Research Article - (2025) Volume 10, Issue 1
A Comparative Evaluation: Dexmedetomidine vs. Magnesium Sulfate for Hemodynamic Stability in Laparoscopic Cholecystectomy
2Smt. NHL Municipal Medical College, Ahmedabad, India
Received Date: Jan 24, 2025 / Accepted Date: Feb 27, 2025 / Published Date: Mar 25, 2025
Copyright: ©©2025 Dr. Vismit Gami, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Bilwal, K., Shah, S. M., Shah, S., Umath, N., Bera, R., Gami, V. (2025). A Comparative Evaluation: Dexmedetomidine vs. Magnesium Sulfate for Hemodynamic Stability in Laparoscopic Cholecystectomy. J Anesth Pain Med, 10(1), 01-21.
Abstract
Laparoscopic cholecystectomy, a minimally invasive procedure, poses significant anesthetic challenges due to hemodynamic fluctuations caused by pneumoperitoneum and endotracheal intubation. This study compares the efficacy of intravenous dexmedetomidine and magnesium sulfate in stabilizing hemodynamic parameters during anesthesia. A randomized controlled trial was conducted on 60 ASA I-II patients undergoing laparoscopic cholecystectomy. Parameters such as heart rate, blood pressure, sedation, and analgesia were assessed. Results indicate that both drugs effectively attenuate hemodynamic responses, with magnesium sulfate offering superior stability. Dexmedetomidine provided better postoperative sedation. Both agents are valuable anesthetic adjuncts, enhancing patient safety and recovery.
Keywords
Laparoscopic Cholecystectomy, Dexmedetomidine, Magnesium Sulfate, Pneumoperitoneum, Endotracheal Intubation, Postoperative Analgesia
Abbreviations
ASA – American Society of Anesthesiologists
HR – Heart Rate
BP – Blood Pressure
MAP – Mean Arterial Pressure
SBP – Systolic Blood Pressure
DBP – Diastolic Blood Pressure
NMDA – N-Methyl-D-Aspartate
NS – Normal Saline
CO – Cardiac Output
SVR – Systemic Vascular Resistance
ETCO2 – End-Tidal Carbon DioxideRSS – Ramsay Sedation Score
VAS – Visual Analogue ScaleIV – Intravenous
NMJ – Neuromuscular Junction
ACTH – Adrenocorticotropic Hormone
ICU – Intensive Care Unit
IAP – Intra Abdominal Pressure
CNS – Central Nervous System
RFT – Renal Function Test
LFT – Liver Function Test
ECG – Electrocardiogram
SpO2 – Peripheral Oxygen Saturation
NIBP – Non-Invasive Blood Pressure
Introduction
Laparoscopic cholecystectomy is now the preferred surgical method for gallbladder removal, offering patients less pain, quicker recovery, and minimal scarring. However, the procedure comes with its own challenges, especially during anesthesia. The use of carbon dioxide to inflate the abdomen (pneumoperitoneum) and the process of inserting a breathing tube (endotracheal intubation) can cause sudden spikes in heart rate and blood pressure, increasing the risk of complications.
To manage these effects, anesthesiologists use medications that help stabilize the body’s response. Dexmedetomidine, a drug that calms the nervous system, reduces stress responses and provides pain relief without affecting breathing. Magnesium sulfate, on the other hand, relaxes blood vessels and helps control blood pressure.
This study compares these two drugs to see which one better maintains stable vital signs, reduces the need for anesthesia, and improves patient comfort during and after surgery, ultimately aiming for safer, smoother recoveries.
Aims
To compare the effects of intravenous dexmedetomidine versus magnesium sulfate as an adjunct during anesthesia for laparoscopic cholecystectomy, focusing on hemodynamic stability, anesthetic requirements, and postoperative recovery.
Objectives
• Evaluate Hemodynamic Stability: Compare the impact of dexmedetomidine and magnesium sulfate on heart rate (HR), blood pressure (BP), and mean arterial pressure (MAP) during laryngoscopy, intubation, and pneumoperitoneum.
• Assess Anesthetic Requirements: Determine the effect of both drugs on the total dose of induction agents needed for anesthesia.
• Monitor Perioperative Changes: Observe variations in cardiovascular responses throughout surgery.
• Postoperative Recovery: Compare sedation levels using the Ramsay Sedation Score (RSS) and analgesia using the Visual Analogue Scale (VAS).
• Identify Complications: Assess and document any intraoperative or postoperative adverse effects associated with
Pharmacology of Dexmedetomidine [1-3]
Dexmedetomidine is a highly selective and potent α2 adrenergic agonist with sedative, anxiolytic and analgesic agent.
Chemical Structure:
Dexmedetomidine hydrochloride is the S-enantiomer of Medetomidine.
It is chemically described as (+)-4-(s)- [1-(2, 3- di-methylphenyl) ethyl] -3 H imidazole mono hydrochloride12
Molecular Weight: 236.7 gm/mole
Chemical Formula: C13 H16 N2 HCl
It is relatively unique in its ability to provide sedation without causing respiratory depression.
The α2 adrenergic receptor is a G-protein coupled receptor and are divided into three subtypes including α2A, α2B, α2C. Subtype B found in peripheral vasculature and it is responsible for short term hypertensive response. Subtype C is found in CNS which is responsible for anxiolytic effect.
α2 adrenergic receptors are also found in locus coeruleus, the predominant nor adrenergic nuclei of the brainstem which inhibits the release of norepinephrine by negative feedback and results in the sedative and hypnotic effect, seen with Dexmedetomidine.
Figure: Physiology of alpha-2a Adreno- receptor
PHARMACODYNAMICS
Central nervous system: Sedation, anxiolysis, hypnosis and amnesia Dexmed provides a dose dependent increase in anxiolysis and sedation through the endogenous sleep promoting pathways, generating natural sleep patterns.
Analgesia: Dexmed appears to exert its analgesic effects by an action at α2 receptors within the locus coeruleus and within the spinal cord. It stimulates α2C and α2A receptors in the dorsal horn, thus directly suppressing pain transmission by reducing the release of pro-nociceptive transmitters, substance P and glutamate.
Respiratory effect: Dexmed is able to achieve its effects without causing any respiratory depression.
Cardiovascular effects Dexmed does not appear to have any direct effects on the heart [4]. The administration of a bolus of 1 µg/kg body weight, initially results in a transient increase of BP and a reflex decrease in HR, especially in young healthy patients. The initial reaction can be explained by the peripheral α2 adrenoceptors stimulation of vascular smooth muscles and can be attenuated by a slow infusion over 10 or more minutes. These temporary effects are successfully treated with Atropine or Epinephrine and volume infusions.
Effect on adrenocorticotropic hormone (ACTH) secretion Cortisol’s response to ACTH may be reduced after prolonged use.
Effect on thermoregulation Dexmed is associated with lower rates of shivering. i.v. infusion of Dexmed reduces vasoconstriction and shivering threshold possibly by their activity at α 2B receptors in the hypothalamic thermoregulatory center of brain.
Effect on renal function α2 agonists exert a diuretic effect by inhibiting the anti-diuretic action of arginine and vasopressin at the collecting duct, resulting in decreased expression of aquaporin 2 receptors and water absorption.
Pharmacokinetics
Distribution, Metabolism and Elimination
Dexmed exhibits a rapid distribution phase with a half life of approximately 6 min., a terminal elimination half life of approximately 2 hours after i.v. administration.
The average protein binding is 94% and constant across different plasma concentrations in all gender, which is decreased in patient with hepatic impairment as compared to healthy subjects.
Dexmed undergoes almost total biotransformation, which involves both direct glucuronidation as well as cytochrome P-450 mediated metabolism.
A mass balanced study demonstrated that after nine days, an average of 95% of the radioactivity following i.v. administration of radiolabeled Dexmed, recovered in the urine and 4% in faeces.
Indications
Sedation
It is used for sedation of intubated and mechanically ventilated patients in an ICU. It is also used for non-intubated patient prior to and/or during surgical and other procedures.
Premedication
Especially in pediatric patients, Dexmed is used as a premedication for its anxiolytic, sedative, analgesic, and sympatholytic properties.
Surgical Procedures
It is used as an adjuvant to balanced general anesthesia. It reduces the requirements for volatile anesthetics, sedative and analgesics without causing significant respiratory depression.
• CONTRAINDICATIONS: Hypersensitivity to drug
• PRECAUTIONS
Hypotension, bradycardia and sinus arrest:
Clinically significant bradycardia and sinus arrest have been associated with Dexmed in young, healthy volunteers with high vagal tone or with different routes of administration, including rapid i.v. bolus administration. Rx includes stopping or decreasing the rate of infusion, increasing the rate of fluid administration, elevation of lower extremities and use of pressor agents. For bradycardia, i.v. anticholinergic agents (Atropine) should be considered to modify vagal responses.
Hepatic Impairment: Clearance decreases, dose reduction should be considered.
Use in Pregnancy: No study has been carried out for use of Dexmed during pregnancy.
Adverse Effects
Most frequently observed side effects include hypotension and bradycardia.
Dosage and Administration
Dexmed should be administered using a controlled infusion device. It is not indicated for infusions lasting longer than 24 hours.
It is generally initiated with a loading i.v. infusion of 1 µg/kg over 10 minutes, followed by a maintenance infusion of 0.2-0.7 µg/kg/ hr. The rate of maintenance infusion should be adjusted according to the desired level of sedation. Preparation of the infusion is the same for both loading and maintenance.
Antidote [5]
Atipamazole an α2 antagonist, is indicated for the reversal of the sedative and analgesic and cardiovascular side effects.
Pharmacology of Magnesium Sulphate [6-8]
It is a bivalent ion like calcium with an atomic weight 24.312 amu. Human body contains 24 gm of magnesium. It is the fourth common mineral salt in the body after phosphorus, calcium and potassium and second intracellular cation after potassium. Serum magnesium is divided into three fractions- ionized, protein bound and anion complexes.
Three fractions account for 65%, 27% and 8% in serum concentration respectively.
Structure of Magnesium Sulphate
It acts as a regulator of different ion channel. It has a competitive antagonist action against calcium inflows thereby limit the outflow of calcium from the sarcoplasmic reticulum.so it is a calcium channel blocker and calcium channel modulator. It is involved in hundreds of enzyme reaction in the body.
Pharmacodynamics
Cardio Vascular System
It acts on calcium channels in the cardiac muscle by inhibiting the calcium uptake on the troponin C of myocyte and thereby influencing myocardial contractility. Its vasodilatory action is due to its activation of cyclic AMP.
Neuro Muscular Transmission
It has a preponderant presynaptic and postsynaptic effects. Magnesium competitively blocks the entry of calcium into the presynaptic nerve endings. It also produces progressive inhibition of catecholamine release from adrenal medulla, adrenal nerve endings and adrenergic post ganglionic sympathetic fibers.
Acts as an antagonist of NMDA receptors and this explains its use in post op analgesia.
Magnesium sulphate increases production of prostaglandins causing vasodilatation of small intra cranial vessels which is responsible for its anticonvulsant action.
Respiratory System
It has bronchodilatory action due to the inhibition of smooth muscle contraction, histamine release from the mast cells and acetylcholine release from the cholinergic nerve endings.
Pharmacokinetics
Absorption, Distribution and Elimination
Absorbed in GI tract from diet, Regulated by renal re absorption and excretion.
Clinical Uses
For severe pre eclampsia and eclampsia Magnesium sulphate has a tocolytic effects at serum levels of 8- 10 mEq/L.
Loading dose of 4-6 gm over 20 min i.v., then after the contraction ceases maintenance is done using 2-4 gm per hour i.v. for 12-24 hour.
To reduce stress response during intubation, it is used in the dosage of 30 - 50 mg/kg i.v.
In surgery for pheochromocytoma, it helps to maintain hemodynamic balance because it inhibits the catecholamine release from adrenal medulla and adrenergic nerve endings.
It is also used in the treatment of the Torsades de pointes, as i.v or intra-osseous in the dosage of 25 to 50 mg/kg (up to 2 gm).
In barium poisoning: 1-2 gm, to counteract the intense muscle stimulating effects of barium.
Hypomagnesemia: in case of mild deficiency 1 gm every 6 hours for 4 doses, in severe cases 1-5 gm in divided doses, repeated until the serum levels are normal.
Recent studies shows its use in tetanus patient, at a serum concentration of 2- 4 mEq/L, it gives good control of spasms and muscle rigidity.
Precautions
The drugs should be used with caution in patients with renal impairment.
Monitoring serum magnesium levels and the patient’s clinical status is essential to avoid the consequences of over dosage in toxemia.
Clinical indications of a safe dosage regimen include the presence of the patellar reflex (knee jerk) and absence of respiratory depression (approximately 16 breaths or more /minutes). Serum magnesium levels usually sufficient to control convulsions ranges from 3-6 mEq/100ml (2.5 to 5.0 mEq/L). The strength of the deep tendon reflexes begins to diminish when magnesium level exceeds 4 mEq/L. Reflexes may be absent at 10 mEq/L magnesium, where respiratory paralysis is a potential hazard. An injectable calcium salt should be immediately available to counteract the potential hazards of magnesium intoxication in eclampsia.
Preparations Available
Parenteral injection: Magnesium sulphate - 10%, 12.5%, 50%
Drug Interactions
Central Nervous System Depressants: it has additive central depressant effects with barbiturates, opiates, general anesthetics or other CNS depressants, so dosage of these agents must be carefully adjusted.
Neuromuscular Blocking Agents: Excessive neuromuscular blockade has occurred in patients receiving parenteral magnesium sulphate, these drugs should be administered with caution.
Cardiac Glycosides: Magnesium salts should be administered with extreme caution in digitalized patients, because serious changes in cardiac conduction can result in heart block.
Adverse Reactions
The adverse effects are usually as the result of magnesium intoxication.
These include flushing, sweating, hypotension, depressed reflexes, flaccid paralysis, hypothermia, circulatory collapse, cardiac and CNS depression proceeding to respiratory paralysis.
Hypocalcaemia, with signs of tetany secondary to magnesium sulphate therapy for eclampsia, has been reported.
Symptoms and Treatment of Overdose
In the event of over dosage, artificial ventilation must be provided.
In adults, i.v. administration of 5 to 10 mEq of 10 % calcium gluconate will usually reverse respiratory depression. In extreme cases, peritoneal dialysis or hemodialysis may be required
Catecholamine Release Associated with Tracheal Intubation
• Laryngoscopy and endotracheal intubation cause intense reflex increase in HR, BP and serum concentration of catecholamines.
• Sympathetic innervation via cardio accelerator fiber from the upper five thoracic segments increases the rhythmicity of SA node and enhances the rate and force of contraction.
• Sympathetic system plays a little part in control of heart rate at rest.
• The tracheal intubation following laryngoscopy is not only accompanied by increased sympathetic activity but also increased sympathoadrenal activity. Increased hypothalamic activity and increased traffic in sympathetic efferent tracts are also observed.
• Release of trophic hormones from hypothalamus stimulates release of ACTH, TSH, GH, FSH, LH and PROLACTIN in addition to ADH from pituitary.
• Afferent impulses are carried through trigeminal, glossopharyngeal, vagus and sympathetic nerves from the airway. These impulses are relayed in cranial nerve nuclei, vasomotor and autonomic regulatory areas. The areas that integrate cardiovascular system responses and maintain homeostasis are nucleus solitarius, dorsal vagal nucleus, nucleus ambiguous and parabrachial nucleus.
• The nucleus solitarius is the area of primary central synapse for baroreceptor mediated reflexes and relay station for peripheral information to hypothalamic sympathetic control centers. It projects directly to intermediate lateral nucleus of the spinal cord, the common pathway for pre ganglionic sympathetic outflow. This along with nucleus ambiguous plays an important role in control of secretion of vasopressin.
• Different studies have shown that during intubation, there is rise in MAP of 25mmHg compared to pre intubation values and elevation of plasma nor adrenaline and adrenaline by 45% and 40% respectively.
• Norepinephrine levels may double from 160 to 300 pg/ml and continue for 4 to 8 minutes. Epinephrine levels may quadruple from 70-280 pg/ml.
Laparoscopic Cholecystectomy Hemodynamic and Neuroendocrine Responses After Pneumoperitoneum
Although “minimally invasive” to the patient, a laparoscopic surgery requires artificial pneumoperitoneum which produces significant physiological changes. They can be broadly divided into the following categories:
1. Changes due to the position of the patient
2. Changes due to pneumoperitoneum (raised IAP and CO2 insufflation)
3. Complications
Changes Due to the Position of the Patient
The patients are placed in the Trendelenburg, Reverse Trendelenburg or lithotomy position depend on the surgeries, for better exposure of the surgical field. Pulmonary and cardiovascular changes in these positions are generally similar to, but more extreme than, those associated with the supine position.
Effect on the respiratory system: The patient is placed in the Trendelenburg position intraoperatively during trochar insertion and creating pneumoperitoneum. So, abdominal viscera fall towards diaphragm. Limitation of diaphragmatic movement because of the pressure of viscera on the diaphragm may cause Decreased FRC, lung volumes and compliance, Increased risk of atelectasis, ventilation perfusion mismatch
Raised intracranial pressure and intraocular pressure
Passive regurgitation
Peripheral Nerve injury to brachial plexus, common peroneal nerve in lithotomy position.
Effect on the cardiovascular system: Generally, the Trendelenberg position increases both the VR as well as the CO. During laparoscopic abdominal procedures like in cholecystectomy the reverse Trendelenberg position is used. This position further reduces venous return, which may lead to more fall in cardiac output and arterial pressure. The lithotomy position will induce auto-transfusion by redistributing blood from vessels of the lower extremities into the central body compartment, which thus will increase the preload of the heart.
Changes Due to Pneumoperitoneum
Changes due to CO2 insufflation: The exogenous gas insufflation produces a physiological trespass, which may result in untoward side effects. CO2 is most suitable gas of choice for peritoneal insufflation during laparoscopy as it is odorless, relatively inert, highly soluble in blood hence chances of gas embolism are rare, non-combustible and rapidly buffered in the blood by bicarbonates and excreted via lungs. However, it is irritant and causes referred pain to shoulder tip. Absorption of CO2 from peritoneal cavity causes hypercarbia and is responsible for rise in ETCO2. Hypercarbia causes sympathetic stimulation that results in tachycardia, increased SVR, BP, CVP and CO. This hampers the diastolic filling of coronary arteries.
Changes due to increased intra-abdominal pressure: Normal intra-abdominal pressure (IAP) is 0 to 5 mmHg. Increases in IAP above 10 mmHg are clinically significant, and above 15mmHg can result in an abdominal compartment syndrome, which affects multiple organ systems.
Complications
CO2 subcutaneous emphysema: It is the most common complication due to extraperitoneal insufflation of CO2.
Pneumothorax: It occurs through weak areas and defects in the diaphragm. It may be asymptomatic or may manifest as increased peak airway pressure, decreased SpO2 and hypotension. In severe cases, there can be profound hypotension and cardiac arrest. In case of pneumothorax, the surgery should be stopped and depending on the degree of cardiopulmonary compromise, the pneumothorax may be observed or treated with a thoracostomy tube.
Endobronchial intubation: Elevation of the diaphragm can alter the position of the endotracheal (ET) tube within the trachea. In some cases, the lungs are pushed such that the ET tube is advanced past beyond the carina and into a mainstem bronchus. When this occurs, only one lung ventilation may occur. The non ventilated lung still remains perfused, and as such becomes a large source of intrapulmonary shunt.
Gas (CO2) Embolism: Gas embolism has a mortality rate of nearly 30%. Profound hypotension, arrhythmias, or asystole can occur as a result of a “gas lock” in the vena cava or right ventricle (RV) that interrupts circulation. An increase in ETCO2 is observed. This may be followed by an acute decrease in ETCO2 if there is severe hypotension. The major cause is intravascular insufflation of gas from misplacement of the veress needle or trocar either directly into a vessel or into a parenchymal organ. Initial steps include immediate deflation of the pneumoperitoneum, 100% FiO2, placement of the patient in the left lateral, head down position to remove air from the RV outflow track, and hyperventilation to eliminate the increased PaCO2 caused by the sudden increase in pulmonary dead space. A central line may be required to aspirate gas from the RV. CPR may required. Hyperbaric O2 treatment should be considered if there is suspicion of cerebral gas embolism.
Gastrointestinal System: Patients undergoing laparoscopy might be considered to be at risk for acid aspiration syndrome.
Mesenteric and Hepatorenal Circulation: The Mesenteric and Hepatorenal blood flow is reduced because of compression during raised IAP
Renal Function: Oliguria may occur during intra and postoperative period, which results from alterations in CO and a direct reduction of renal blood flow.
Thromboembolism: An IAP above 14 mmHg, reverse Trendelenburg position, obesity, pelvic surgery and long surgical duration promote venous stasis and increase the chances of thromboembolism.
Temperature Regulation: During laparoscopic surgeries, continuous flow of dry gases can lead to fall in the body temperature.
Neurohormonal Stress Response: Laparoscopy induces classic stress response elucidated by the HPA, simultaneously affecting regulation of glucose metabolism.
Review of Literature:
Laryngoscopy, intubation, pneumoperitoneum and extubation causes a reflex sympathetic discharge characterized by tachycardia and rise in BP [9,10]. This study is to compare effect of preoperative i.v administration of Dexmedetomidine and Magnesium sulphate on hemodynamic changes after study drugs, after induction drug and intubation, after pneumoperitoneum and then at various time intervals, also compared total dose of induction agent, postoperative sedation, analgesia requirement.
In 2009, D. Jee, D. Lee et al studied Magnesium sulphate on attenuation of arterial pressure increase during laparoscopic cholecystectomy [11]. They randomly assigned 32 patients into two groups and control group was given saline 0.5 ml/kg i.v. and other group received 50 mg/kg i.v MgSO4. They observed that arterial pressure was significantly higher in control group than in Magnesium group at 10,20,30 min and post pneumoperitoneum. Norepinephrine/ Epinephrine level was higher in control group than study group each time. No significant difference in baseline cortisol level between two groups. They concluded that administration of magnesium sulphate before pneumoperitoneum attenuated arterial pressure increase which is resulted from decrease in catecholamine release and vasodilatory effect of Magnesium sulphate also provides hemodynamic stability during pneumoperitoneum.
In 2009, Islam M. M, Wafa A Mohsen et al done a study to evaluate a balanced anesthesia with Dexmed on post operative nausea and vomiting after laparoscopic gynecological surgery [12].
A prospective double blind randomized study was done in patients of age group of 17-48 years, ASA 1 and 2 grade, scheduled for elective diagnostic laparoscopic surgery under general anesthesia. Group D received Dexmed infusion at the rate of 0.5 µg/kg while group P received 0.9% NS infusion at same rate. They concluded that combining dexmed to other anesthetic agents results in significant decrease in incidence of postoperative nausea and vomiting with decreasing overall consumption of anesthetic drugs after laparoscopic gynecological surgeries.
In 2012, Ghodki PS, Thombre SK et al done observational study of dexmed as an anesthetic adjuvant in laparoscopic surgery using entropy monitoring, with the aim of avoidance of awareness [13]. They found that there is 62.5% reduction in induction dose of propofol and 30% less requirement of end tidal concentration of isoflurane for maintenance, with adequate depth of anesthesia. They concluded that Dexmed is an effective anesthetic adjuvant and can be used safely without the threat of awareness under anesthesia.
In 2014, Chaithanya K et al done comparison between i.v. 50% Magnesium sulphate and Dexmed for attenuation of cardiovascular stress response during laryngoscopy and endotracheal intubation [14]. They took 60 patients divided into two groups 30 each, group M received 30 mg/kg of Magnesium sulphate and group D received 1 µg/kg Dexmed 10 min before intubation. Various hemodynamic parameters assessed at different time intervals and post-extubation. They observed both magnesium sulphate and dexmed attenuate increase in SBP and DBP but Dexmed effectively controlled HR. They proved that Magnesium sulphate is as effective as Dexmed in attenuating the cardiovascular stress response to laryngoscopy and endotracheal intubation.
In 2015, Pierre zarif, ahmed abdelaal at al done a clinical study to compare Dexmed versus Magnesium sulfate as adjunct during anesthesia for lap [15]. colectomy. They randomly allocated 51 patients into 3 groups: group C received NS infusion 20 ml, group D received Dexmed 1 µg/kg in 20 ml NS and group M received MgSO4 2 gm in 20 ml NS over 15 min and then normal saline, Dexmed at 0.4 µg/kg/min or MgSO4 at 15 gm/kg/min infusion for maintenance. They measured HR, SBP, DBP, MAP at different time intervals, postoperative analgesia and sedation assessment. They found either drugs MgSO4 or dexmed could ameliorates the pressure responses to laryngoscopy and surgical manipulation in lap. colectomy under pneumoperitoneum in trendelenburg position with comparable recovery time and sedation score.
In April 2015, Azim Honarmand, Mohammadreza safavi et al compared different doses of i.v. Magnesium sulphate on cardiovascular changes following the laryngoscopy and tracheal intubation [16]. In this study 120 patients undergoing elective surgery were divided equally into four groups (n=30) and received different doses of MgSO4 (group 1: 30 mg/kg, group 2: 40 mg/ kg, group 3: 50 mg /kg) and equal volume of normal saline as a control group. The patient’s hemodynamic status was recorded at baseline, before laryngoscopy and at various time intervals after laryngoscopy. They concluded that MgSO4 at dose of < 50 mg/ kg can be effective to attenuate cardiovascular instability after laryngoscopy and intubation without significant effect on heart rate changes.
In 2018, Mahajan L et al studied attenuation of pressure response to laryngoscopy and endotracheal intubation with intravenous Dexmed versus MgSO4 under Bi-spectral index controlled anesthesia [17]. They randomized 120 patients into three groups, Dexmed group received 1 µg/kg, Magnesium sulphate group received 30 mg/kg diluted in 100 ml NS, 100 ml NS in group NS 15 min before induction of anesthesia. HR, SBP, DBP were recorded at various time intervals. They found SBP, DBP and HR fell in the group D and group M at laryngoscopy and intubation. They concluded both Dexmed and Magnesium sulphate significantly reduced HR and BP from baseline.
In 2018, Balata AAH et al studied Dexmed versus Magnesium sulphate or Lidocaine for blunting stress response to direct laryngoscopy and endotracheal intubation in abdominal surgeries [18]. Total 87 patient posted for elective surgery randomly divided into three groups, Group D received 1 µg/kg dexmed, group M received 30 mg/kg of Magnesium sulphate and group L given 1.5 mg/kg lidocaine i.v. infusion over 10 min before induction. They observed changes in hemodynamic parameters at different time intervals, blood glucose and cortisol level measured at pre infusion and 10 min post intubation, RSS and postoperative pain. They observed significant attenuation in MAP in group D and group M but failed in group L, also found decreased in mean HR, S. cortisol, S. glucose level, RSS and postoperative pain significantly in group D. They concluded that in both magnesium sulphate and Dexmed groups achieved better hemodynamic stability compared to lignocaine group.
In Dec 2019, Devyani J Desai, Shailee Shah et al conducted a prospective randomized comparative study of intravenous Dexmed and Magnesium sulphate as an adjunct during anesthesia for laparoscopic cholecystectomy [19]. They divided patients of 18 – 60 years of age, ASA grade 1 and 2, into two groups of 35 each. In Group D, Dexmed 1 µg/kg and in group M, Magnesium sulphate 40 mg/kg over 15 min in 20 ml NS were given i.v. before induction of anesthesia. They noticed fall in MAP and HR after giving study drugs, which even fall more after giving propofol in both groups. They observed rise in both parameters after intubation and after pneumoperitoneum but remained lower than baseline throughout surgery in both the groups. Fall in MAP and HR were more in group M than in group D. They recorded mean time for first rescue analgesic and cumulative (24hrs) analgesic dose requirement, which was low in group M but not significant. They noticed comparable sedation score in both groups. They concluded that both drugs were able to attenuate hemodynamic response to laryngoscopy and intubation and surgical manipulation during laparoscopic cholecystectomy, though Magnesium sulphate produced better hemodynamic stability compared to dexmed.
In 2019, Chandrakala M et al compared Magnesium sulphate and Dexmed for Attenuation of hemodynamic stress Response to Intubation [20]. They divided 60 patients into two equal groups, Group-M received 25 mg/kg of Magnesium sulphate while Group-D received 1 µg/kg Dexmed 15 minutes before laryngoscopy and intubation. Hemodynamic parameters noted at 0, 1, 3, 5, 10 minutes after intubation. They noted both Magnesium Sulphate and Dexmed attenuate the rise in systolic and diastolic blood pressures and proved that Magnesium Sulphate is as effective as Dexmed in attenuating the hemodynamic stress response to laryngoscopy and endotracheal intubation. In 2020, Arora V Compared Magnesium Sulphate and Dexmedetomidine for attenuation of vasopressor stress response during laryngoscopy and endotracheal intubation [21]. 90 patients of ASA I and II, divided into three groups of 30 each. Group I was administered i.v. dexmed 1 μg/kg, group II received i.v. magnesium sulphate 30 mg/kg and group III was control group, received NS i.v. and measured HR, SBP, DBP at various intervals and concluded that both Magnesium sulphate and Dexmed significantly reduced the heart rate and blood pressure from baseline.
In 2020, Pathak deba and singha et al done a comparative study between i.v. Dexmed and 50% Magnesium sulfate in attenuation of cardiovascular response to laryngoscopy and endotracheal intubation [22]. They randomized 100 patients into two groups of 50 patients each. Group A received inj. dexmed (1 μg / kg) and Group B received inj. Magnesium sulfate (50 mg/kg). Hemodynamic parameters of patients (HR, SBP, DBP, and MAP) were recorded at different time intervals for the study purpose. They found mean fall in HR, SBP, DBP and MAP values at 2 and 5 minutes of intubation and results were compared with various other similar studies which had analyzed the effect of Dexmedetomidine and Magnesium sulfate for attenuation of cardiovascular response during laryngoscopy and intubation. In 2020, Ghodki PS, Sawle VM et al compared Magnesium sulphate and Dexmedetomidine for attenuation of vasopressor stress response during laryngoscopy and endotracheal intubation [23]. They divided 60 patients between 18 -65 years randomly into two groups. Group D received dexmed 1 µg/kg and group M received 30 mg/kg of MgSO4 diluted to 10 ml normal saline, i.v. over 10 min before intubation, Both groups were observed for changes in hemodynamic parameters. They concluded that Magnesium sulphate is as effective as Dexmed to attenuate stress response to laryngoscopy and intubation, however dexmed may be better choice for controlling HR.
In 2021, Prithiv Rishardhan et al done A Comparative study of Magnesium sulphate and Dexmed in the attenuation of pressor response to intubation and on intraoperative haemodynamic parameters in laparoscopic cholecystectomy [24]. They randomized 60 patients ASA I and II undergoing laparoscopic cholecystectomy into two groups, Group A received Magnesium sulphate 50mg/ kg and Group B Dexmed 1 µg/kg before induction. Baseline values compared with intraoperative hemodynamic changes. Demographic variables were comparable in both the groups. They found Magnesium sulphate was better in controlling the stress response of intubation and pneumoperitoneum with lower VAS scale & better analgesic effect on immediate post-operative pain, which makes it better choice than Dexmedetomidine to use as anesthetic adjuvant during laparoscopic surgery.
In 2022, Mohamed A. M, Handy Magdy Fahim at al compared Dexmed versus Magnesium sulfate as an adjunct to general anesthesia in patients undergoing video assisted thoracoscopy [25]. Patients were randomly allocated into group C (control group), group M and group D, 30 in each, of ASA grade 1 and 2. They observed lower MAP and HR in group D than other two groups. No significant differences in HR in group M and group C, whereas MAP was lower in group M than group C. Postoperative RSS were significantly higher at 1st hour in group D and M, than in group C. VAS score and postoperative analgesic consumption were lowest in group D and lower in group M, compared to group C. They found that group D patients had better hemodynamic stability with better quality of post operative analgesia and sedation compared to group M.
In 2023, Mohan A, Saha G et al compared efficacy of intravenous Magnesium sulphate and intravenous Dexmed in attenuating haemodynamic response to laryngoscopy and endotracheal intubation [4]. They randomly allocated 120 patients into three groups. Group A received i.v. normal saline 50 ml, Group B received i.v Dexmed 1 µg/kg into 50 ml, Group C received i.v. Magnesium sulphate 30 mg/kg in 50 ml NS over 10 min, 10 min before induction. They observed hemodynamic parameters like HR, SBP, DBP, MAP after study drug, 1 min, 3 min, 5 min, 10 min and 15 min after intubation in all three groups. There is no significant difference in baseline Vitals, but significant increase in all parameters in group A at all time intervals, in group B there is significant decrease in all hemodynamic parameters compared to group C which is significant at different time intervals. Based on study they came with conclusion that both Dexmed and Magnesium sulphate effectively controlled the hemodynamic stress response during laryngoscopy and intubation but dexmed demonstrated more significant reduction in sympathetic response.
Material and Methodology
After taking written and informed consent, a total 60 patients of 18-60 yrs ASA status 1 and 2 of either sex, posted for elective laparoscopic cholecystectomy surgery lasting for ≤ 2 hours under general anesthesia were randomly selected and divided into two groups (n=30 for each group). Randomization was done by computer generated random numbers and they were randomly allocated by sealed envelope method.
Exclusion Criteria
Patient refusal
Patient with cardiovascular, respiratory or renal disorders, diabetes, Hypertension, psychiatric illness, known hypersensitivity to drugs
Pregnant and lactating patients.
Patients having baseline heart rate <60/min or taking medications like digoxin or beta-blocker.
Patient with history of sleep apnoea.
Patient with intubation attempt lasting longer than 15 seconds, multiple intubation attempts (2 or more attempts) or having difficult airway (Mallampati grade 3 or 4)
Pre-Anesthetic Analysis
Pre-anesthetic checkup was conducted and a detailed history and complete physical examination and investigations were recorded.
Before Induction
Multipara monitor was applied in each patient.
Patient’s HR, NIBP and SpO2 were recorded before induction (baseline).
An 18 gauge intravenous cannula was inserted and Inj. RL pint started. Patients were randomly divided in two groups, 30 patients in each group.
Group D: Inj. Dexmed 1µg/kg infusion in 20ml NS over 15 min.
Group M: Inj. MgSO4 40 mg/kg infusion in 20ml NS over 15 min.
Both were given through the syringe infusion pump Before premedication.
All patients were received premedication, 0.004mg/kg glycopyrrolate, Inj. Ondansetron 0.08 mg/kg, Inj. Fentanyl 1.5 µg/ kg i.v.
Pre oxygenation was done for 3 minutes after completion of the study drug infusion.
Induction
Inj. Propofol 1.5 - 2 mg/kg
inj. Succinyl choline 2 mg/kg (after check ventilation)
Amount of drug needed for induction was noted down.
Laryngoscopy and tracheal intubation done with appropriate size oral cuffed endotracheal tube. Close circuit and ETCO2 monitor attached. Anesthesia was maintained through closed circuit with O2:N2O(50:50) and sevoflurane, fresh gas flow of 2 liters/ min and intermittent inj. Atracurium (controlled ventilation).
Pneumoperitoneum was created and intraabdominal pressure maintained at 12 - 14 mm Hg. IPPV was delivered with tidal volume 6-8 ml/kg and respiratory rate adjusted to maintain ETCO2 between 35- 45 mmHg. The surgical technique used was same in all groups. Once pneumoperitoneum was created, all patients were operated with head up tilt.
During surgery, Ringer lactate solution was administered according to fasting and maintenance volume.
Intra operative, routine monitoring of HR, NIBP, SpO2 and ETCO2 were done. NIBP measurements were taken in the same arm throughout study. Vitals were recorded at baseline, after study drug infusion, after intubation, before pneumoperitoneum, 1 min, 5 min, 15 min, 30 min, 45 min, 60 min, 90 min, 120 min after pneumoperitoneum, after extubation and 1st and 2nd hour post operatively.
Any intra operative bradycardia was treated with inj. Atropine 0.6 mg and Hypotension was treated with fluid and inj. Mephentermine 6 mg i.v., if needed.
Reversal and Extubation
Inj. Glycopyrrolate 0.008 mg/kg
Inj. Neostigmine 0.05 mg/kg
extubation was done after all extubation criteria fulfilled and vitals were recorded.
Post-Operative
Patient were watched for vitals, any adverse event. All patients were monitored for sedation using Ramsay sedation score and pain via VAS score for upto 120 min.
Post operative pain intensity was assessed using 10 point VAS score, if VAS >3, inj. Diclofenac 75 mg i.v given as a rescue analgesia and was repeated when required. Time for first rescue analgesia and total analgesic drug required in 24 hr were noted.
Postoperative adverse event like Hypotension / Hypertension, bradycardia / tachycardia or nausea / vomiting, if occur were noted and treated accordingly.
Statistical analysis for intra-group comparison was done via student’s paired t- test and unpaired t test was used for inter group comparison of categorical data. Data are expressed as mean (SD) and P value of <0.05 considered significant while, P value <0.001 highly significant.
Performa:
Name:
Age/sex:
Registration no:
ASA grade:
Preoperative assessment:
H/o present illness:
Past medical / surgical history:
P/H/O Anesthesia:
Family history:
General examination:
Temperature: Teeth:
Pulse: Mallampati grade:
Blood pressure:
Respiratory rate: Mouth opening:
Height: Neck extension:
Weight: Spine & back:
Systemic examination:
RS: CNS:
CVS: GIT:
Investigations:
CBC: S. ELECTROLYTES:
RBS: CHEST X-RAY:
RFT: ECG:
LFT: OTHERS:
General Anaesthesia
Patients are divided into two groups, 30 patients in each.
Group D: Inj. Dexmedetomidine 1 µg/kg i.v diluted in 20 ml NS over 15 min.
Group M: Inj. MgSO4 40 mg/kg i.v diluted in 20 ml NS over 15 min.
Pre-Medication
Inj. Ondansetron 0.08 mg/kg i.v.
Inj. Glycopyrrolate 0.004 mg/kg i.v.
Inj. Fentanyl 1.5 µg/kg i.v.
Induction
Inj. Propofol 1.5 - 2 mg/kg i.v.
Inj. Succinyl choline 2 mg/kg i.v.
Intubation:
intubation done with appropriate size, oral cuffed ET tube.
Maintenance
O2 + N2O+ Sevoflurane + Inj. Atracurium intermittently.
Total induction agent dose requirement------------------ Mg
Intra operative Hemodynamic parameter:
|
|
HR |
SBP |
DBP |
MAP |
|
|
Preoperative (Baseline) |
|
|
|
|
|
|
After study drug |
|
|
|
|
|
|
After induction agent |
|
|
|
|
|
|
After intubation |
|
|
|
|
|
|
Before pneumoperitoneum |
|
|
|
|
|
|
After 1 min of pneumoperitoneum |
|
|
|
|
|
|
After 5 min of pneumoperitoneum |
|
|
|
|
|
|
After 15 min of pneumoperitoneum |
|
|
|
|
|
|
After 45 min of pneumoperitoneum |
|
|
|
|
|
|
After 60 min of pneumoperitoneum |
|
|
|
|
|
|
After 90 min of pneumoperitoneum |
|
|
|
|
|
|
At 120 min of pneumoperitoneum |
|
|
|
|
|
|
After extubation |
|
|
|
|
|
|
Post operative |
1st hr |
|
|
|
|
|
|
2nd hr |
|
|
|
|
Duration of surgery---------------- Min.
Post-Operative
Ramsay Sedation Score
1. Patient is anxious and agitated or restless or both.
2. Patient is cooperative, oriented and tranquil.
3. Patient responds to commands only.
4. Patient exhibits brisk response to light glabellar tap or loud auditory stimulus
5. Patient exhibit no response
Table:
|
|
Ramsay sedation score (RSS) |
VAS Scale |
|||||
|
|
1 |
2 |
3 |
4 |
5 |
|
|
|
1 min after extubation |
|
|
|
|
|
|
|
|
15 min |
|
|
|
|
|
|
|
|
30 min |
|
|
|
|
|
|
|
|
60 min |
|
|
|
|
|
|
|
|
120 min |
|
|
|
|
|
|
|
|
|
|
||||||
|
Time for 1st rescue analgesia (Min.) |
|
||||||
|
Total analgesic doses in 24 hours (Mg) |
|
||||||
Complications:
|
|
Intra operative |
Post operative |
|
Bradycardia |
|
|
|
Tachycardia |
|
|
|
Hypertension |
|
|
|
Hypotension |
|
|
|
Nausea |
|
|
|
Vomiting |
|
|
Result and Observation
Total 60 patients are randomly selected and enrolled in our study.
Patients are divided into two groups of 30 each.
Group D: Inj. Dexmedetomidine 1 µg/kg i.v. diluted in 20 ml NS over 15 min
Group M: Inj. MgSO4 40 mg/kg i.v. diluted in 20 ml NS over 15 min
|
Demographic data |
Group M |
Group D |
P Value |
|
|
MEAN±SD |
MEAN±SD |
|
|
AGE |
38.36±7.06 |
40.43±5.23 |
>0.05 |
|
SEX (MALE: FEMALE) |
9:21 |
10:20 |
- |
|
WEIGHT |
61.9±10.36 |
62.8±7.69 |
>0.05 |
|
DURATION OF SURGERY (min) |
100.93±15.86 |
99.06±17.14 |
>0.05 |
|
HEIGHT (cm) |
153.2±5.03 |
155±7.63 |
>0.05 |
Table 1: Demographic Data
Table 1 shows no statistically significant difference in age, sex, weight and duration of surgery
|
Total induction agent dose (mg) |
Group M (MEAN±SD) |
Group D (MEAN±SD) |
P Value |
|
|
121.66±9.49 |
118.33±13.91 |
>0.05 |
Table 2: Total Induction Agent Dose Required
Table 2 shows no significant difference (P value >0.05) in total induction dose of inj. propofol between both group
Hemodynamic Parameters
|
|
MEAN HR |
P VALUE |
MEAN MAP |
P VALUE |
|
Baseline |
73.2 ± 4.31 |
- |
84.48±3.99 |
- |
|
After study drug |
70.2± 4.88 |
<0.05 |
80.53±3.57 |
<0.001 |
|
After induction agent |
69.2± 4.31 |
<0.05 |
77.15± 3.58 |
<0.001 |
|
After intubation |
70.2± 5.64 |
<0.05 |
78.48±3.07 |
<0.001 |
|
Before pneumoperitoneum |
68.66±6.56 |
<0.05 |
77.33±2.95 |
<0.001 |
|
1 min after pneumoperitoneum |
69.46±5.94 |
<0.05 |
79.86±2.98 |
<0.001 |
|
5 min after pneumoperitoneum |
69.53±5.55 |
<0.05 |
81.37±3.43 |
<0.05 |
|
15 min after pneumoperitoneum |
70.46±5.32 |
<0.05 |
82.33±2.89 |
<0.05 |
|
30 min after pneumoperitoneum |
70.53±5.40 |
<0.05 |
82.13±2.87 |
<0.05 |
|
45 min after pneumoperitoneum |
71.33±1.91 |
<0.05 |
81.88±2.71 |
<0.05 |
|
60 min after pneumoperitoneum |
71±2.017 |
<0.05 |
81.95±2.99 |
<0.05 |
|
90 min after pneumoperitoneum |
70.57± 1.80 |
<0.05 |
81.75±2.39 |
<0.05 |
|
At 120 min pneumoperitoneum |
70.5±1 |
<0.05 |
78.5±2.89 |
<0.001 |
|
After extubation |
71.13±1.71 |
<0.05 |
78.35±2.65 |
<0.001 |
|
Post op 1st Hr |
71.06±2.14 |
<0.05 |
80.44±2.08 |
<0.001 |
|
Post op 2nd Hr |
71.4±1.75 |
<0.05 |
82.02±2.89 |
<0.001 |
Table 3: Vitals in Group M
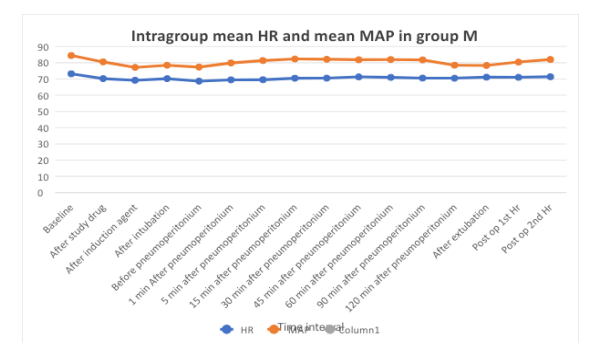
Table 3 shows statistically significant difference in mean HR and mean MAP from baseline value in group M
|
|
MEAN HR |
P VALUE |
MEAN MAP |
P VALUE |
|
Baseline |
73.67±4.55 |
- |
84.84±3.73 |
- |
|
After study drug |
67.66±3.75 |
<0.001 |
82.57±3.43 |
<0.05 |
|
After induction agent |
64.73±4.47 |
<0.001 |
81.02±3.72 |
<0.001 |
|
After intubation |
67.8±4.67 |
<0.001 |
79.86±3.29 |
<0.001 |
|
Before pneumoperitoneum |
66±4.13 |
<0.001 |
79.11±3.35 |
<0.001 |
|
1 min After pneumoperitoneum |
67.06±3.55 |
<0.001 |
81.4±3.19 |
<0.001 |
|
5 min after pneumoperitoneum |
67.43±3.96 |
<0.001 |
83.02±3.01 |
<0.05 |
|
15 min after pneumoperitoneum |
68.2±4.55 |
<0.001 |
85.06±3.20 |
<0.05 |
|
30 min after pneumoperitoneum |
68.27±4.66 |
<0.001 |
87.42±3.02 |
<0.05 |
|
45 min after pneumoperitoneum |
69.06±3.004 |
<0.001 |
88.46±2.80 |
<0.001 |
|
60 min after pneumoperitoneum |
69.53±2.90 |
<0.001 |
88.73±2.40 |
<0.001 |
|
90 min after pneumoperitoneum |
69.13±2.81 |
<0.001 |
88.10±2.18 |
<0.001 |
|
At 120 min pneumoperitoneum |
68±2.96 |
<0.001 |
85.75±3.21 |
<0.001 |
|
After extubation |
69.6±3.53 |
<0.001 |
88.31±2.88 |
<0.001 |
|
Post op 1st Hr |
69.46±3.71 |
<0.001 |
89.37±2.88 |
<0.001 |
|
Post op 2nd Hr |
70.06±3.17 |
<0.05 |
90.91±2.93 |
<0.001 |
Table 4: Vitals in Group D
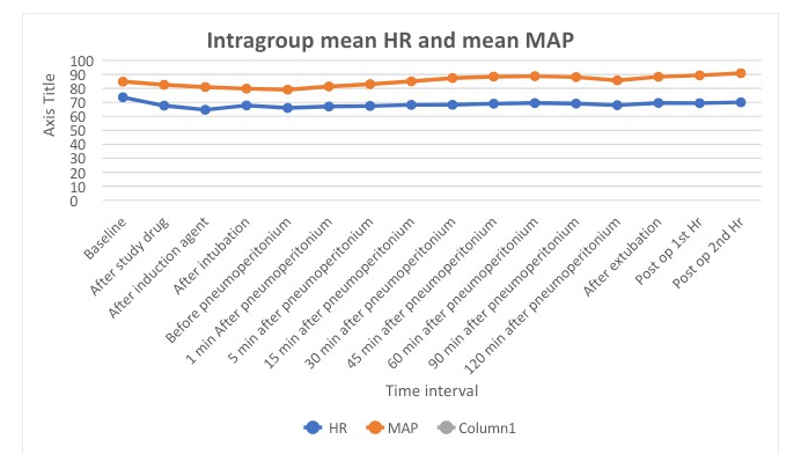
Table 4 shows significant fall in mean HR and mean MAP compared to baseline in group D.
|
|
Group M MEAN±SD |
Group D |
MEAN ± SD |
P Value |
|
Preoperative |
Baseline |
73.2±4.31 |
73.67±4.55 |
>0.05 |
|
|
After study drug |
70.2±4.88 |
67.66±3.75 |
<0.05 |
|
|
After induction drug |
69.2±4.31 |
64.73±4.47 |
<0.001 |
|
|
After intubation |
70.2±5.64 |
67.8±4.67 |
<0.05 |
|
Intraoperative |
Before pneumoperitoneum |
68.66±6.56 |
66±4.13 |
<0.05 |
|
|
1 min After pneumoperitoneum |
69.46±5.94 |
67.06±3.55 |
<0.05 |
|
|
After 5 minutes of pneumoperitoneum |
69.53±5.55 |
67.43±3.96 |
<0.05 |
|
|
After 15 minutes of pneumoperitoneum |
70.46±5.32 |
68.2±4.55 |
<0.05 |
|
|
After 30 minutes of pneumoperitoneum |
70.53±5.40 |
68.27±4.66 |
<0.05 |
|
|
After 45 minutes of pneumoperitoneum |
71.33±1.91 |
69.06±3.004 |
<0.001 |
|
|
After 60 minutes of pneumoperitoneum |
71±2.01 |
69.53±2.90 |
<0.05 |
|
|
After 90 minutes of pneumoperitoneum |
70.57±1.80 |
69.13±2.81 |
<0.05 |
|
|
At 120 minutes of pneumoperitoneum |
70.5±1 |
68±2.96 |
<0.05 |
|
|
After extubation |
71.13±1.71 |
69.6±3.53 |
<0.05 |
|
Post operative |
1st hour |
71.06±2.14 |
69.46±3.71 |
<0.05 |
|
|
2nd hour |
71.4±1.75 |
70.06±3.17 |
<0.05 |
Table 5: Comparison Heart rate (HR) Between Two Groups
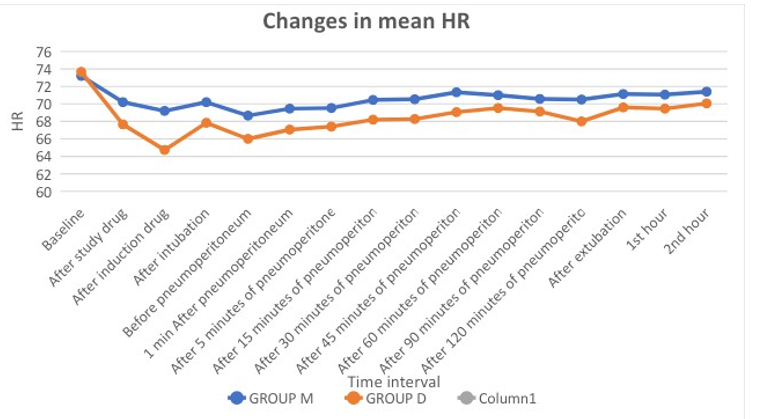
Table 5 shows statistically significant difference in HR in between group M and group D intraoperatively and postoperatively.
|
|
|
Group M Mean ± SD |
Group D Mean ± SD |
P Value |
|
Preoperative |
Baseline |
113.3±3.37 |
113.5±2.67 |
>0.05 |
|
|
After study dose |
108.93±3.00 |
111.46±2.72 |
<0.001 |
|
|
After induction drug |
105.73±3.35 |
109.46±2.92 |
<0.001 |
|
|
After intubation |
105.26±2.94 |
107.46±2.92 |
<0.05 |
|
Intraoperative |
Before pneumoperitoneum |
106.53±2.92 |
108.26±4.41 |
<0.05 |
|
|
After 1 min pneumoperitoneum |
109.2±2.44 |
110.46±3.22 |
<0.05 |
|
|
After 5 minutes of pneumoperitoneum |
110.53±2.82 |
111.73±2.44 |
<0.05 |
|
|
After 15 minutes of pneumoperitoneum |
112.2±2.69 |
113.6±3.12 |
<0.05 |
|
|
After 30 minutes of pneumoperitoneum |
112.9±2.66 |
115.7±3.09 |
<0.001 |
|
|
After 45 minutes of pneumoperitoneum |
109.13±3.54 |
117.93±3.21 |
<0.001 |
|
|
After 60 minutes of pneumoperitoneum |
112.4±2.64 |
117.93±2.25 |
<0.001 |
|
|
After 90 minutes of pneumoperitoneum |
111.63±1.91 |
116.13±2.52 |
<0.001 |
|
|
At 120 minutes of pneumoperitoneum |
105.5±5 |
113.63±3.88 |
<0.05 |
|
|
After extubation |
107.86±2.28 |
117.2±2.60 |
<0.001 |
|
Postoperative |
1st hour |
110±2.10 |
118.4±2.64 |
<0.001 |
|
|
2nd hour |
111.8±2.18 |
119.93±2.54 |
<0.001 |
Table 6: Changes in SBP
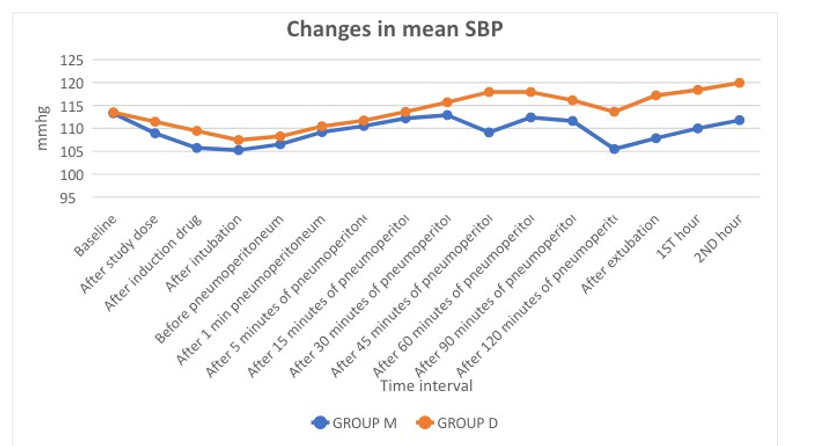
Table 6 shows statistically significant change in SBP intraoperatively and postoperatively between two groups.
|
|
|
Group M MEANS±SD |
Group D MEAN±SD |
P value |
|
Preoperative |
Baseline |
70.06±4.88 |
70.53±4.69 |
>0.05 |
|
|
After study dose |
66.13±4.54 |
68.13±4.36 |
<0.05 |
|
|
After induction drug |
62.87±4.41 |
66.8±4.6 |
<0.001 |
|
|
After intubation |
63.4±3.79 |
66.06±4.05 |
<0.05 |
|
Intraoperative |
Before pneumoperitoneum |
62.73±3.80 |
64.53±4.13 |
<0.05 |
|
|
After 1 min pneumoperitoneum |
65.2±3.95 |
66.86±3.54 |
<0.05 |
|
|
After 5 minutes of pneumoperitoneum |
66.8±4.35 |
68.66±4.07 |
<0.05 |
|
|
After 15 minutes of pneumoperitoneum |
67.4±3.97 |
70.8±3.98 |
<0.05 |
|
|
After 30 minutes of pneumoperitoneum |
66.73±3.76 |
73.26±3.58 |
<0.001 |
|
|
After 45 minutes of pneumoperitoneum |
68.26±3.13 |
73.73±3.43 |
<0.001 |
|
|
After 60 minutes of pneumoperitoneum |
66.73±4.11 |
74.13±3.01 |
<0.001 |
|
|
After 90 minutes of pneumoperitoneum |
66.81±3.24 |
74.08±2.98 |
<0.001 |
|
|
At 120 minutes of pneumoperitoneum |
65±2.58 |
71.81±3.51 |
<0.001 |
|
|
After extubation |
63.6±3.37 |
73.86±3.36 |
<0.001 |
|
Postoperative |
1st hour |
65.66±2.52 |
74.86±3.26 |
<0.001 |
|
|
2nd hour |
67.13±3.91 |
76.4±3.42 |
<0.001 |
Table 7: Changes in DBP
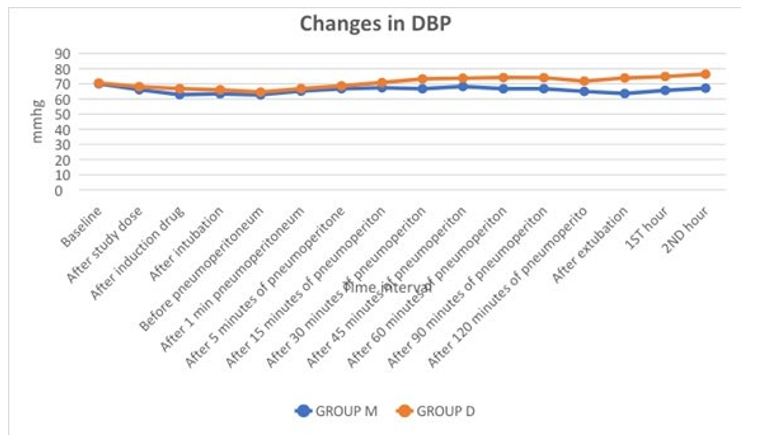
Table 7 shows significant changes in DBP between both groups at various time intervals.
|
|
|
Group M MEANS±SD |
Group D MEAN±SD |
P Value |
|
Preoperative |
Baseline |
84.48±3.99 |
84.84±3.73 |
>0.05 |
|
|
After study drug |
80.53±3.57 |
82.57±3.43 |
<0.05 |
|
|
After induction drug |
77.15±3.58 |
81.02±3.72 |
<0.001 |
|
|
After intubation |
78.48±3.07 |
79.86±3.29 |
<0.05 |
|
Intraoperative |
Before pneumoperitoneum |
77.33±2.95 |
79.11±3.35 |
<0.05 |
|
|
After 1 min pneumoperitoneum |
79.86±2.98 |
81.4±3.19 |
<0.05 |
|
|
After 5 minutes of pneumoperitoneum |
81.37±3.43 |
83.02±3.01 |
<0.05 |
|
|
After 15 minutes of pneumoperitoneum |
82.33±2.89 |
85.06±3.20 |
<0.001 |
|
|
After 30 minutes of pneumoperitoneum |
82.13±2.87 |
87.42±3.02 |
<0.001 |
|
|
After 45 minutes of pneumoperitoneum |
81.88±2.71 |
88.46±2.80 |
<0.001 |
|
|
After 60 minutes of pneumoperitoneum |
81.95±2.99 |
88.73±2.40 |
<0.001 |
|
|
After 90 minutes of pneumoperitoneum |
81.75±2.39 |
88.10±2.18 |
<0.001 |
|
|
At 120 minutes of pneumoperitoneum |
78.5±2.89 |
85.75±3.21 |
<0.001 |
|
|
After extubation |
78.35±2.65 |
88.31±2.88 |
<0.001 |
|
Postoperative |
1st hour |
80.44±2.08 |
89.37±2.88 |
<0.001 |
|
|
2nd hour |
82.02±2.89 |
90.91±2.93 |
<0.001 |
Table 8: Changes in MAP
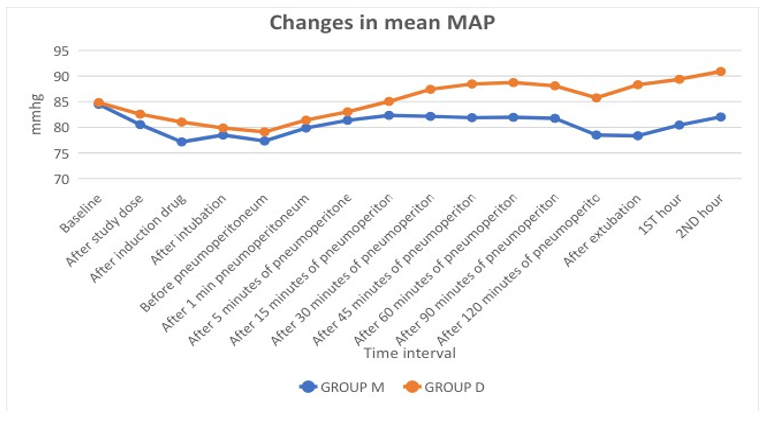
Table 8 shows control of MAP is better in group M compared to group D.
|
|
Group M |
Group D |
|
After study drug |
↓ 4.09% |
↓ 8.1% |
|
After induction |
↓5.46% |
↓12.13% |
|
After intubation |
↓4.09% |
↓7.96% |
|
After pneumoperitoneum |
↓5.10% |
↓8.9% |
|
After extubation |
↓2.82% |
↓5.52% |
Table 9: Changes in HR % from Baseline values
|
|
Group M |
Group D |
|
After study drug |
↓4.6% |
↓2.67% |
|
After induction |
↓8.6% |
↓4.50% |
|
After intubation |
↓7.1% |
↓5.86% |
|
After pneumoperitoneum |
↓5.4% |
↓4.05% |
|
After extubation |
↓7.2% |
↑4.09% |
Table 10: Changes in MAP % from Baseline values
Table 9 and 10 shows changes in HR % and MAP % from baseline value in both groups.
|
|
Group-M |
Group-D |
||
|
Intraoperative |
Postoperative |
Intraoperative |
Postoperative |
|
|
Bradycardia |
- |
- |
2 patients |
- |
|
Tachycardia |
- |
- |
- |
- |
|
Hypotension |
- |
- |
- |
- |
|
Hypertension |
- |
- |
- |
- |
|
Nausea |
- |
- |
- |
- |
|
Vomiting |
- |
- |
- |
- |
Table 11: Complications
Table 11 shows intraoperative bradycardia in two patients in group D.
|
|
Ramsay sedation score (RSS) |
Visual analogue scale (VAS) |
||||
|
|
Group M |
Group D |
P value |
Group M |
Group D |
P value |
|
1 min after extubation |
2.66±0.71 |
2.6±0.67 |
>0.05 |
0 |
0 |
- |
|
15 min |
1.96±0.71 |
1.96±0.66 |
>0.05 |
1.26±0.69 |
1.03±0.7 |
>0.05 |
|
30 min |
1.63±0.49 |
1.5±0.50 |
>0.05 |
2.43±0.63 |
1.87±0.63 |
>0.05 |
|
60 min |
1±0 |
1.06±0.25 |
>0.05 |
3.5±0.57 |
3.2±0.92 |
>0.05 |
|
120 min |
2±0 |
2± 0 |
- |
1.63±0.55 |
2.03±0.71 |
>0.05 |
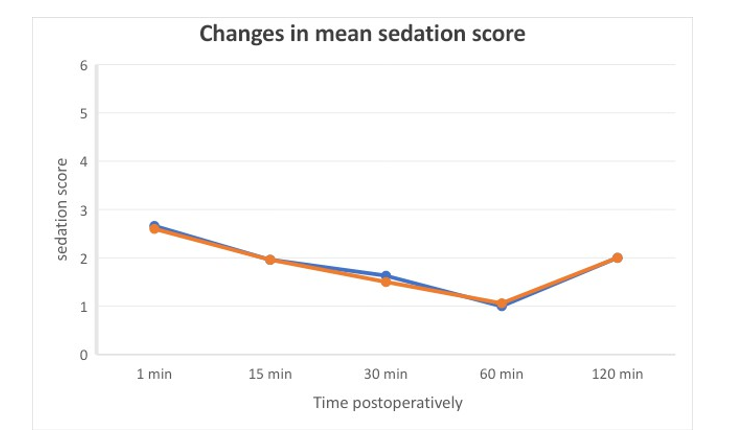
Table 12: RSS and VAS:
Above table shows RSS and VAS score, which is comparable in both groups.
|
|
Group M |
Group D |
P Value |
|
1st Rescue analgesic agent (at --- min) MEAN±SD |
70.03±10.20 |
71.96±12.46 |
>0.05 |
|
Total cumulative dose in 24 hrs (mg) MEAN±SD |
257.16±47.16 |
242.5±50.91 |
>0.05 |
Table 13: Rescue Analgesia and Total Cumulative Dose
Table 13 shows comparable 1st rescue analgesia time and total cumulative dose requirement in between groups
Discussion
Different authors [19,20,21,24] had successfully used Inj. Dexmed 1 µg/kg without major side effects to counteract these hemodynamic changes. In 2009 D Jee at al and many others recommended bolus dose of 40-50 mg/kg magnesium sulphate before pneumoperitoneum to exert this beneficial effect during laparoscopy [11,16,22,24]. In 2015 Azim Honarmad et al compared different doses of magnesium sulphate i.v. (30 mg, 40 mg and 50 mg) and found all of them safe to prevent cardiovascular changes following laryngoscopy and intubation [16]. These justify our study of single bolus dose of Dexmed 1 µg/kg i.v with Magnesium sulphate 40 mg/kg i.v. before induction as an adjunct in patients undergoing laparoscopic cholecystectomy.
Dexmed and Magnesium sulphate significantly reduce the release of catecholamines, especially nor epinephrine, thereby attenuating the increase in systemic vascular resistance [1,2,3,6,7,11]. Bhana et al and Kallio et al reported completely blocked nor epinephrine secretion of sympathetic nerves by single dose of intravenous Dexmed and 92% reduction in plasma norepinephrine levels leading to decrease in MAP and HR. The effect of Magnesium sulphate on hemodynamics is due to its interaction and activation of membrane Ca-ATPase and Na-K-ATPase which are involved in trans membrane ion exchange during depolarization and re polarization phases, thus acting as a cell membrane stabilizer [1,2,6,7]. This calcium inhibitory effect of magnesium sulphate and attenuation of vasopressin stimulated vasoconstriction, causes central arterioler vasodilatation and acts against vasospasm [7,8].
In our study, demographic parameters are comparable in both groups for age, sex, weight, duration of surgery and they are not statistically significant (P value >0.05). Number of female patients having gall stones and cholecystectomy are more in both groups, may be due to excess fat, high cholesterol and excess estrogen from pregnancy, HRT or birth control pills (Wang S. et al Sarah S. Jackson et al [26,27].
Ghodki PS et al and others used dexmed or/and magnesium sulphate in their study and found significant reduction in requirement of dose of induction agent [13,28,29,30]. We also found the same in both groups in our study. Our study supports these studies.
Hemodynamic Parameters
Intra Group Comparison
Heart Rate: Comparison to baseline value at various time intervals, after bolus dose of drug in group M, there are significant drop (p value <0.05) in HR while in group D, drop in HR is highly significant (p value <0.001) throughout study except at postoperative 2nd hr which is significant. Devyani desai et al, Prithiv et al and others studied only bolus dose of dexmed and magnesium sulphate and our results are comparable with their results [19, 21,23,24].
Map: Comparison to baseline value, in group M, highly significant (<0.001) difference is found after study drug, after induction agent, after intubation, before pneumoperitoneum, at extubation while Throughout surgery MAP difference remains significant (p value<0.05).
In group D, highly significant (p value <0.001) drop is found after induction then throughout surgery and postoperatively except at 5, 15 and 30 min, it is significant (p value <0.05).
We have also calculated changes in HR % and MAP % from baseline values in both groups and found significant drop in HR % in group D, while reduction in HR % and MAP % are better controlled in group M.
Thus, in individual group comparison, both groups provided good hemodynamic stability with better controlled vitals in group M compared to group D. Devyani j desai et al and others compared dexmed and magnesium sulphate in and found better hemodynamic stability with magnesium. Our study supports their study [19,20,21,24].
Inter Group Comparison
We observed comparable (P value >0.05) baseline vitals in both groups.
Heart Rate: After giving study drugs, there is significant difference (P value <0.05) between two groups at all time intervals except after induction drug and 45 min after pneumoperitoneum it is highly significant (p value <0.001).
Blood Pressure: Regarding SBP and DBP, we found significant (p value <0.05) or highly significant (p value <0.001) difference at various time intervals between both study groups.
Map: is detected highly significant (p value <0.001) at all time intervals except after giving study drug, after intubation, before pneumoperitoneum, 1 and 5 min after pneumoperitoneum, MAP difference is significant (p value <0.05), with Group M patients have better control compared to group D patients.
Ramsay Sedation Score: is observed in both groups for two hours after extubation and it is comparable and nonsignificant with P value >0.05. In both groups patients are tranquil, oriented and responds to commands.
VAS score 3-4 is considered for 1st rescue analgesia dose. Inj. Diclofenac 75 mg is used for analgesia. In Group M, mean time for it is 70.03± 10.20 min and in Group D, 71.96± 12.46 min, which are comparable (P value >0.05).
At these times, VAS score increases and pts became anxious and slightly agitated. After giving analgesia, again pts were calm, and RSS score was 2 and VAS score decreases.
Total cumulative dose for analgesia in 24 hours is 257.14± 47.16 mg in group M and 242.5± 50.91 mg in group D, which is comparable (P value >0.05) in both groups.
Jung kyu park et al, Mohmed A. M et al [25] also used dexmed 1 µg/kg and found similar response after giving study drug. Azim et al [16] and others [31,19] used magnesium sulphate and found similar response. Our study supports their study.
We have not found any intraoperative or postoperative complication in Group M. In group D, we noticed intraoperative bradycardia in two patients at 30 min in one patient While at 45 min in other, which was treated with inj. Atropine 0.6 mg.
Islam M.M et al, Massad IM et al studied dexmed infusion and found decrease incidence of nausea and vomiting. In our study not, a single patient experienced nausea or vomiting postoperatively [12,32].
Devyani desai et al, Mohamed A et al [] nd many authors did comparison between dexmed and magnesium sulphate [4,14,15,17- 23, 25]. Chaithanya et al and others found magnesium sulphate equal with dexmed and Mohamed A et al [] found dexmed better than magnesium sulphate. Our results are similar to Devyani desai et al [], Prithiv et al [], Chandrakala et al [20], Ghodki PS et al [], in which both drugs are efficient to attenuate hemodynamic responses throughout surgery with better and sustained hemodynamic stability in group M [4,14,15,17,18, 19,21-25,].
Limitation of our study is small sample size. We have not taken surgery lasted >2 hours, we have not measured S. Catecholamines level and also drug concentration of Dexmed and Magnesium sulphate as we had not used infusion. We recommend future study with inclusion of these limitations.
Conclusion
Total 60 patients scheduled for laparoscopic cholecystectomy belonging to ASA grade 1 and 2, aged 18-60 years were included in our study. Patients were divided into two groups.
Group M (n=30): received inj. Magnesium sulphate 40 mg/kg in 20 ml NS i.v. over 15 min.
Group D (n=30): received inj. Dexmedetomidine 1 µg/kg in 20 ml NS i.v. over 15 min.
After completion of study drug vitals were noted.
All patients were pre-medicated and general anesthesia was given.
The induction of anesthesia was same in both groups and then HR, SBP, DBP, MAP was noted at different time intervals intra operatively and postoperatively for two hours.
We had compared vitals between both groups and also in individual group i.e. intra group vitals compared with baseline value among each group separately.
Patient demographic data compared, which is not significant in both group for age, sex, weight and height and ASA grade.
Duration of surgery is comparable in between both groups.
Total induction dose is decreased but comparable in both groups.
Mean HR and Mean MAP are significantly decreased (P Value<0.05) compared to baseline in individual group comparison at every time intervals in both groups.
All vital parameters HR, SBP, DBP and MAP were significantly decreased at every time intervals in both group M and group D with better hemodynamic stability related to MAP in group M.
Patients were assessed postoperatively two hours for sedation by RSS and analgesia by VAS score which were comparable.
Time for 1st rescue analgesia was noted and comparable in both groups.
Total cumulative analgesia for 24 hours is noted and is comparable in both study groups.
No major complication was noted intra or post operatively in group M. Two patients in group D had bradycardia intra operatively which was not much distressing and was treated by inj. Atropine i.v once. On the basis of our study we concluded that both the study drugs decrease vitals and they were within a physiological limit.
Magnesium sulphate provides a reliable and effective control over all vital parameters and better hemodynamic stability intraoperatively and postoperatively with comparable sedation and analgesic requirement postoperatively to dexmedetomidine [25-37].
References
- Bhana, N., Goa, K. L., & McClellan, K. J. (2000). Dexmedetomidine. Drugs, 59, 263-268.
- Kallio, A., Scheinin, M., Koulu, M., Ponkilainen, R., Ruskoaho, H., Viinamäki, O., & Scheinin, H. (1989). Effects of dexmedetomidine, a selective α2â?adrenoceptor agonist, on hemodynamic control mechanisms. Clinical Pharmacology & Therapeutics, 46(1), 33-42.
- Lee, S. (2019). Dexmedetomidine: present and future directions. Korean journal of anesthesiology, 72(4), 323-330.
- Mohan, A., Saha, G. A. (2023). Comparative Study of the Efficacy of Intravenous Magnesium Sulphate and Intravenous Dexmedetomidine in Attenuating Haemodynamic Response to Laryngoscopy and Endotracheal Intubation. Indian J Clin Anaesth;10(4):351-357.
- Pertovaara, A., Haapalinna, A., Sirviö, J., & Virtanen, R. (2005). Pharmacological properties, central nervous system effects, and potential therapeutic applications of atipamezole, a selective α2â?adrenoceptor antagonist. CNS drug reviews, 11(3), 273-288.
- Fawcett, W. J., Haxby, E. J., & Male, D. A. (1999). Magnesium: physiology and pharmacology. British journal of anaesthesia, 83(2), 302-320.
- Altura, B. M., & Altura, B. T. (1978). Magnesium and vascular tone and reactivity. Journal of Vascular Research, 15(1-3), 5-16.
- Stoelting, R. K., & Hillier, S. C. (2012). Pharmacology and physiology in anesthetic practice. Lippincott Williams & Wilkins.
- Walder, A. D., & Aitkenhead, A. R. (1997). Role of vasopressin in the haemodynamic response to laparoscopic cholecystectomy. British journal of anaesthesia, 78(3), 264- 266.
- O'leary, E., Hubbard, K., Tormey, W., & Cunningham, A.J. (1996). Laparoscopic cholecystectomy: haemodynamic and neuroendocrine responses after pneumoperitoneum and changes in position. British journal of anaesthesia, 76(5), 640-644.
- Jee, D., Lee, D., Yun, S., & Lee, C. (2009). Magnesium sulphate attenuates arterial pressure increase during laparoscopic cholecystectomy. British journal of anaesthesia, 103(4), 484-489.
- Massad, I. M., Mohsen, W. A., Basha, A. S., Al-Zaben, K. R., Al-Mustafa, M. M., & Alghanem, S. M. (2009). A balanced anesthesia with dexmedetomidine decreases postoperative nausea and vomiting after laparoscopic surgery. Saudi Med J, 30(12), 1537-1541.
- Ghodki, P. S., Thombre, S. K., Sardesai, S. P., & Harnagle,K. D. (2012). Dexmedetomidine as an anesthetic adjuvant in laparoscopic surgery: An observational study using entropy monitoring. Journal of Anaesthesiology Clinical Pharmacology, 28(3), 334-338.
- Chaithanya, K., Vaddineni, J., Reddy, N., Gandra, S., Kumar, C., Rao, V., & Sekhar, V. (2014). A comparative study between IV 50% magnesium sulphate and dexmedetomidine for attenuation of cardiovascular stress response during laryngoscopy and endotracheal intubation. Journal of Evolution of Medical and Dental Sciences, 3(32), 8741-8750.
- Zarif, P., Abdelaal, A., Mahmoud, A., Abdelhaq, M., Mikhail,H. M. S. Et al. (2016). Dexmedetomidine versus Magnesium Sulphate as an adjunct during anaesthesia for laparoscopic colectomy. Anesth Res Prac 1-7.
- Honarmand, A., Safavi, M., Badiei, S., & Daftari-Fard, N. (2015). Different doses of intravenous Magnesium sulfate on cardiovascular changes following the laryngoscopy and tracheal intubation: A double-blind randomized controlled trial. Journal of research in pharmacy practice, 4(2), 79-84.
- Mahajan, L., Kaur, M., Gupta, R., Aujla, K. S., Singh, A., & Kaur, A. (2018). Attenuation of the pressor responses to laryngoscopy and endotracheal intubation with intravenous dexmedetomidine versus magnesium sulphate under bispectral index-controlled anaesthesia: A placebo-controlled prospective randomised trial. Indian journal of anaesthesia, 62(5), 337-343.
- Balata, A. A. H., Abdel Latif, H. K., Waly, S. H., & Mohamed, A.B. (2018). DEXMEDETOMIDINE VERSUS MAGNESIUM SULPHATE OR LIDOCAINE FOR BLUNTING STRESS RESPONSE TO DIRECT LARYNGOSCOPY AND ENDOTRACHEAL INTUBATION IN ABDOMINALSURGERIES. Zagazig University Medical Journal, 24(6), 492-500.
- Desai, D. J., Shah, S., & Upadhyay, M. R. (2019). A Prospective Randomized Comparative Study of Intravenous Dexmedetomidine versus Magnesium Sulphate as an Adjunct during Anesthesia for Laparoscopic Cholecystectomy. J Anesth Clin Res, 10(930), 2.
- Mohan, A., & Saha, G. (2023). A comparative study of the efficacy of intravenous magnesium sulphate and intravenous dexmedetomidine in attenuating haemodynamic response to laryngoscopy and endotracheal intubation. Indian Journal of Clinical Anaesthesia, 10(4), 351-357.
- Arora, V. A. (2020). Comparative Study between Magnesium Sulphate and Dexmedetomidine for Attenuation of Vasopressor Stress Response During Laryngoscopy and Endotracheal Intubation. Ann. Int. Med. Den. Res. 2020; 6(6).
- Pathak, Deba. & Singha, E. (2020). “A COMPARATIVE STUDY BETWEEN INTRAVENOUS FENTANYL AND MAGNESIUM SULFATE IN ATTENUATION OF CARDIOVASCULAR RESPONSE TO LARYNGOSCOPY AND ENDOTRACHEAL INTUBATION”. PARIPEX INDIAN JOURNAL OF RESEARCH. 148-150.
- Ghodki, P. S., Sawle, V. M. (2020) Comparative Study Between Magnesium Sulphate and Dexmedetomidine For Attenuation of Vasopressor Stress Response During Laryngoscopy and Endotracheal Intubation. International Journal of Medical Anesthesiology 3(3): 63-67.
- Rishardhan, P., Chavan, R. V., Mina, B. N., Manisha, C. (2021). Prithiv Rishardhan, Rashmee Vijay Chavan, Mina Bhaarathi N, C Manisha. Comparative study of magnesium sulphate and dexmedetomidine in the attenuation of pressor response to intubation and on intraoperative haemodynamic parameters in laparoscopic cholecystectomy.International Journal of Anesthesiology 19(3):105-110.
- Menshawi, M. A., Fahim, H. M. (2022). Dexmedetomidine Versus Magnesium Sulfate asAnAdjunct to GeneralAnesthesia in Patients Undergoing Video Assisted Thoracoscopy. Journal of Anesthesiology (2022)14: 11.
- Wang, S., Wang, Y., Xu, J., & Chen, Y. (2017). Is the oral contraceptive or hormone replacement therapy a risk factor for cholelithiasis: A systematic review and meta-analysis. Medicine, 96(14), e6556.
- Jackson, S. S., Pfeiffer, R. M., Gabbi, C., Anderson, L., Gadalla, S. M., & Koshiol, J. (2022). Menopausal hormone therapy and risk of biliary tract cancers. Hepatology, 75(2), 309-321.
- Ray, M., Bhattacharjee, D. P., Hajra, B., Pal, R., & Chatterjee,N. (2010). Effect of clonidine and magnesium sulphate on anaesthetic consumption, haemodynamics and postoperative recovery: A comparative study. Indian Journal of anaesthesia, 54(2), 137-141.
- Kalaskar, V. P., Ruparel, D. H., & Wakode, R. P. (2021). Effects of dexmedetomidine infusion in low dose on dose reduction of propofol, intraoperative hemodynamics, and postoperative analgesia in patients undergoing laparoscopic cholecystectomy. Anesthesia Essays and Researches, 15(4), 391-394.
- Khare, A., Sharma, S. P., Deganwa, M. L., Sharma, M., & Gill, N. (2017). Effects of dexmedetomidine on intraoperative hemodynamics and propofol requirement in patients undergoing laparoscopic cholecystectomy. Anesthesia Essays and Researches, 11(4), 1040-1045.
- Kamble, S. P., Bevinaguddaiah, Y., Nagaraja, D. C., Pujar,V. S., & Anandaswamy, T. C. (2017). Effect of magnesium sulfate and clonidine in attenuating hemodynamic response to pneumoperitoneum in laparoscopic cholecystectomy. Anesthesia Essays and Researches, 11(1), 67-71.
- Massad, I. M., Mohsen, W. A., Basha, A. S., Al-Zaben, K. R., Al-Mustafa, M. M., & Alghanem, S. M. (2009). A balanced anesthesia with dexmedetomidine decreases postoperative nausea and vomiting after laparoscopic surgery. Saudi Med J, 30(12), 1537-1541.
- Wahba, R. W., Béïque, F., & Kleiman, S. J. (1995). Cardiopulmonary function and laparoscopic cholecystectomy. Canadian journal of anaesthesia, 42, 51-63.
- Miller’s Anesthesia 7th edition by Ronald D. Miller, 8th edition, volume 1, chapter-30, 857-858.
- Fasil, F., Saundattikar, G., & Jawale, R. H. (2022). Study of hemodynamic effects of preoperative single-bolus dexmedetomidine in elective laparoscopic surgeries. Journal of Pharmacology and Pharmacotherapeutics, 13(1), 85-91.
- James, M. F., Beer, R. E., & Esser, J. D. (1989). Intravenous magnesium sulfate inhibits catecholamine release associated with tracheal intubation. Anesthesia & Analgesia, 68(6), 772-776.
- Park, J. K., Cheong, S. H., Lee, K. M., Lim, S. H., Lee, J.H., Cho, K., ... & Kim, H. T. (2012). Does dexmedetomidine reduce postoperative pain after laparoscopic cholecystectomy with multimodal analgesia?. Korean journal of anesthesiology, 63(5), 436-440.



