Research Article - (2019) Volume 4, Issue 3
A Comparative Open Labelled Study on Ivabradine and Bisoprolol Prescribed in Combination Versus Maximum Dose Titration of Bisoprolol in Patients with Systolic Heart Failure and Left Ventricular Systolic Dysfunction
2Department of Cardiology, Rehman Medical Institute, Peshawar, Pakistan
Received Date: Dec 08, 2019 / Accepted Date: Dec 17, 2019 / Published Date: Dec 31, 2019
Copyright: ©Sadia Akram, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Increased resting heart rate is associated with cardiovascular outcomes in patients with heart failure and reduced ejection fraction (HFrEF). Despite high volume prescribers of beta blockers patients does not achieve recommended target heart rate. The primary objective of this study was to assess the efficacy of ivabradine as adjunct therapy with beta blockers in south east Asian population systolic heart failure and left ventricular systolic dysfunction.
Methodology: This single center, open labelled, randomized study included 113 patients in sinus rhythm with HFrEF and left ventricular systolic dysfunction from outpatient department. Ivabradine was initiated in 45% patients with SR. Patients with LVEF < 35% by Teichholz method, NHYA class II-III, sinus rhythm and resting HR > 70 bpm, already on bisoprolol 5 mg were divided into 2 groups; Group 1 (n= 56) patients were uptitrated to bisoprolol 10 mg and Group 2 (n= 57) patients received ivabradine 5 mg b.i.d in addition to bisoprolol 5 mg. Blood samples for NTproBNP level, an ECG, echocardiogram, NYHA functional class, systolic and diastolic BP were taken at baseline and at the end of 6 months follow-up in both groups
Results: After 6 months HR decreased significantly from 94.82±7.03 to 68.75±5.35 bpm (p < 0.0001), with more patients in NHYA functional Class I than Class II and III and decrease in BNP level from 969.8.3±348.9 to 348.6±230.2 pg/ml (p < 0.0001) in group 2 patients. A significant increase in LVEF was observed with the addition of ivabradine from 31.40±5.37 to 41.68±5.33 % (p < 0.0001). However, mean systolic and diastolic blood pressure was not affected by the addition of ivabradine.
Conclusion: This study concludes that patients with HFrEF demonstrated good tolerability, efficacy and NYHA functional class with the combination of ivabradine and bisoprolol therapy.
Keywords
Ivabradine, Bisoprolol, Combination therapy, Heart failure with Reduced Ejection Fraction, Left ventricular systolic dysfunction, Heart rate, BNP
Abbreviations
LV: Left ventricle
ACC: American College of Cardiology
AHA: American Heart Association
HF: Heart failure
NHYA: New York Heart Association
CHF: Congestive Heart Failure
HFrEF: Heart failure with reduced ejection fraction 2D: Two dimensional
LVEDD: Left ventricle end diastolic diameter
LVESD: Left ventricle end systolic diameter
ECG: Electrocardiogram NT- pro
BNP: N terminal pro brain natriuretic peptide
LVEF: Left ventricular ejection fraction
ACEI: Angiotensin converting enzyme inhibitor
ARB: Angiotensin receptor blocker
MI: Myocardial infarction b.p.m: Beats per minute
AV: Atrio ventricular
HFpEF: Heart failure with preserved ejection fraction
NSR: Normal sinus rhythm
SHIFT: Systolic heart failure treatment with the If inhibitor
BEAUTIFUL: morbidity-mortality evaluation of the If inhibitor ivabradine in patients with coronary disease and left ventricular dysfunction.
Introduction
Heart failure is a clinical syndrome which occurs due to structural or functional abnormalities characterized by inability of heart to fill or eject blood. Mostly all types of cardiac diseases may lead to heart failure with reduced ejection fraction (HFREF).
Cardiac diseases which causes myocardial pressure/volume overload, or decreased contractility triggers adaptive responses to improve cardiac output and maintain blood flow to vital organs. However, when these responses become persistent, they lead to the structural and molecular changes which characterizes ventricular remodeling [1].
It is estimated that heart failure affects more than 23 million people worldwide, with the prevalence of symptomatic heart failure ranging from 0.4-2%. In the Framingham Heart Study, at 40 years of age, the lifetime risk for the development of heart failure for both men and women is 1 in 5 [2].
The annual incidence rates per 1,000 population in the age groups for those 65-74 years of age is 15.2 for males and 8.2 for females, for those 75-84 years of age is 31.7 for males and 19.8 for females and for those ≥ 85 years of age is 65.2 for males and 45.6 for females [3].
Elevated heart rate is a modifiable risk factor for cardiovascular morbidity and mortality as it causes the activation of sympathetic nervous system and withdrawal of parasympathetic activity which are components of neurohumoral response to heart failure. It produces detrimental effects as it decreases the myocardial perfusion and increases myocardial oxygen consumption.
Beta blockers and ivabradine both are effective in treatment of chronic heart failure of ischemic etiology. They have shown to be efficacious in reducing LV remodeling by prolonging the diastolic time & improving left ventricle filling, thus increasing the stroke volume by heart rate reduction [4]. Beta blocker therapy has been a cornerstone in the treatment of patients with HFREF based on strong data from multiple randomized trials including CIBIS II. Besides suppression of ventricular arrhythmias, they improve the diastolic filling time in CHF patients. Beta blocker uptitration has been a focal point in the establishment of CHF clinic across Europe and US since sudden hemodynamic changes associated with beta blocker could determine the long term compliance of such an important therapy in these patients. It is for this reason ACC/ AHA focused update 2005 (management of CHF in adults) recommended beta blocker for symptomatic and asymptomatic patients with heart failure who have LVEF < 45% [5].
Ivabradine is a novel selective inhibitor of the sinoatrial node “If” channels which induces a selective heart rate reduction in humans and animals. It was mainly used as antianginal drug and was given in combination with the beta blockers. It is also used in the management of patients with sinus rhythm (SR) who suffer from moderate to severe heart failure with impaired left ventricular systolic function to reduce morbidity and mortality rate based on SHIFT data. Ivabradine is considered a significant drug in the management of angina pectoris and HFrEF with beneficial effects on cardiac remodeling and capillary density [6].
Despite the safe use of beta blockers as a first line therapy for heart rate reduction in patients with systolic heart failure, patients may not tolerate the target dose well because of side effects such as hypotension, light headedness, lethargy, dyspnea, depression, gastrointestinal disturbances, bronchospasm, excessive bradycardia and AV blocks. In contrast, Ivabradine in combination is safe with lesser adverse effects like headache, dizziness, vertigo, diarrhea [7].
This study compares the efficacy and effects of ivabradine used in combination with beta blocker versus uptitration of beta blocker on HR, LVEF, LV systolic and diastolic dimensions, BNP, and blood pressure in Southeast Asian population with systolic heart failure and left ventricular systolic dysfunction.
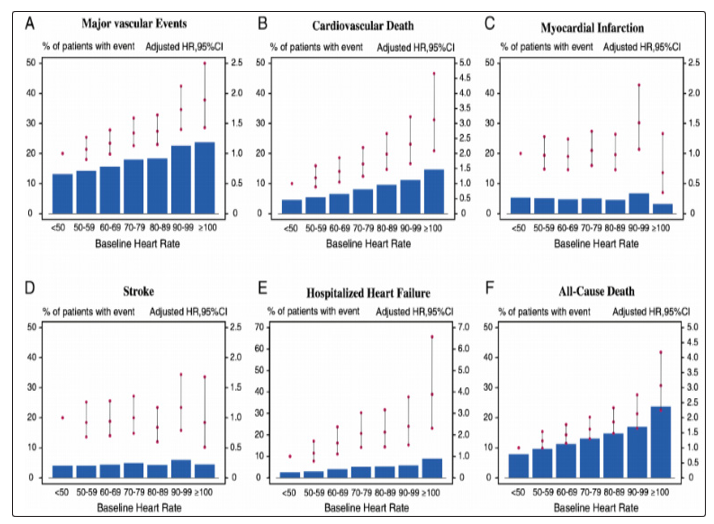
Figure 1.1: Heart rate is associated with increased risk of major cardiovascular events, cardiovascular and all-cause death, chronic heart failure, and stroke, but not with myocardial infarction. Data from secondary analysis of ONTARGET/TRANSCEND study. CI, confidence interval; HR, hazard ratio [8].
Protocol and Study Design
This was a simple, randomized, controlled open labelled, single center based, interventional, parallel group longitudinal study in patients with moderate to severe chronic heart failure and left ventricular systolic dysfunction. The study was planned according to the Good Clinical Quality standards. The protocol was approved by the Ethics Committee of the Khyber Medical University, Peshawar.
Patients were already on standard treatment for heart failure and other comorbidities that included diuretics, angiotensin converting enzyme inhibitors (ACEI) or angiotensin II receptor blockers (ARB), aldosterone antagonist, and beta blocker (mainly bisoprolol).
Inclusion Criteria
Male or female out patients with stable symptomatic chronic heart failure alongside functional capacity of NYHA Class I-III for ≥ 4 weeks with ischemic or non- ischemic etiology and a prior history of hospitalization with worsening heart failure in past 12 months. Left ventricular systolic dysfunction is defined by reduction in left ventricular ejection fraction due to reduced myocardial contractility documented within past 3 months. It is graded as mild (LVEF 41- 45%), moderate (LVEF 36-40%) and severe (LVEF≤ 35%). Patients were in sinus rhythm with resting heart rate ≥ 70 b.p.m selected on two consecutive visits (duration between two visits was 14 days) before randomization measured by a 12- lead electrocardiogram performed after at least 5 minutes rest.
Exclusion Criteria
Exclusion criteria includes mainly male or female patients with left ventricular diastolic dysfunction or preserved left ventricular systolic function with heart failure preserved ejection fraction (HFpEF) and recent MI within 2 months of presentation. Patients with paroxysmal or permanent atrial fibrillation, sick sinus syndrome, history of symptomatic sustained or non sustained ventricular arrhythmias, sinoatrial block, bradyarrhythmias, second and third degree AV block, pacemaker with atrial or ventricular pacing are excluded. Patients with family history of QT prolongation or congenital prolong QT syndrome or treated with certain QT- prolonging products are also excluded. Patients who are eligible candidates for cardiac transplantation or have started CRT within previous 6 months are excluded as well.
Screening, Selection and Data Collection
This study included 113 ambulatory, clinically stable symptomatic patients selected from Rehman Medical Institute Peshawar, with their written informed consent agreed by their physicians. Patients already on bisoprolol 5 mg od/p.o (control group) were randomly divided into 2 groups. Patients in control group 1 (n= 56) on bisoprolol 5 mg od/p.o were up titrated to bisoprolol 10 mg od/p.o (treatment group 1) while patients in control group 2 (n= 57) received ivabradine 5 mg b.i.d/p.o in addition to bisoprolol 5 mg od/p.o (treatment group 2). Patients were evaluated for full clinical workup at selection (visit 0), after 2 weeks (visit 1), at 4 weeks (visit 2), at 12 weeks (visit 3), and finally at 24 weeks after completion of treatment follow up of study (visit 4). Following data was collected at all the visits I) medical history with physical examination and duration of illness II) NYHA class of heart failure III) concomitant medications IV) physical examination of patient V) reported adverse effects VI) systolic and diastolic blood pressure VII) laboratory analysis for BNP level VIII) 12- lead electrocardiogram and IX) echocardiography.
After the initiation of treatment at day 14th, patients were seen on 4th week to check for possible signs of bradycardia, if there were any signs reported the patient was to be withdrawn from the study. Patients were informed to visit immediately if any symptoms that led to withdrawal side effects reported with the medication.
Table 1.1: Design of Study
|
|
Patients with stable chronic heart failure, NYHA Class I-III, treated by Bisoprolol 5mg |
|
|
Visit 0 |
Assessment of patients by eligibility criteria Allocation of patients to control groups (1 & 2) |
|
|
Visit 1 (randomization, Day 14th) |
Standard therapy: uptitration of Bisorpolol 5mg od to Bisoprolol 10mg od (Treatment group 1) |
Addition of Ivabradine 5mg/day b.i.d (Treatment group 2) |
|
Visit 2 (Week 4) |
Check for possible signs of bradycardia and other adverse effects |
|
|
Visit 3 (Week 12) |
Assessment of treatment efficacy and tolerability |
|
|
Visit 4 (Week 24) |
Assessment of treatment efficacy and tolerability End of Study |
|
Table 1.2: Main characteristics of Group 1 (SMT) & Group 2 (SMT + Ivab)
|
|
Group 1 (SMT) |
Group 2 (SMT + Ivab) |
|
Age (Mean ± SD) |
68.4 ± 5.7 |
67.8 ± 6.2 |
|
Gender (Males/ Females) (%) |
71.4/28.5 |
73.6/26.3 |
|
Smokers (%) |
33.9 |
36.8 |
|
Diabetes (%) |
78.5 |
80.7 |
|
Hypertension (%) |
82.1 |
84.2 |
|
Previous MI (%) |
87.5 |
89.4 |
|
Previous Revascularization (PCI/CABG) (%) |
80.3 |
82.4 |
|
NYHA Functional Class (II/III) (%) |
26.7/73.2 |
24.5/75.4 |
|
Aspirin/Antiplatelet drugs (%) |
87.5 |
89.4 |
|
ACEI/ARB (%) |
82.1 |
84.2 |
|
Diuretics (%) |
98.2 |
100 |
|
Statins (%) |
89.2 |
91.2 |
Statistical Analysis
The analysis was performed using intention to treat method. The mean values of variables in both groups were compared with their respective baseline control groups. Data of qualitative parameters were expressed in frequencies and percentages and quantitative parameters were expressed as mean arithmetic value ± 1 standard deviation (SD). Changes in the quantitative variables between groups at the end of 6 months treatment were analyzed by applying column statistics and using paired Student’s t test at 95% confidence interval. A p value of < 0.05 was considered statistically significant. This analysis was done using Graph Pad Prism 5.
Results
Echocardiography
Standard 2D and M-mode imaging were obtained for the measurements of left ventricle prior to the initiation of therapy and post therapy. Using M mode method from the leading edge of one interface to the leading edge of other interface the diameters of left ventricle both systolic and diastolic were measured. With the help of ultrasound software, LVEF was measured by Teichholz formula [9].
LVEF increased from 33.68±4.52% baseline to 39.79±5.77%, with a significant p value < 0.0001 with the uptitration of bisoprolol fumarate 5 mg od/p.o to bisoprolol fumarate 10 mg od/p.o od after 6 months of therapy. While combination of ivabradine 5 mg b.i.d/p.o with bisoprolol fumarate 5 mg od/p.o improved LVEF to 41.68±5.33 from 31.40±5.37% baseline, with a significant p value < 0.0001 after 6 months of therapy.
Electrocardiogram
Standard 12- lead ECGs were obtained. Heart rate was determined by counting the small boxes between two consecutive R waves (R-R interval) and divided this number into 1500. The rhythm was determined by an upright P wave in lead II that is followed by QRS complex then it is said to be a normal sinus rhythm (NSR). ECGs were also evaluated for any intra cardiac block and intraventricular conduction delay, rhythm abnormalities i.e. atrial fibrillation specifically in ivabradine group as it has been reported [10].
The heart rate decreased from 96.33±8.03 to 81.58±8.87 b.p.m baseline, with a significant p value < 0.0001 with the uptitration of bisoprolol fumarate 5 mg od/p.o to bisoprolol fumarate 10 mg od/p.o after 6 months of therapy. While combination of ivabradine 5 mg b.i.d/p.o with bisoprolol fumarate 5 mg od/p.o decreased heart rate to 68.75±5.35 from 94.82±7.03 b.p.m baseline, with a significant p value < 0.0001 after 6 months of therapy.
BNP
Standard BNP test was performed to measure the level of brain natriuretic peptide hormone in the blood at the start and endpoint of treatment. BNP test was performed by drawing a blood sample from patient. The values were evaluated in picograms per milliliter (pg/ml) [11].
The level of BNP decreased in bisoprolol fumarate 10 mg od/p.o group from baseline 927.3±350.3 to 460.4±311.9 pg/mL, with a significant p value < 0.0001 after 6 months of therapy. While with the addition of ivabradine 5 mg b.i.d/p.o to bisoprolol fumarate 5 mg od/p.o BNP level was reduced from 969.8±348.9 at baseline to 348.6±230.2 pg/mL, with a significant p value < 0.0001 after 6 months of therapy.
Blood Pressure
Standard mercurial sphygmomanometer was used for measurement of blood pressure. Patient was allowed to rest for 5 to 10 minutes to stabilize the heart rate and blood pressure. The values are evaluated in mmHg [12,13].
Pretreatment systolic and diastolic blood pressure was 133.5 ± 9.95 mmHg and 92.37 ± 8.71 mmHg which reduced to 119.6 ± 7.75 mmHg and 80.61 ± 6.88 mmHg respectively, with a significant p value < 0.0001 with the uptitration of bisoprolol fumarate 5 mg od/ p.o to bisoprolol fumarate 10 mg od/p.o od after 6 months of therapy. While combination of ivabradine 5 mg b.i.d/p.o with bisoprolol fumarate 5 mg od/p.o decreased systolic blood pressure from 127.1 ± 7.55 mmHg to 124.8 ± 5.97 mmHg, with a significant p value 0.0144 and diastolic blood pressure from 85.53 ± 7.17 mmHg to 83.77 ± 6.14 mmHg, with a significant p value 0.0011.
Table 1.3: Mean values of changes in different clinical parameters observed in Group 1 patients with bisoprolol 5 mg od/p.o (control group 1) vs. bisoprolol 10 mg od/p.o (treatment group 1) in patients with systolic heart failure and left ventricular systolic dysfunction
|
Groups |
Heart Rate (bpm) |
LVEF (%) |
LVEDD (mm) |
LVESD (mm) |
BNP (pg/ml) |
Systolic BP (mmHg) |
Diastolic BP (mmHg) |
|
Control 1 (Bisoprolol 5 mg) |
96.33 ± 8.03 |
33.68 ± 4.52 |
62.18 ± 5.65 |
51.96 ± 6.06 |
927.3 ± 350.3 |
133.5 ± 9.95 |
92.37 ± 8.71 |
|
Treatment 1 (Bisoprolol 10 mg) |
81.58 ± 8.87 **** |
39.79 ± 5.77 **** |
57.23 ± 5.32 **** |
45.93 ± 6.35 **** |
460.4 ± 311.9 **** |
119.6 ± 7.75 **** |
80.61 ± 6.88 **** |
**** = P < 0.0001
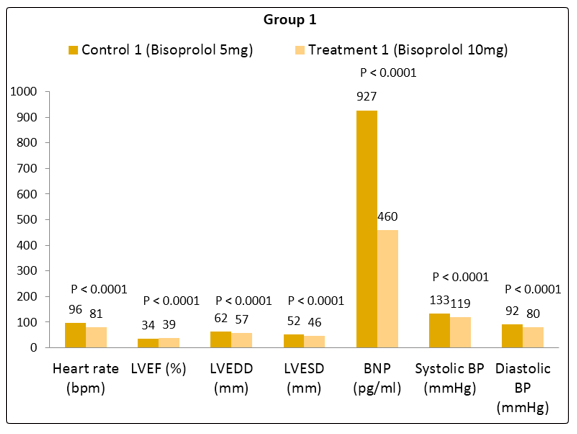
Figure 1.2: Mean values of changes in different clinical parameters observed in Group 1 patients
Table 1.4: Mean values of changes in different clinical parameters observed in Group 2 patients with bisoprolol 5 mg od/p.o (control group) vs. bisoprolol 5 mg od/p.o+ ivabradine 5 mg b.i.d/p.o (treated group) in patients with systolic heart failure and left ventricular systolic dysfunction
|
Groups |
Heart Rate (bpm) |
LVEF (%) |
LVEDD (mm) |
LVESD (mm) |
BNP (pg/ml) |
Systolic BP (mmHg) |
Diastolic BP (mmHg) |
|
Control 2 (Bisoprolol 5 mg) |
94.82 ± 7.03 |
31.40 ± 5.37 |
61.11 ± 6.25 |
51.44 ± 6.22 |
969.8 ± 348.9 |
127.1 ± 7.55 |
85.53 ± 7.17 |
|
Treatment 2 (Bisoprolol 5 mg + Ivabradine 5 mg) |
68.75 ± 5.35 **** |
41.68 ± 5.33 **** |
54.40 ± 4.86 **** |
42.56 ± 5.65 **** |
348.6 ± 230.2 **** |
124.8 ± 5.97 * |
83.77 ± 6.14 ** |
**** = P < 0.0001
** = P = 0.0011
* = P = 0.0144
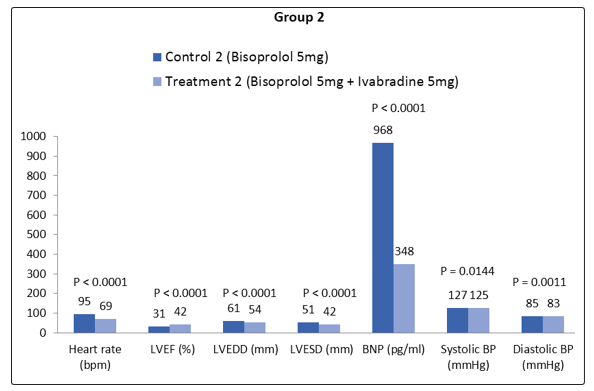
Figure 1.3: Mean values of changes in different clinical parameters observed in Group 2 patients
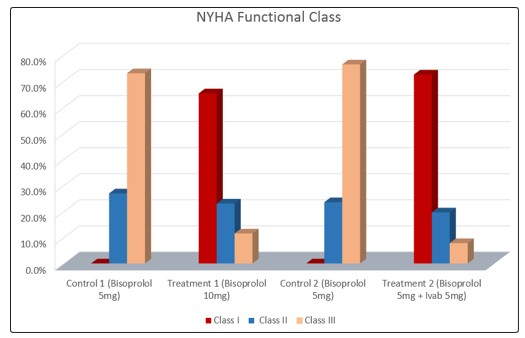
Figure 1.4: Percentage change in NYHA Functional class observed in Control and Treatment groups
Adverse Events
Six patients (10%) reported paroxysmal atrial fibrillation in the first four weeks of initiation of therapy with ivabradine, which required discontinuation of therapy [14]. Five patients reported lethargy (8.7%), six patients reported dizziness (10%) and four patients reported sexual dysfunction (7%) with bisoprolol 10mg od/p.o [15,16].
Discussion
It has been noticed in a clinical practice that only few patients receive target dose of beta blocker that is recommended for systolic heart failure according to ACC/ AHA guidelines and uptitration of beta blocker has been a focal point in the treatment of heart failure patients as it requires time and effort [17].
Ivabradine is a novel selective inhibitor of the sinoatrial node “If” channels which induces a selective heart rate reduction in humans and animals. It is given in combination with the beta blockers in the management of patients with NSR who suffer from moderate to severe heart failure and impaired left ventricular systolic function to reduce morbidity and mortality rate based on SHIFT data [7]. Heart rate reduction with ivabradine could be helpful in a group of patients with systolic heart failure,
(i) who may not tolerate the target dose well because of side effects such as hypotension, lethargy, light headedness, dyspnea, depression, sexual dysfunction,
ii) who have contraindications to beta blockers such as bronchospasm and AV blocks,
(iii) whose heart rate remains high ≥70 b.p.m despite maximum tolerated dose of beta-blockers [18].
This shows that patients who cannot tolerate maximum dose of beta blocker and in whom sufficient heart rate reduction is not achieved despite beta blocker, combination of ivabradine with beta blockers appears to be an appropriate therapeutic regimen. The dose of ivabradine given in this study was clinically well tolerated without any evidence of adverse effects.
The objective of this study was to compare the therapeutic effects of ivabradine 5 mg b.i.d/p.o in combination with bisoprolol 5 mg od/p.o vs. bisoprolol 10 mg od/p.o in patients with sinus rhythm and systolic heart failure with left ventricular systolic dysfunction.
Besides clinical parameters pathologic BNP findings, electrocardiogram monitoring and echocardiographic changes confirm the severity of disease in patients with HFrEF.
Heart rate reduction is a modifiable prognostic risk factor and potential therapeutic target in general population as well as in those with coronary artery disease and heart failure with reduced ejection fraction (HFrEF). Increased resting heart rate is inversely correlated with cardiovascular outcomes and all cause mortality [19].
An elevated heart rate is caused by the activation of sympathetic nervous system and withdrawal of parasympathetic activity which are components of neurohumoral response to heart failure. It produces detrimental effects as it decreases the myocardial perfusion and increases myocardial oxygen consumption [20].
According to the findings of study by Paul Dubach et al. with magnetic resonance myocardial imaging, at baseline mean heart rate was 81±12 (b.p.m) and LVEF was 25.0±7 (%). After 6 months of therapy with bisoprolol fumarate, mean heart rate decreased to 64±13 (bpm) and LVEF increased to 29.2±8 (%), respectively [21].
Left ventricular ejection fraction (LVEF) is a most important prognostic indicator of cardiovascular outcomes and death in patients with systolic heart failure. It is inversely related to mortality, as lower the LVEF, the higher is the mortality rate in heart failure patients with systolic dysfunction [22].
It has been shown that in patients with < 45% ejection fraction, every 10% decrease in LVEF is associated with 39% increase risk of all cause mortality and with every 5% increase in LVEF there is 19% lower relative risk of mortality [23].
Increase in cardiac dimensions such as left ventricular end systolic (LVESD) and left ventricular end diastolic diameters (LVEDD) are important determinant of clinical outcomes in heart failure patients. Patients with heart failure and left ventricular systolic dysfunction show marked improvement in symptoms and reduction in risk of congestive heart failure with relatively reduced size of cardiac chambers [24,25].
Similar findings were observed in a study conducted by Luis Beck da Silva et al. where the baseline heart rate was 84±14 (b.p.m) and left ventricular ejection fraction was 21.7±9.4 (%). The heart rate decreased significantly to 70±10 (b.p.m) and LVEF increased significantly by 7.9% (p = 0.0003) after 4 months of treatment with bisoprolol fumarate [26].
According to the findings of BEAUTIFUL (trial) there is 16% reduction in new onset or worsening of heart failure with the use of ivabradine [27].
The findings of systolic heart failure treatment with the If inhibitor ivabradine trial (SHIFT) shows 18% reduction in risk of cardiovascular death and worsening of heart failure. According to the echocardiographic findings of SHIFT trial, left ventricular ejection fraction was increased by 2.4±7.7% in ivabradine group with 36% patients having ≥5% increase in left ventricular ejection fraction [28].
It has also shown that heart rate and left ventricular ejection fraction are inversely related, as decrease in heart rate is associated with improvement in LVEF [29].
Similarly another echocardiographic study in rats model also shows improved LVEF and marked reduction in LV diameters with the use of ivabradine. BNP is a natriuretic peptide, which is released as a result of increased myocardial wall stress/remodeling. Its activation helps in natriuresis, vasodilation and diuresis [30].
It is an independent predictor of cardiovascular mortality and is used as a diagnostic tool for the management and treatment of heart failure. The rise in the level of BNP is directly proportional to the worsening of heart failure symptoms and myocardial failure [31].
Serkan Ordu et.al demonstrated a study that revealed in ivabradine group vs. non-ivabradine group, the heart rate decreases from baseline 84.10 ± 8.76 to 68.36 ± 8.32 b.p.m after 6 months of therapy, with a significant value (p = 0.001). The NT-proBNP level in ivabradine group was significantly reduced from baseline 1353.02 ± 1453.77 to 717 ± 834.76 pg/mL after 6 months of therapy, with a significant value p < 0.001 [32].
Hypertension is a major independent risk factor for the cardiovascular diseases. Increased vascular tone is directly related to increase in blood pressure and cardiovascular outcomes in hypertensive patients [33].
The risk of heart failure increases by two to three folds affecting 50- 60% population in hypertensive patients. According to Framingham study, about one quarter of heart failure cases are associated with hypertension [34].
Conclusion
Multicenter trials have shown that ivabradine is effective in the treatment of patients with systolic heart failure and left ventricular systolic dysfunction. The main finding from this study is that long- term reduction of heart rate with ivabradine 5 mg b.i.d/p.o and bisoprolol 5 mg od/p.o when used in combination has proven to improve primary efficacy endpoint, left ventricular ejection fraction, symptoms of heart failure and BNP levels significantly. These results indicate that ivabradine can reverse the cardiac remodeling in patients with systolic heart failure and severe LV systolic dysfunction. Thus the combination of ivabradine 5 mg b.i.d/p.o and bisoprolol 5 mg od/p.o used in patients with systolic heart failure has proven to be more efficacious as compared to beta blocker uptitration with tolerated safety profile in a group of Asian population.
Acknowledgements
The authors want to thank Rehman Medical Institute for providing the data of patients.
Ethics Approval and Consent to Participate
Ethical Board of the Khyber Medical University, Peshawar accorded approval of the study protocols via Approval No. Dir/KMU-EB/ EI/000262. Informed consent was obtained from all the individual participants included in the study.
References
- Gheorghiade M BR (1998) A Manifestation of Coronary Artery Disease Chronic heart failure in the United States. Circulation 97: 282-289.
- Sytkowski PA, Kannel WB, D’Agostino RB (1990) Changes in risk factors and the decline in mortality from cardiovascular disease: the Framingham Heart Study. New England Journal of Medicine 322: 1635-1641.
- Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, et al. (2015) Heart disease and stroke statistics-2015 update: a report from the american heart association. Circulation 131: e29.
- Amosova E, Andrejev E, Zaderey I, Rudenko U, Ceconi C, et al. (2011) Efficacy of ivabradine in combination with beta-blocker versus uptitration of beta-blocker in patients with stable angina Cardiovascular drugs and therapy 25: 531-537.
- Investigators C-I (1999) The cardiac insufficiency bisoprolol study II (CIBIS-II): a randomised trial. The Lancet 353: 9-13.
- Fox K, Komajda M, Ford I, Robertson M, Böhm M, et al. (2013) Effect of ivabradine in patients with left-ventricular systolic dysfunction: a pooled analysis of individual patient data from the BEAUTIFUL and SHIFT trials. European heart journal 34: 2263-2270.
- Swedberg K, Komajda M, Böhm M, Borer JS, Ford I, et al. (2010) Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. The Lancet 376: 875-885.
- Lonn EM, Rambihar S, Gao P, Custodis FF, Sliwa K, et al. (2014) Heart rate is associated with increased risk of major cardiovascular events, cardiovascular and all-cause death in patients with stable chronic cardiovascular disease: an analysis of ONTARGET/TRANSCEND. Clinical Research in Cardiology 103: 149-159.
- Lang RM BM, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, et al. (2006) Recommendations for chamber quantification. European Heart Journal-Cardiovascular Imaging 7: 79-108.
- Kligfield P GL, Bailey JJ, Childers R, Deal BJ, Hancock EW, et al. (2007) Recommendations for the Standardization and Interpretation of the Electrocardiogram. Circulation 115: 1306- 1324.
- Fischbach FT DM (2009) A manual of laboratory and diagnostic tests: Lippincott Williams & Wilkins.
- Beevers G LG, O’brien E (2001) Blood pressure measurement. BMJ: British Medical Journal 322: 1043.
- Gheorghiade M, Abraham WT, Albert NM, Greenberg BH, O’Connor CM, et al (2006) Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure. Jama 296: 2217-2226.
- Ruairidh I R Martin OP, Mauro Santibáñez Koref, John P Bourke, M Dawn Teare, Bernard D Keavney (2014) Atrial fibrillation associated with ivabradine treatment: meta-analysis of randomised controlled trials. Heart 100: 1506-1510.
- Jane K McGavin GMK (2002) Bisoprolol: a review of its use in chronic heart failure. Drugs 62: 2677-2696.
- Alberto Cordero VBM, Pilar Mazón, Lorenzo Fácila, Vicente Bertomeu-González, Pedro Conthe, et al. (2010) Erectile Dysfunction in High-Risk Hypertensive Patients Treated with Beta-Blockade Agents. Cardiovascular Therapeutics 28: 15-22.
- Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, Ganiats TG, et al (2009) focused update incorporated into the ACC/AHA 2005 guidelines for the diagnosis and management of heart failure in adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines developed in collaboration with the International Society for Heart and Lung Transplantation. Journal of the American College of Cardiology 53: e1-e90.
- Swedberg K, Komajda M, Böhm M, Borer J, Robertson M, et al. (2012) Effects on outcomes of heart rate reduction by ivabradine in patients with congestive heart failure: is there an influence of beta-blocker dose?: findings from the SHIFT (Systolic Heart failure treatment with the If inhibitor ivabradine Trial) study. Journal of the American College of Cardiology 59: 1938-1945.
- Fox K BJ, Camm AJ, Danchin N, Ferrari R, Lopez Sendon JL, et al. (2007) Resting heart rate in cardiovascular disease. J Am Coll Cardiol 50: 823-830.
- Böhm M SK, Komajda M, Borer JS, Ford I, Dubost- Brama A, et al. (2010) Heart rate as a risk factor in chronic heart failure (SHIFT): The association between heart rate and outcomes in a randomised placebo-controlled trial. Lancet 376: 886-894.
- Dubach P, Myers J, Bonetti P, Schertler T, Froelicher V, et al (2002) Effects of bisoprolol fumarate on left ventricular size, function, and exercise capacity in patients with heart failure: analysis with magnetic resonance myocardial tagging. American heart journal 143: 676-683.
- Redfield MM, Jacobsen SJ, Burnett Jr JC, Mahoney DW, Bailey KR, et al. (2003) Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic. Jama 289: 194-202.
- McMurray JJ, Östergren J, Swedberg K, Granger CB, Held P, et al. (2003) Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-Added trial. The Lancet 362: 767-771.
- Sutton MSJ, Pfeffer MA, Plappert T, Rouleau JL, Moyé LA, et al. (1994) Quantitative two-dimensional echocardiographic measurements are major predictors of adverse cardiovascular events after acute myocardial infarction. The protective effects of captopril. Circulation 89: 68-75.
- Greenberg B, Quinones MA, Koilpillai C, Limacher M, Shindler D, et al. (1995) Effects of long-term enalapril therapy on cardiac structure and function in patients with left ventricular dysfunction. Circulation 91: 2573-2581.
- Beck-da-Silva L, De Bold A, Davies RA, Chow BJ, Ruddy TD, et al. (2004) Effect of bisoprolol on right ventricular function and brain natriuretic peptide in patients with heart failure. Congestive Heart Failure 10: 127-132.
- Fox K, Ford I, Steg PG, Tendera M, Ferrari R, et al. (2008) Ivabradine for patients with stable coronary artery disease and left-ventricular systolic dysfunction (BEAUTIFUL): a randomised, double-blind, placebo-controlled trial. The Lancet 372: 807-816.
- Mulder P, Barbier S, Chagraoui A, Richard V, Henry JP, et al. (2004) Long-term heart rate reduction induced by the selective If current inhibitor ivabradine improves left ventricular function and intrinsic myocardial structure in congestive heart failure. Circulation 109: 1674-1679.
- Tardif JC, O’meara E, Komajda M, Böhm M, Borer JS, et al. (2011) Effects of selective heart rate reduction with ivabradine on left ventricular remodelling and function: results from the SHIFT echocardiography substudy. European heart journal 32: 2507-2515.
- Hobbs RE ML, Bott-Silverman C, James KB, Rincon G, Grossbard EB (1996) Hemodynamic effects of a single intravenous injection of synthetic human brain natriuretic peptide in patients with heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. The American journal of cardiology 78: 896-901.
- Maisel AS KP, Nowak RM, McCord J, Hollander JE, Duc P, et al. (2002) Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. New England Journal of Medicine 347: 161-167.
- Ordu S, Yildiz BS, Alihanoglu YI, Ozsoy A, Tosun M, et al. (2015) Effects of ivabradine therapy on heart failure biomarkers. Cardiol J 22: 501-509.
- Julius S, Palatini P, Kjeldsen SE, Zanchetti A, Weber MA , et al. (2012) Usefulness of Heart Rate to Predict Cardiac Events in Treated Patients With High-Risk Systemic Hypertension. Am J Cardiol 109: 685-692.
- Kannel WB CJ (1992) Left ventricular hypertrophy and mortality--results from the Framingham Study. Cardiology 81: 291-298.




