is linked with soil organic carbon chemical composition in plantations in subtropical China. Journal of Soils and Sediments, 15, 1094-1103.
15. Manici, L. M., Caputo, F., Fornasier, F., Paletto, A., Ceotto, E., & De Meo, I. (2024). Ascomycota and Basidiomycota fungal phyla as indicators of land use efficiency for soil organic carbon accrual with woody plantations. Ecological Indicators, 160, 111796.
16. Lan, J., Wang, S., Wang, J., Qi, X., Long, Q., & Huang, M. (2022). The shift of soil bacterial community after afforestation influence soil organic carbon and aggregate stability in karst region. Frontiers in Microbiology, 13, 901126.
17. Zhang, C., Lei, S., Wu, H., Liao, L., Wang, X., Zhang, L., ... & Song, Z. (2023). Microbial network complexity supports the microbial structure stability and soil functionality: Evidence from a natural aridity gradient in alpine ecosystem.
18. Lin, S., Wang, W., Peñuelas, J., Sardans, J., Fernández- Martínez, M., Su, C., ... & Fang, Y. (2022). Combined slag and biochar amendments to subtropical paddy soils lead to a short-term change of bacteria community structure and rise of soil organic carbon. Applied Soil Ecology, 179, 104593.
19. Liu, L., Gunina, A., Zhang, F., Cui, Z., & Tian, J. (2023). Fungal necromass increases soil aggregation and organic matter chemical stability under improved cropland management and natural restoration. Science of the Total Environment, 858, 159953.
20. Lladó, S., López-Mondéjar, R., & Baldrian, P. (2017). Forest soil bacteria: diversity, involvement in ecosystem processes, and response to global change. Microbiology and Molecular Biology Reviews, 81(2), 10-1128.
21. Wang, C., & Kuzyakov, Y. (2024). Mechanisms and implications of bacterial–fungal competition for soil resources. The ISME Journal, 18(1), wrae073.
22. Wang, X., Zhang, Z., Yu, Z., Shen, G., Cheng, H., & Tao, S. (2020). Composition and diversity of soil microbial communities in the alpine wetland and alpine forest ecosystems on the Tibetan Plateau. Science of the Total Environment, 747, 141358.




